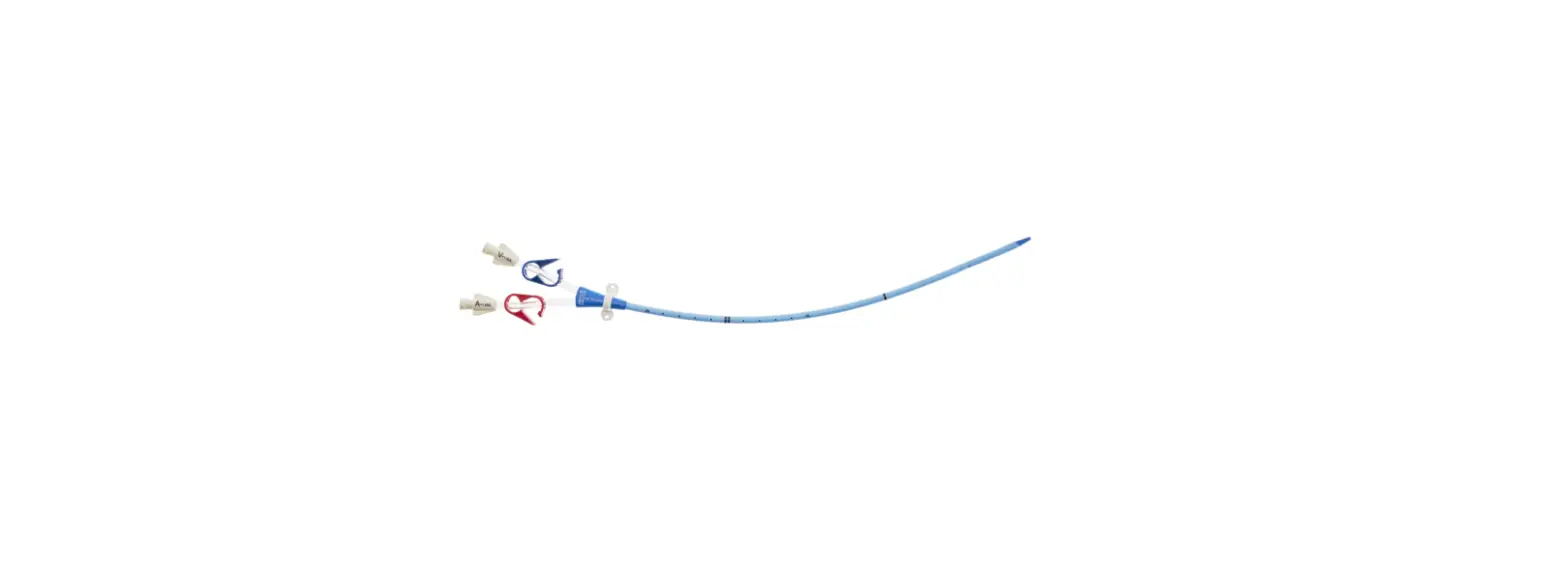
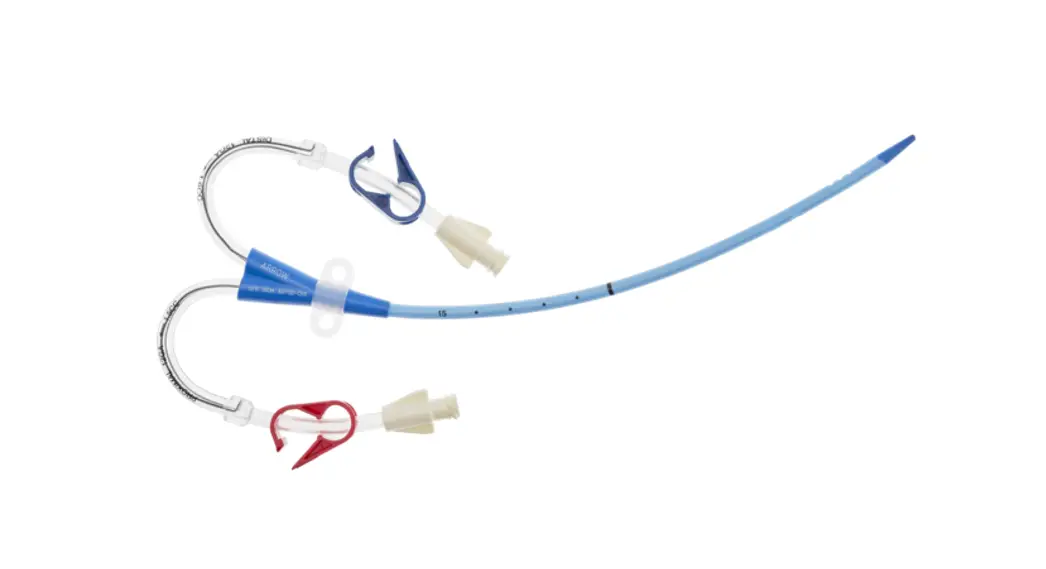
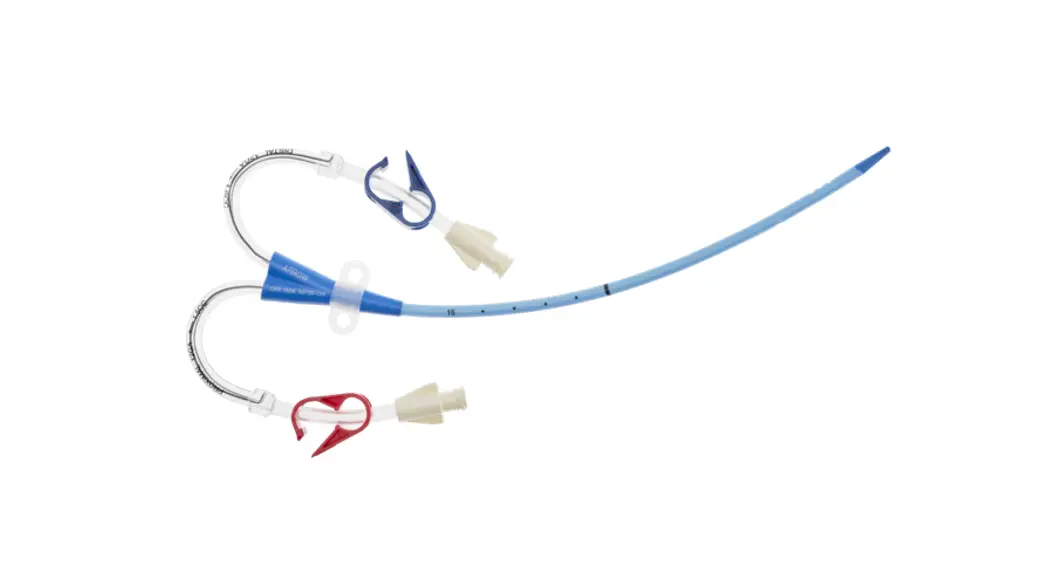
CDC-15123-XPCN1A Pressure Injectable Three-Lumen Hemodialysis Catheterization for High Volume Infusions
Product Information
- Product Name: CDC-15123-XPCN1A
- Catheter Size: 12 3 Fr.
- Lumen: Three-Lumen
- Catheter Length: 20cm
- Spring-Wire Guide Diameter: 0.035 inch
- Intended Use: Pressure Injectable Three-Lumen Hemodialysis
Catheterization for High Volume Infusions - Contents:
- Catheter: 18 Ga. x 2-1/2 (6.35 cm) Radiopaque over 20 Ga. RW
Introducer Needle - Raulerson Spring-Wire Introduction Syringe
- Injection Needle: SafetyGlideTM1 22 Ga. x 1-1/2 (3.81 cm) and 5
mL Luer-Slip - Hi-Lite OrangeTM Tint
- Catheter: 18 Ga. x 2-1/2 (6.35 cm) Radiopaque over 20 Ga. RW
- Trademarks: SafetyGlideTM1 is a trademark of Becton, Dickinson
and Company. Hi-Lite OrangeTM is a registered trademark of
CareFusion or one of its subsidiaries. - Contraindications: The Multi-Lumen Acute Hemodialysis Catheter
(AHDC) is not designed for long-term (30 days) hemodialysis or for
insertion into thrombosed vessels. - LBL068959 R00 (2023-04)
Product Usage Instructions
To use the CDC-15123-XPCN1A Hemodialysis Catheter:
- Ensure that the catheter is sterile and undamaged before
use. - Prepare the insertion site according to standard aseptic
techniques. - Locate the vessel for catheterization.
