DESCRIPTION OF SYMBOLS USED
| WARNING: Important information |  |
| Please wear gloves |  |
| Visual examination | |
| Name of manufacturer |  |
| medical device complying with European Directive 93/42/CEE |  |
| General indications with no danger to people or objects |  |
| Please read this ! |  |
| Medical device | MD |
| Thermo-disinfectable | |
| Sterilisable up to 135°C | |
| Single-Use |
AREA OF APPLICATION
The prosthetic wrench (MD) is intended for dental applications in the fields of surgery and implant prosthetics. The INCC ratchet wrench is used for the rotation of tools with a “wrench” type connection and to apply the torques required for the execution of clinical procedures. Any form of incorrect use is prohibited and may be dangerous.
For this MD, we applied Community Directive 93/42/CEE (amended 2007/47), transposed by the French regulations on medical equipment (Official Bulletin – L169 July 12, 1993).
The MD must be used by someone with experience of dental medicine, especially in implantology and in prosthesis on implants, for the application described and in compliance with the provisions in effect in relation to the prevention of accidents at work, protection of employment and the instructions for use.
In accordance with these provisions, the user must:
 only use working instruments without defects,
only use working instruments without defects,- observe the principle of correct use,
- protect themselves and the patient or third party against any danger
- prevent any contamination by the product.
GENERAL SAFETY INFORMATION
Before use, check that the MD is not damaged and that no part is missing.![]() After insertion of a rotary instrument, check that it is correctly held by a slight rotary movement. Do not handle the rotary instrument during operation. Before using the prosthetic dynamometrical wrench, we strongly advise to test it outside of the patient’s mouth.
After insertion of a rotary instrument, check that it is correctly held by a slight rotary movement. Do not handle the rotary instrument during operation. Before using the prosthetic dynamometrical wrench, we strongly advise to test it outside of the patient’s mouth. In the event of malfunction or damage, immediately stop using the instrument and inform your approved distributor or the manufacturer.
In the event of malfunction or damage, immediately stop using the instrument and inform your approved distributor or the manufacturer. In the event of any questions about the approved distributor or the manufacturer.
In the event of any questions about the approved distributor or the manufacturer.
TECHNICAL SPECIFICATIONS
| Reference | INCC |
| Weight (g) | 15 |
| Energy | Manual |
 The INCC ratchet wrench is for use with Anthogyr instruments only.
The INCC ratchet wrench is for use with Anthogyr instruments only.
USING THE MD
The MD is supplied non-sterile. Before first using it, this MD must be decontaminated and sterilized (see § « Hygiene and maintenance”).
Before use, check that the MD is not damaged or that no part is missing.
5.1. Connecting / Disconnecting the instrument:![]() It is preferable to wear protective gloves for all tool handling. Check the condition of the rotary instruments used and handle them cautiously and carefully.
It is preferable to wear protective gloves for all tool handling. Check the condition of the rotary instruments used and handle them cautiously and carefully.
5.2. Inserting and locking the rotary instrument:
- Insert the rotary instrument and turn it slightly until hearing a clicking sound and the ratchet takes place in the groove.

- Check that the rotary instrument is correctly held with a slight axial movement each time the tool is changed.
5.3. Removing the rotary instrument:
Remove the instrument by pulling it out.
MD lifecycle: If used in a proper manner, all MD parts have a two-year lifecycle (corresponding to 250 sterilization cycles). However, these indications are not a warranty because wear may appear prematurely, depending on how the MD is maintained (cleaning and sterilization).
HYGIENE AND MAINTENANCE
 Sterilization of medical devices must be done by properly trained and protected staff, in compliance with current regulations. The sterilization protocol must be appropriate to the infectious risk. Wear appropriate protective clothing: risk of infection and injury. Only use products for the maintenance of medico-surgical equipment compatible with stainless steel (no chlorinated content). Forbid antiseptics that are intended for use on skin and mucous. Forbid products containing aldehyde, alcohol or other products are likely to bind proteins.
