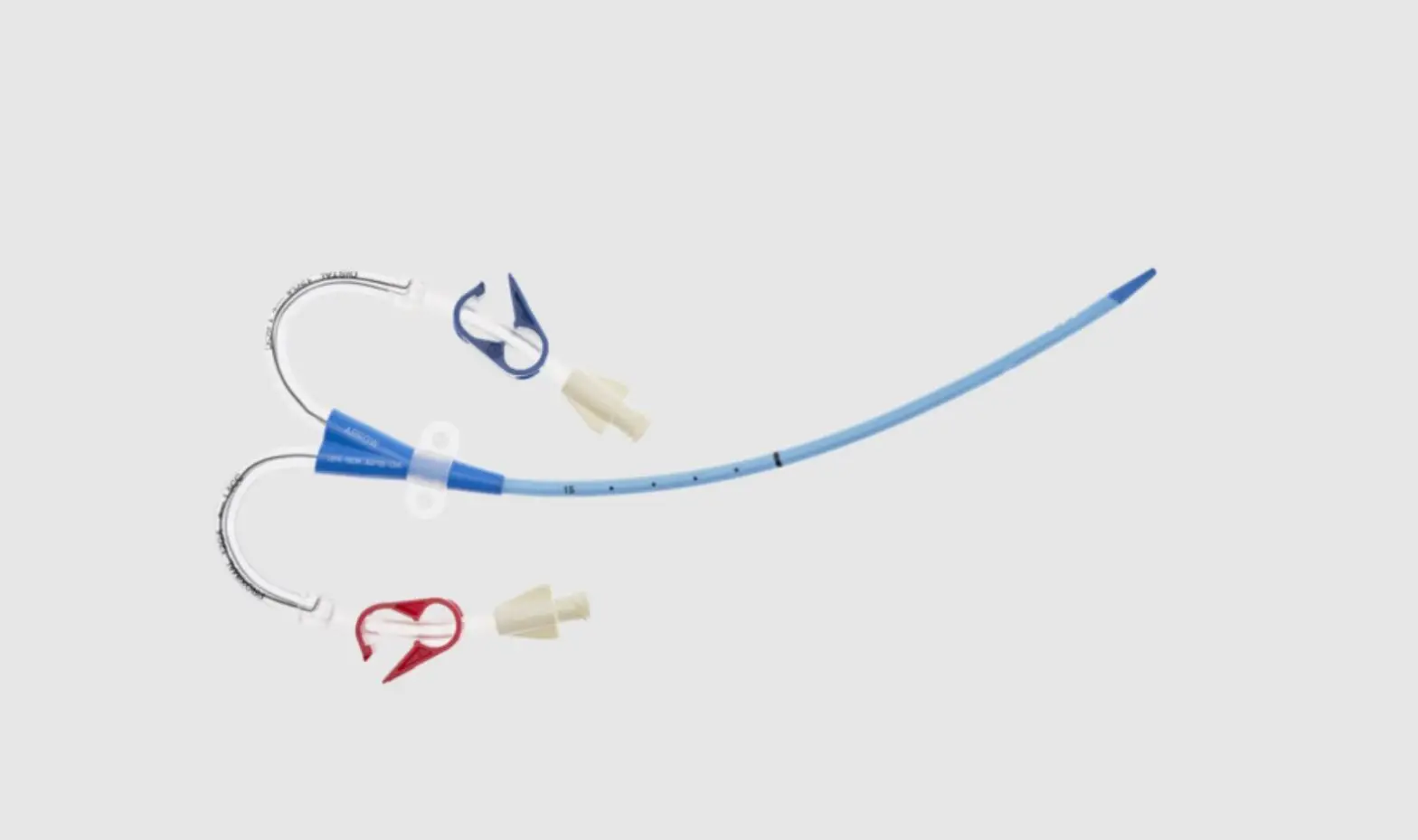
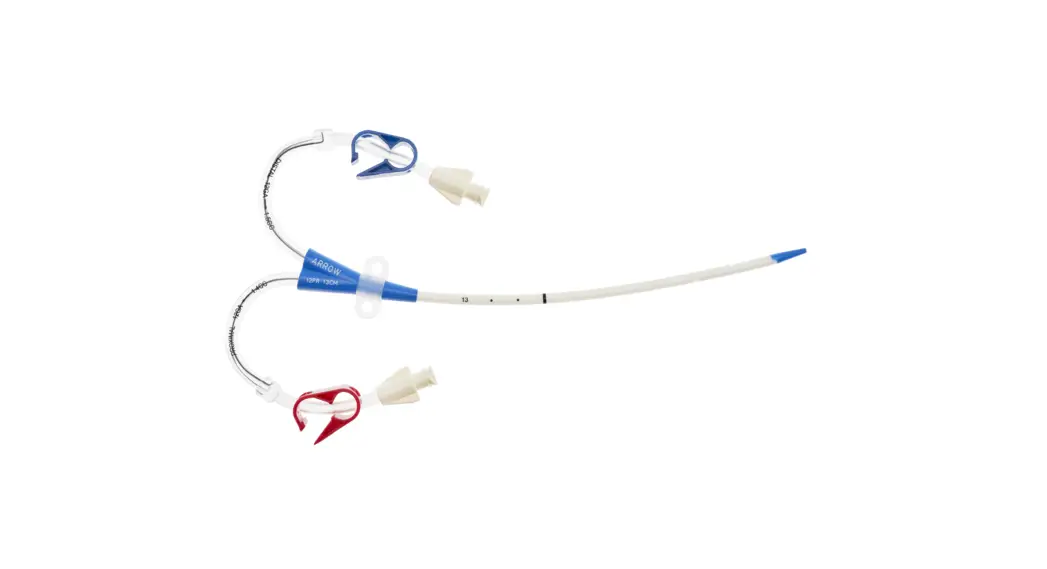
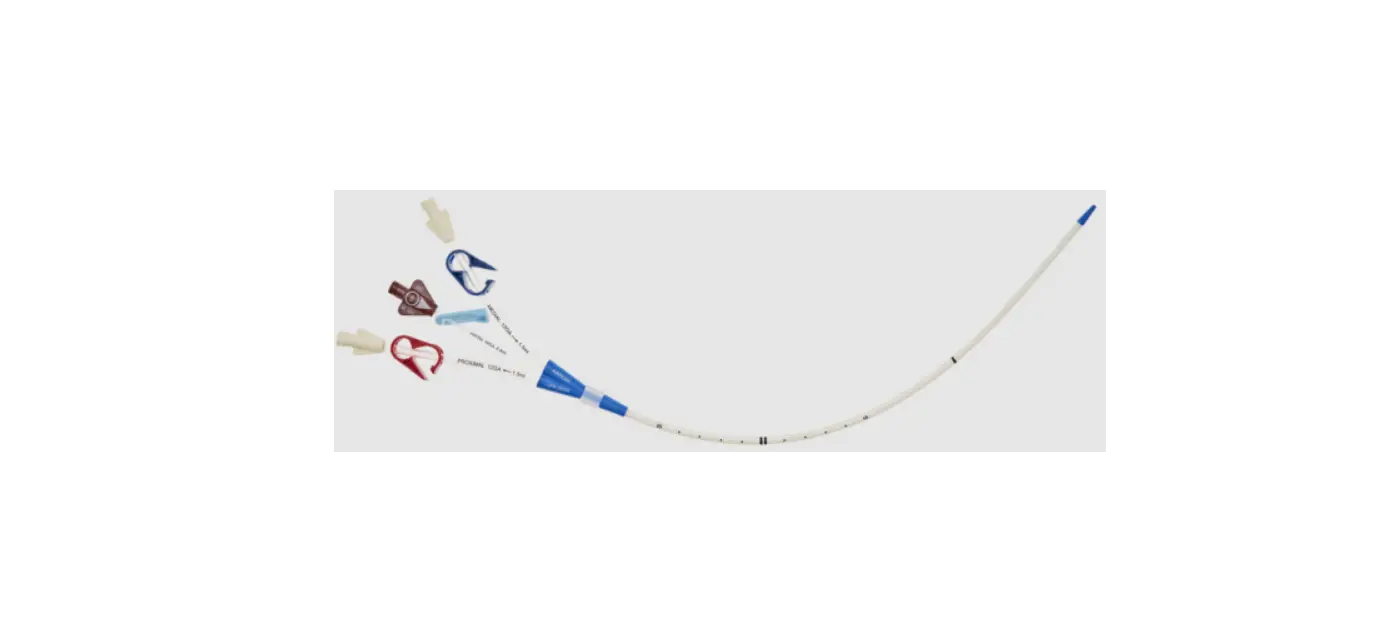
CS-22142-F Arrowg+ard Blue Two-Lumen Hemodialysis Catheter for High Volume Infusions
Contents
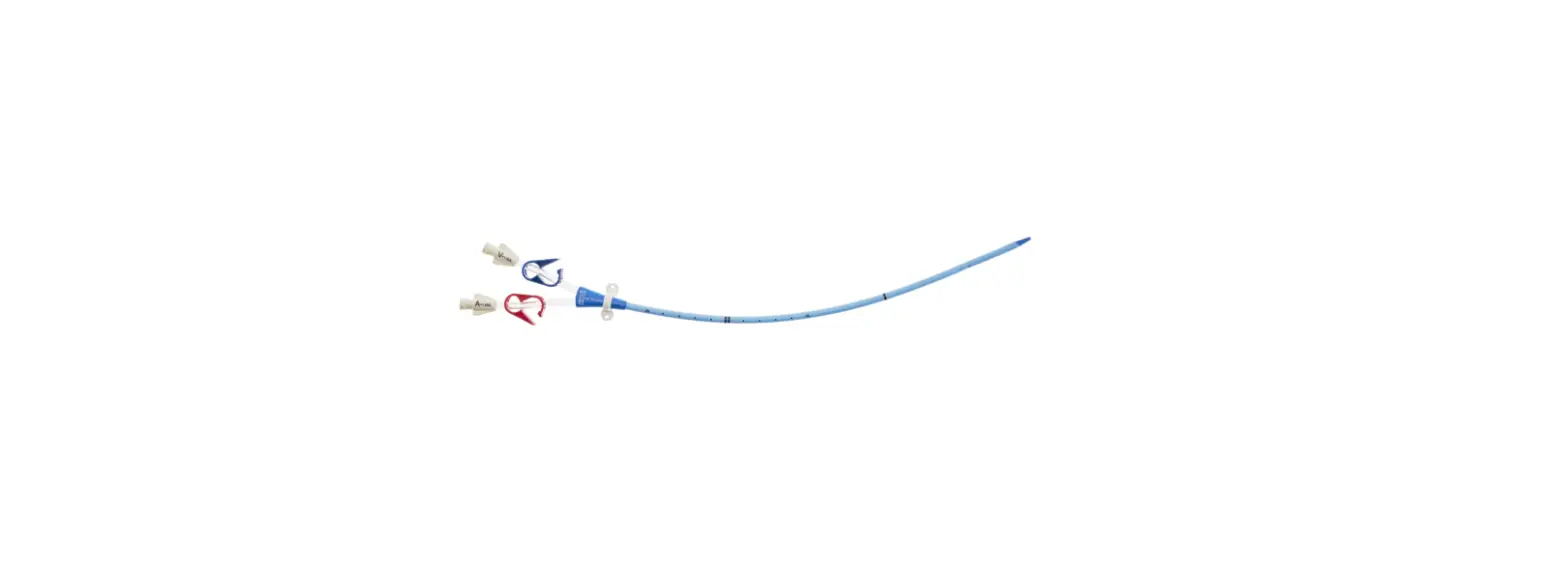
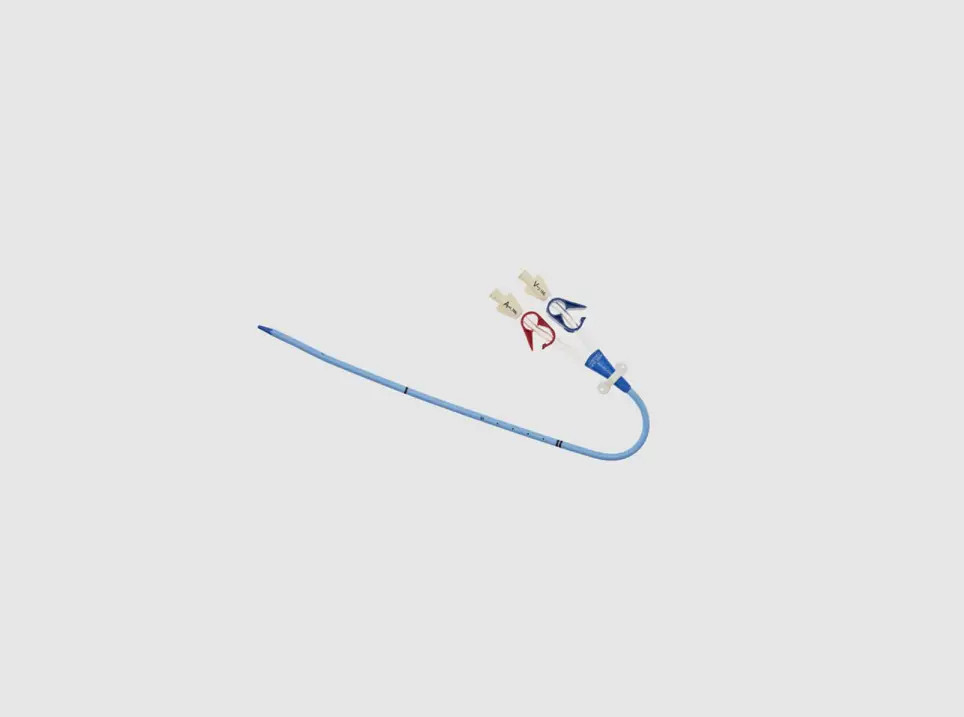
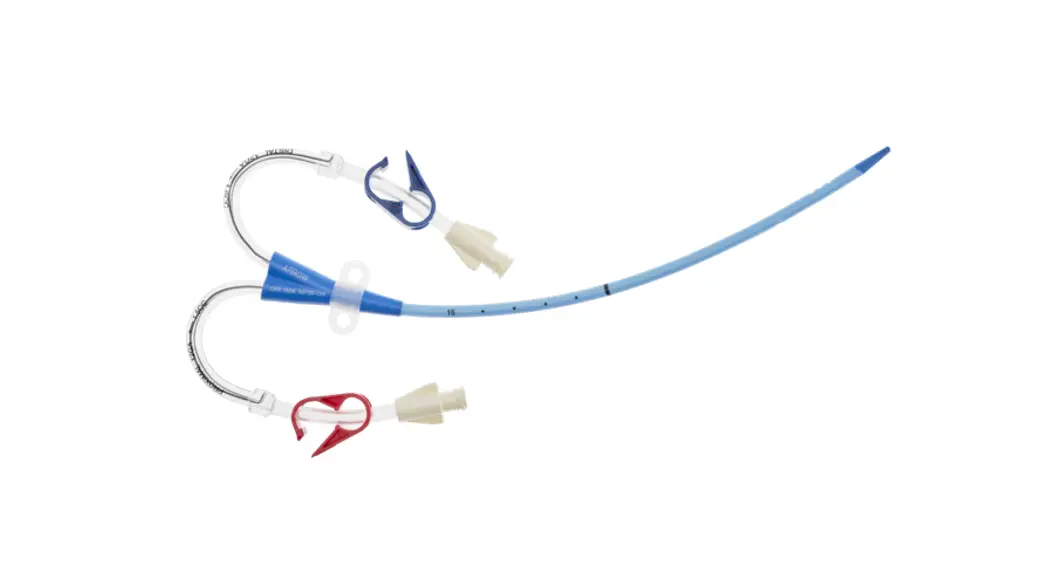
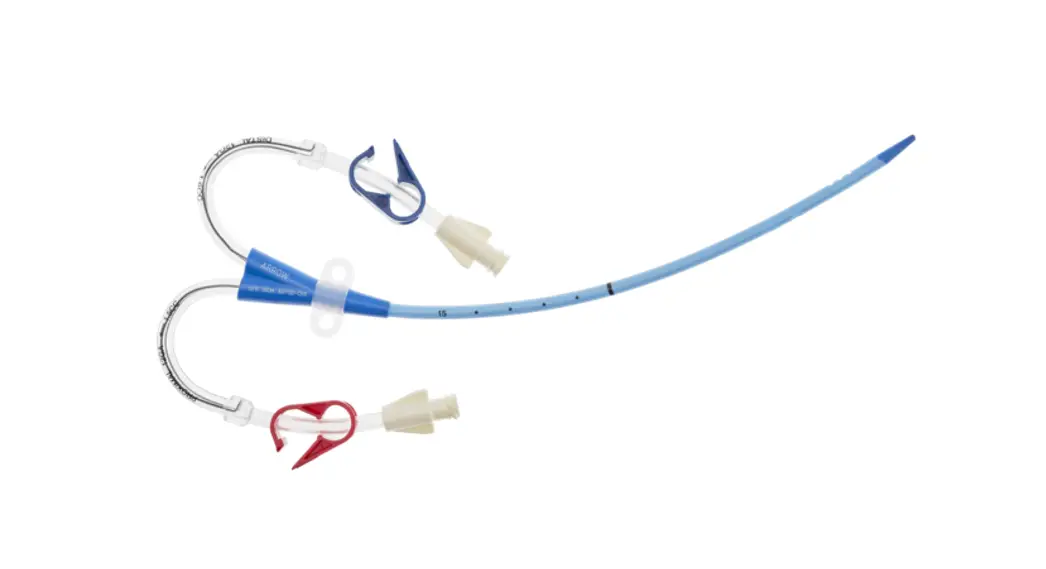