- Insert the Radiopaque over 20 Ga. RW Introducer Needle into the
vessel using the Raulerson Spring-Wire Introduction Syringe. - Advance the catheter through the introducer needle into the
vessel. - Remove the introducer needle while keeping the catheter in
place. - Connect the catheter to the appropriate infusion or
hemodialysis system. - Monitor the patient and follow the recommended protocols for
high volume infusions. - After use, dispose of the catheter and related materials
according to proper medical waste disposal guidelines.
Note: This catheter is not suitable for long-term hemodialysis
or insertion into thrombosed vessels. Please consult a healthcare
professional for alternative options in those cases.
6
NO
NO
CT
CT
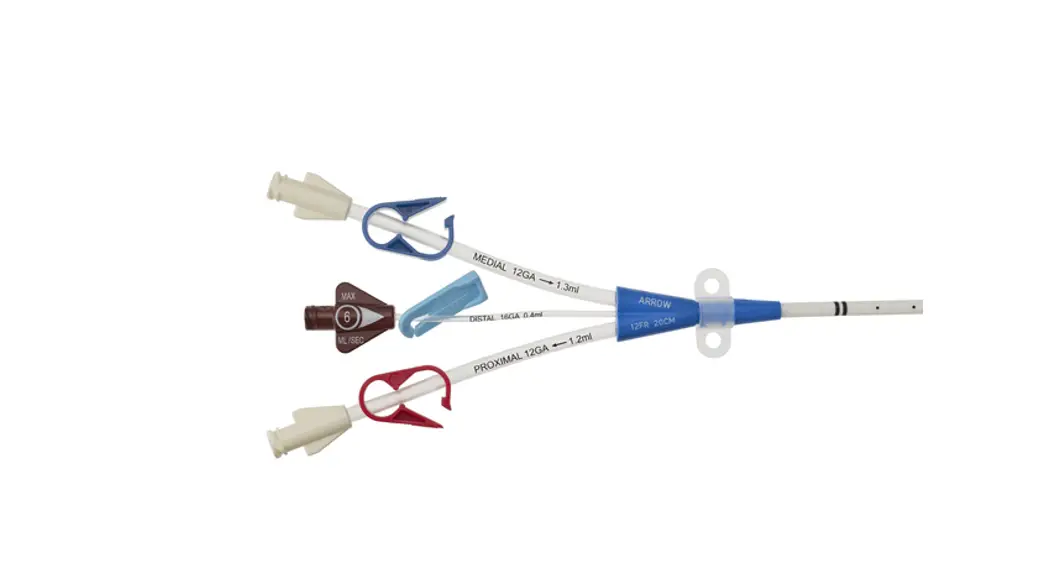
CDC-15123-XPCN1A
12 3 Fr.