Sterilization of medical devices must be done by properly trained and protected staff, in compliance with current regulations. The sterilization protocol must be appropriate to the infectious risk. Wear appropriate protective clothing: risk of infection and injury. Only use products for the maintenance of medico-surgical equipment compatible with stainless steel (no chlorinated content). Forbid antiseptics that are intended for use on skin and mucous. Forbid products containing aldehyde, alcohol or other products are likely to bind proteins. For each product used: refer to the manufacturer’s instructions. Comply with the concentrations, exposure durations, and life span of products. Do not mix the products and follow the instructions for their disposal.
For each product used: refer to the manufacturer’s instructions. Comply with the concentrations, exposure durations, and life span of products. Do not mix the products and follow the instructions for their disposal.
After patient care, the MD should be processed within the next two hours.
6.1. Preparation of MD in the place of use
To be done immediately after surgery: Remove the largest organic contaminants with disinfectant wipes.
6.2 Reprocessing preparation
Disassembling the MD:
Disassemble the MD as described below: Unscrew the head (1) and remove it from the main body (2).
Unscrew the head (1) and remove it from the main body (2). Remove the set «ratchet (3) + rod (4)» from the body, by pushing slightly against each other the back wheel of the rod (4) and the ratchet (3) while simultaneously rotating the ratchet (3) ¼ turn anticlockwise in order to unlock the bayonet.
Remove the set «ratchet (3) + rod (4)» from the body, by pushing slightly against each other the back wheel of the rod (4) and the ratchet (3) while simultaneously rotating the ratchet (3) ¼ turn anticlockwise in order to unlock the bayonet.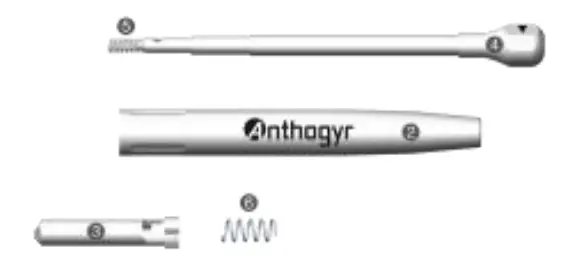 6.2.1 Manual reprocessing preparation
6.2.1 Manual reprocessing preparation
- Brush thoroughly with a soft brush (such as nylon) under running tap water, at room temperature for 3 minutes. Use a smaller brush for hard-to reach areas.
- Immerse it completely in 8mL/L Cidezyme solution, incubated at 38-40°C for 3 minutes.
- Rinse with purified water for at least one minute.
- Rinse again with purified water while brushing with a clean soft brush (such as nylon) for 1 minute.
- Dry immediately and carefully, with a soft lint-free cloth.
6.2.2 Automatic reprocessing preparation![]() The INCC ratchet wrench is designed to withstand thermal disinfection cleaning.
The INCC ratchet wrench is designed to withstand thermal disinfection cleaning.
The thermal disinfection cycle
| Detergent and | ||||
| Pre-wash | Temperature Coldwater | the concentration used Duration Cleaning agent by Anthogyr to approve protocol | ||
| 4 min. | Water | Not applicable | ||
| Washing | 55°C | 5 min. | Enzymatic detergent | Neodisher Mediclean Dental (2mL/L) |
| Neutralization | 55°C | 3 min. | Neutralizer | Neodisher Z Dental (1mUld |
| Rinsing | 40°C | 2 min. | Water | Not applicable |
| Thermal disinfection | 95°C | 10 min. | Not applicable | Not applicable |
| Final rinsing | 40°C | 2 min | Rinsing liquid | Neodisher Mielclear (0.2mL/L) |
| Drying | 140°C | 10 min. | Not applicable | Not applicable |
![]() Check there is no residue and that all the parts of the MD are completely dry at the end of the cycle.
Check there is no residue and that all the parts of the MD are completely dry at the end of the cycle.