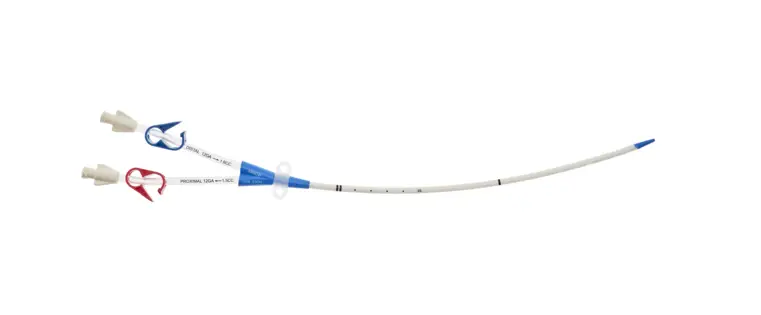
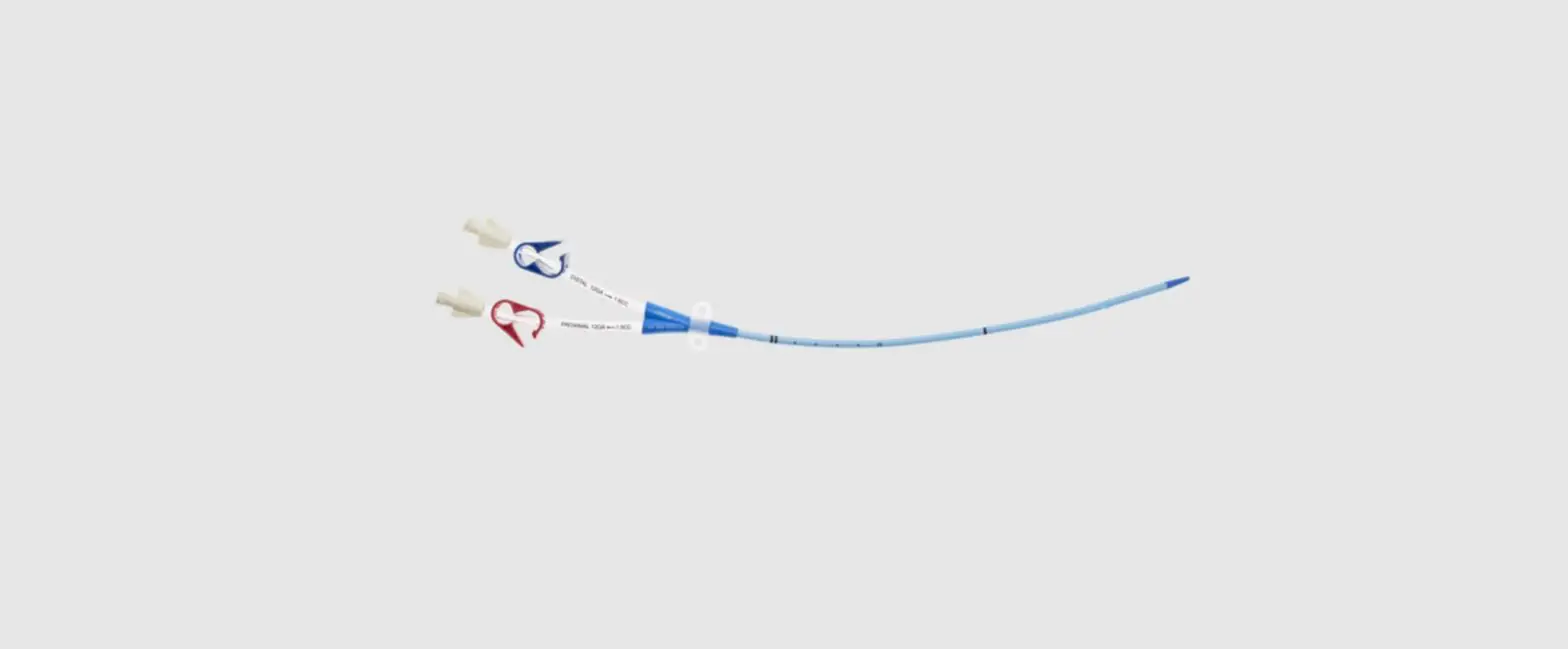
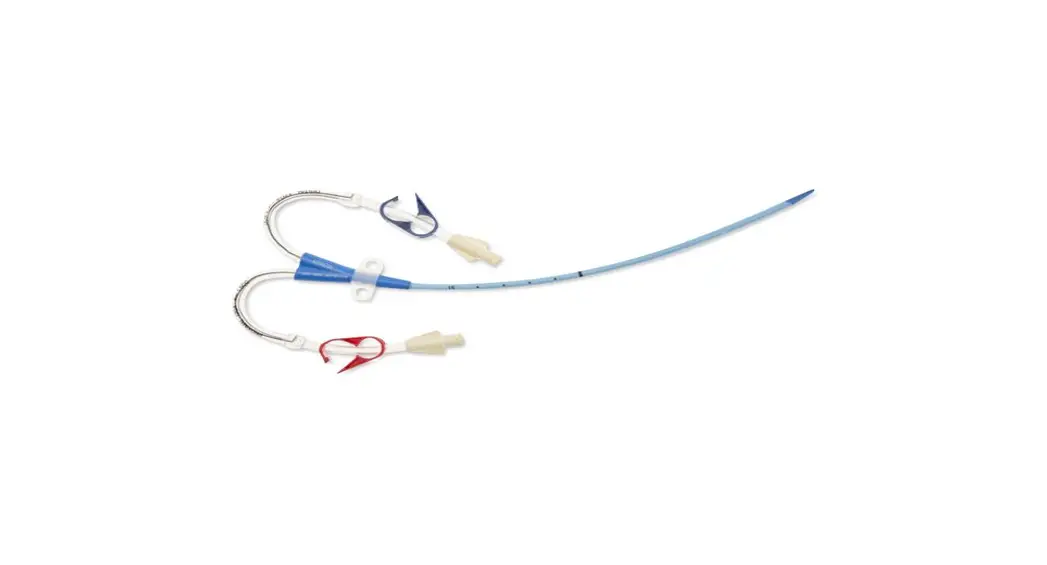
- Two-Lumen Catheter: 14 Fr. (4.72 mm OD) x 15 cm Radiopaque Polyurethane with Blue FlexTip®, ARROWg+ard® Antimicrobial Surface Treatment, Extension Line Clamps
- Spring-Wire Guide, Marked: .038″ (0.97 mm) dia. x 27-1/2″ (70 cm) (Straight Soft Tip on One End – “J” Tip on Other) with Arrow Advancer