Lumen
20cm catheter length
.035 inch dia. spring-wire guide
Pressure Injectable Three-Lumen Hemodialysis Catheterization for High Volume Infusions
Contents:
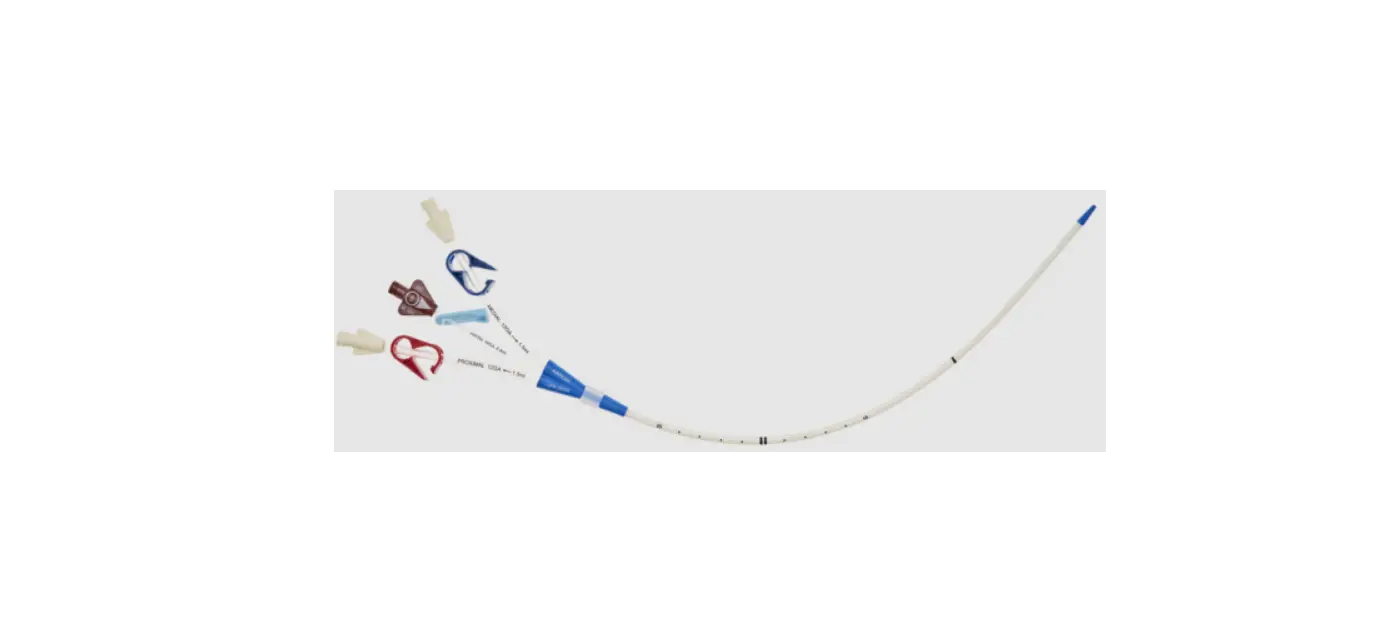
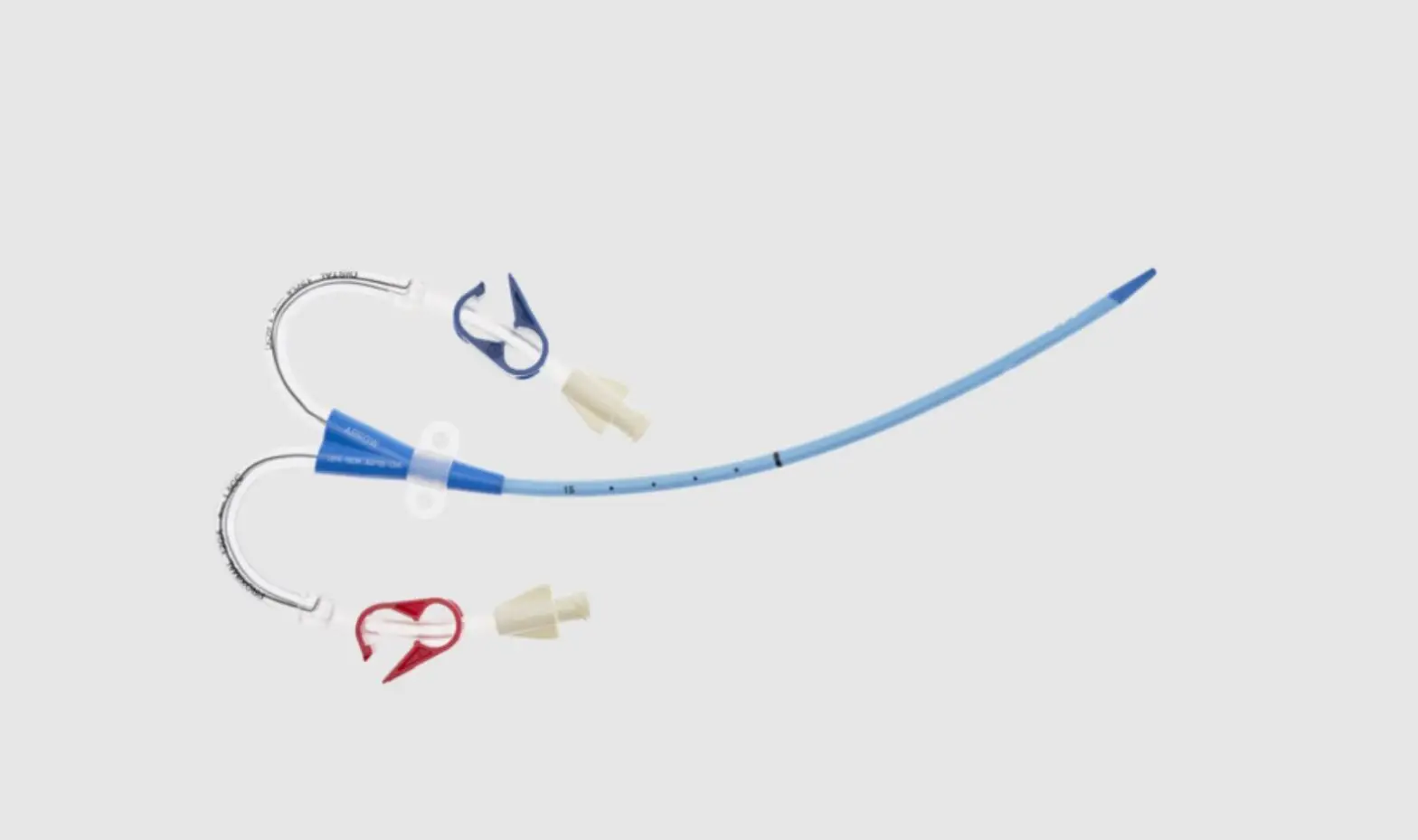
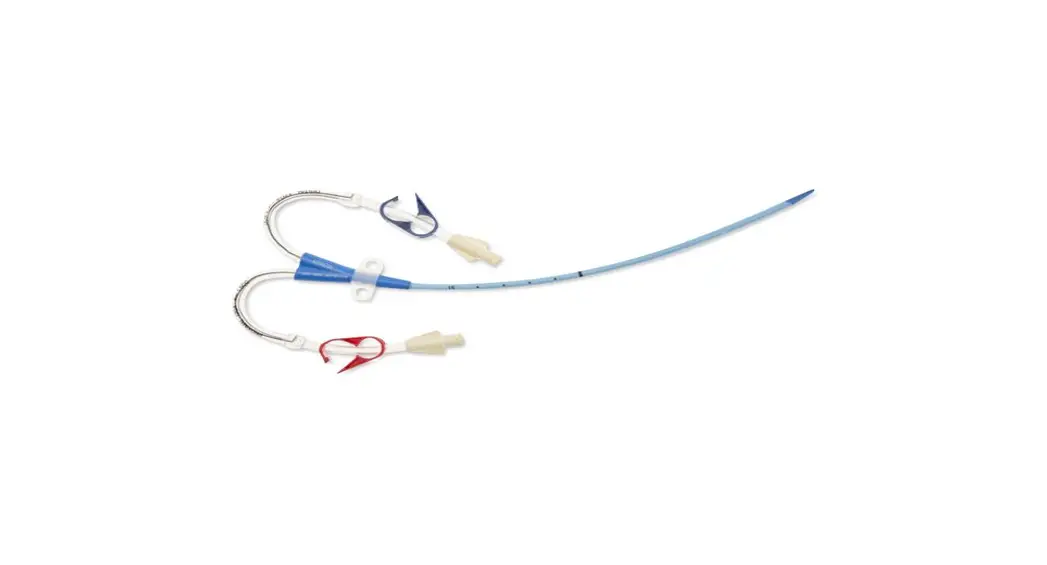
1: Three-Lumen Catheter: 12 Fr. (4.06 mm OD) x 20 cm Pressure Injectable Radiopaque Polyurethane with Blue FlexTip®, Extension Line Clamps
1: Spring-Wire Guide, Marked: .035″ (0.89 mm) dia. x 23-5/8″ (60 cm) (Straight Soft Tip on One End – “J” Tip on Other) with Arrow® GlideWheelTM Wire Advancer
1: Catheter: 18 Ga. x 2-1/2″ (6.35 cm) Radiopaque over 20 Ga. RW Introducer Needle
1: Injection Needle: SafetyGlideTM1 25 Ga. x 1″ (2.54 cm) 1: Blunt Fill Needle: 18 Ga. x 1-1/2″ (3.81 cm) 1: Pressure Transduction Probe 1: Introducer Needle: Echogenic 18 Ga. x 2-1/2″ (6.35 cm) XTW and 5 mL Arrow®
Raulerson Spring-Wire Introduction Syringe 1: Injection Needle: SafetyGlideTM1 22 Ga. x 1-1/2″ (3.81 cm) and 5 mL Luer-Slip
