GIMA 0767/ B Light Alloy Walking Stick

LIGHT ALLOY WALKING STICK
All serious accidents concerning the medical device sup-plied by us must be reported to the manufacturer and com-petent authority of the member state where your registered office is located.
SAFETY WARNINGS
This stick is not a tetrapod walking stick! It must be used like any walking stick and must not be intended as an aid designed to increase the patient’s stability. The function of the 4 feet is a convenience that serves only to allow the stick to stand up by itself when it is not being used. Never use a damaged device
INSTRUCTIONS FOR USE
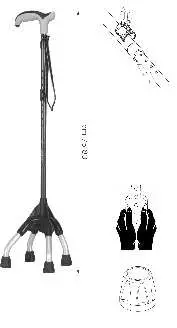
Height adjustment procedure. (Fig.1)
- Disengage the locking spring. (See Fig. 2)
- Adjust the height by removing the telescopic tubes.
- Reposition the spring clip.
ATTENTION
Do not use tools to remove the fixing spring as it could get dam-aged. The spring is hard to remove for safety reasons. (Fig. 2)
CLEANING AND MAINTENANCE
Cleaning: regularly clean the handle with neutral soap; do not use alcohol or solvents.
Tip: The tip resting on the ground (Fig. 3) is subject to wear; we recommend replacing it periodically using original OPO spare parts.
GIMA WARRANTY TERMS
The Gima 12-month standard B2B warranty applies.
 Peso Max. utente Max. user weight Poids maximal de l’utilisateur Peso Máx. usuario 100 kg
Peso Max. utente Max. user weight Poids maximal de l’utilisateur Peso Máx. usuario 100 kg
 Medical Device compliant with Regulation (EU) 2017/745
Medical Device compliant with Regulation (EU) 2017/745 Caution: read instructions (warnings) carefully
Caution: read instructions (warnings) carefully Keep in a cool, dry place
Keep in a cool, dry place Product code
Product code Lot number
Lot number Date of manufacture
Date of manufacture Consult instructions for use
Consult instructions for use Manufacturer
Manufacturer Medical Device
Medical Device
Officina Prodotti Ortopedici S.r.l.
Via Bellini 33, Rastignano (Bologna – Italy)
Made in Italy


