Re-assembling the MD:
Repeat the disassembling operation above in reverse order. Insert the set «rod (4) + spring (5)» through the back part of the body (2). Fit the loose spring (6) around the rod (4) through the front part of the body (2).
Assemble the ratchet (3) by pushing it onto the rod (4) and rotating it ¼ turn clockwise to lock the bayonet. Screw the head (1) onto the body (2).
6.3 Sterilisation:
- The instruments are to be sterilized before first use and after each use.
- Only sterilize cleaned, lubricated, and tested instruments.
- Anthogyr recommends class B autoclave sterilisation according to stand- guard EN 13060 for all devices bearing the logo

- Independently put each MD in a sterilization sachet that complies with current standards and the size of the MD.
- Comply with the space between the sachets and do not overload the autoclave.
- Run a sterilisation cycle according to the following parameters:
| Country | Sterilisation parameters | Drying time |
| European Union | 135°C (-1°C / +2°C) for 3 minutes | 10 minutes |
| France | 135°C (-1°C / +2°C) for 18 minutes | 10 minutes |
| United States | 132°C (0°C / +3°C) (270°F) for 3 minutes | 16 minutes |
- Allow cooling to room temperature for around 10 minutes.
- Indicate the sterilization date and the expiration date on the package in accordance with the data from the package manufacturer (maximum 1 month).
![]() Check that the device is completely dry at the end of the cycle.
Check that the device is completely dry at the end of the cycle. Other products and detergents may be used according to local availability. Approval of these products is the responsibility of the user.
Other products and detergents may be used according to local availability. Approval of these products is the responsibility of the user.
REPAIRS
In the case of device failure, please contact your authorized distributor, or contact our after-sales service department directly.
Repairs may only be carried out by an authorized repair service or by the Anthogyr after-sales service department, and only using original Anthogyr replacement parts.
For any servicing or repair work, the prosthetic wrench must be returned complete and sterile with proof of sterility. It must be accompanied by a document that describes the problem in question and also includes the complete contact details of the practitioner who used the device.
Exchange of replacement parts is possible for 7 years after-sales discontinuation.
CONTACT DETAILS: AFTER-SALES SERVICE
AFTER-SALES SERVICE DEPARTMENT
Author
2237 Avenue André Lasquin – 74700 Sallanches – FRANCE
Direct line: +33 (0)4 50 58 50 53
Mail: [email protected]
WARRANTY
This MD is guaranteed parts and labor against all manufacturing defects for 12 months from the date of invoice. This guarantee does not apply to wear and tear parts. All changes or additions to the product without the express agreement of Anthogyr render this guarantee null and void. The guarantee becomes null and void if the technical instructions are not followed.
Anthogyr cannot be held responsible for damage resulting from or which could result from normal wear, use, cleaning or incorrect maintenance, the non-observance of instructions for use or connection, scaling or corrosion, impurities in the water supply system, or unusual chemical or electrical influences or nonobservance of the instructions, maintenance instructions, and assembly of Anthogyr and other manufacturer’s instructions.
Delivery charges incurred when sending an instrument back to Anthogyr for repair will be paid by the client, even if the repair itself is covered by the guarantee.
Postage and packing fees when returning the instrument to the client are covered by the guarantee.
So that guarantee requests are taken into consideration, please attach a copy of the invoice or a copy of the delivery slip to the MD.
ELIMINATION DISPOSAL
As far as is currently known, the product does not contain any substances which are harmful to the environment. The product must be sterilized before being disposed of. Observe national rules with regard to disposal.
![]() Anthogyr
Anthogyr
2237 avenue André Lasquin
74700 SALLANCHES – FRANCE
Tél. +33 (0) 4 50 58 02 37
Fax +33 (0)4 50 93 78 60
N°SAV / Repairs : +33 (0) 4 50 58 50 53
E-mail : [email protected]
www.anthogyr.com REF 063INCC_NOT-2020-07
REF 063INCC_NOT-2020-07
Made in France