Syringe 1: Syringe: 3 mL Luer-Lock 1: Tissue Dilator: 10 Fr. (3.5 mm) x 10.2 cm 1: Tissue Dilator: 12 Fr. (4.0 mm) x 14 cm 3: 10 mL Luer-Lock Pre-Filled Saline Syringe 1: 5 mL 1% Lidocaine HCl Solution and Alcohol Prep 2: 3 mL Applicator 2% CHG and 70% IPA ChloraPrep®2 One-Step Solution with
Hi-Lite OrangeTM Tint
1: SharpsAway® II Locking Disposal Cup 1: SharpsAway® Disposal Cup 1: Maximal Barrier DrapeTM with 4″ fenestration 1: Towel 1: Needle Holder 1: Safety Scalpel: #11 1: Checklist/CLIP Sheet 1: Sterile Procedure Sign 1: Medication Label: 1% Lidocaine 2: Gauze Pad: 2″ x 2″ (5 cm x 5 cm) 5: Gauze Pad: 4″ x 4″ (10 cm x 10 cm) 1: Surgical Apparel: Impervious Gown 1: Dressing: Tegaderm®3 CHG 3-1/2″ x 4-1/2″ (8.5 cm x 11.5 cm) 1: Transducer Cover: 14 cm x 147 cm 1: Surgical Apparel: Mask with Eye Shield 1: Surgical Apparel: Bouffant Cap 1: Suture: 3-0 Silk with Curved Needle 1: Tubing±: 20″ (50 cm) Extension 1: HemoHopper® Fluid Receptacle
1A trademark of Becton, Dickinson and Company. 2A registered trademark of CareFusion or one of its subsidiaries.
±Precaution: Contains Phthalate: DEHP. Warning: The results of certain animal experiments have shown phthalates to be potentially toxic to reproduction. Proceeding from the present state of scientific knowledge, risks for male premature infants cannot be excluded in the case of long-term exposure or application. Medical products containing phthalates should be used only temporarily with pregnant women, nursing mothers, babies and infants. Not made with natural rubber latex. Store between 20 25°C (68 77°F). Arrow International provides the enclosed medication label(s) for your convenience. Please ensure that these labels are applied to the correct syringe and corresponding medication.
Contraindications: The Multi-Lumen Acute Hemodialysis Catheter (AHDC) is not designed for long-term ( 30 days) hemodialysis or for insertion into thrombosed vessels.
3A registered trademark of 3M Company.
LBL068959 R00 (2023-04)