- Catheter: 16 Ga. x 1-7/8″ (4.8 cm) Radiopaque over 19 Ga. TW Introducer Needle
- Pressure Transduction Probe
- Introducer Needle: 18 Ga. x 2-1/2″ (6.35 cm) XTW and 5 mL Arrow® Raulerson Spring-
Wire Introduction Syringe - Tissue Dilator: 14 Fr. (4.7 mm) x 14 cm
- Tissue Dilator: 16 Fr. (5.3 mm) x 11.4 cm
- Dust Cap: Non-Vented
- Drape: 24″ x 36″ (61 cm x 91 cm)
- Scalpel: #11 All components are CE 2797 unless otherwise noted.
Rx only
Warning: Read all package insert warnings, precautions, and instructions prior to use. Failure to do so may result in severe patient injury or death. www.teleflex.com/IFU Not made with natural rubber latex. Store below 25°C (77°F). Avoid excessive heat above 40°C (104°F). Fluid path components are non-pyrogenic. Contraindications: The Arrow large-bore two-lumen catheter is not designed for long-term (≥ 30 days) hemodialysis or for use in patients with thrombosed vessels. The Arrowg+ard Blue antimicrobial catheter is contraindicated for patients with known hypersensitivity to chlorhexidine, silver sulfadiazine, and/or sulfa drugs.
- Priming volumes are approximate and are done without accessories. †† Venous Pressure settings are approximate and were determined by varying the blood flow rates from 250 to 400 mL/min in an in vitro experiment using blood analog solution.
EU Authorized Representative and
Importer: Teleflex Medical IDA Business and Technology Park Dublin Road, Athlone Co. Westmeath, Ireland
Contents: Arrow International LLC Subsidiary of Teleflex Incorporated 3015 Carrington Mill Blvd. Morrisville, NC 27560 USA