FISHER PAYKEL 900MR860 Temperature Probe

USER INSTRUCTIONS
Reprocessing Diagram
E.g.
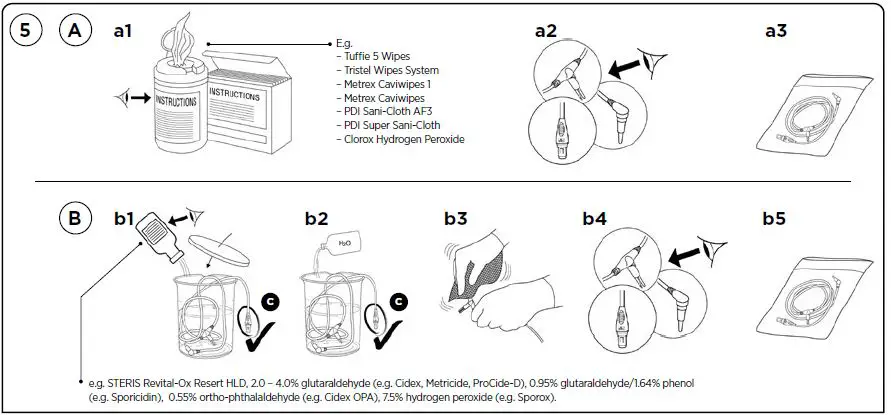
- Tuffie 5 Wipes
- Tristel Wipes System
- Metrex Caviwipes 1
- Metrex Caviwipes
- PDI Sani-Cloth AF3
- PDI Super Sani-Cloth
- Clorox Hydrogen Peroxide
e.g. STERIS Revital-Ox Resert HLD, 2.0 – 4.0% glutaraldehyde (e.g. Cidex, Metricide, ProCide-D), 0.95% glutaraldehyde/1.64% phenol
(e.g. Sporicidin), 0.55% ortho‑phthalaldehyde (e.g. Cidex OPA), 7.5% hydrogen peroxide (e.g. Sporox).
| Equipment | Setting | Reprocessing Cycles |
| STERIS V-PRO 1 | Standard Cycle |
78 |
| STERIS V-PRO 1 Plus | Non-Lumen Cycle | |
| STERIS V-PRO 60 | ||
| STERIS V-PRO maX | ||
| Sterrad 100NX | Standard Cycle | 25 |
| VP4 STERIZONE | Cycle 1 | 78 |

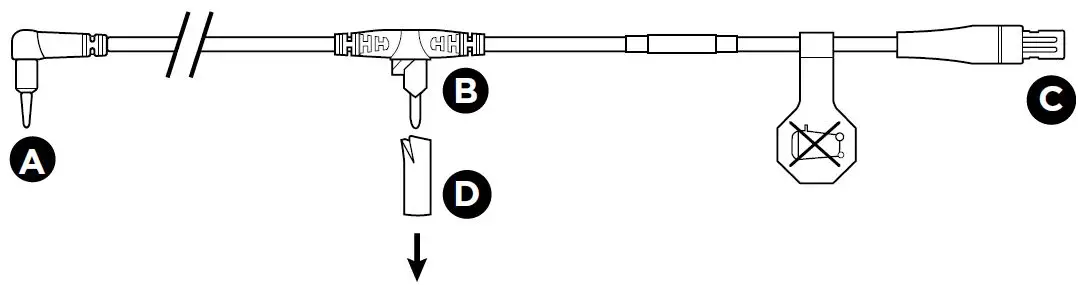
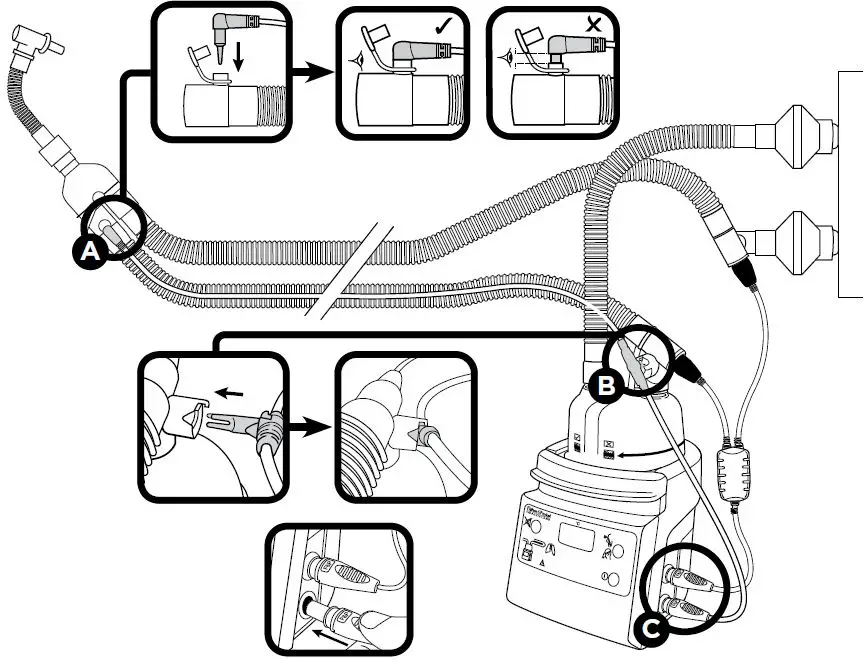
Setup Diagram
Temperature Probe User Instructions
Intended Use:
The Temperature Probe is designed for measuring gas temperature and flow with Fisher & Paykel Healthcare MR850 and HC550 Humidifiers and Fisher & Paykel Healthcare Breathing Circuits.
Specification:
Expected Service Life:
3 years, if used in accordance with this User Instruction. Note: Service life may vary depending on usage pattern.
Setup:
Refer to the setup diagram.
- Before use remove and discard Protective Sleeve D, if present, from Chamber Probe B .
- Visually inspect entire product for damage before use. Discard if damaged.
- Push the Airway Probe A and Chamber Probe B into the Breathing Circuit making sure they are correctly located and pushed into place.
- The probe lead can be restrained using
Breathing Circuit Clips.- If patient is in an incubator, refer to Breathing Circuit User Instructions for probe positioning information.
- Insert the Temperature Probe Connector C into the blue socket on the Humidifier.
- Perform ventilator leak test on the Breathing Circuit before use.
Maintenance:
Probe maintenance and accuracy checks should be routinely performed. For further information, refer to the MR850 or HC550 Technical Manual (available from your local Fisher & Paykel Healthcare representative).
Reprocessing:
The following parts are in contact with the gas path and can become contaminated with body fluids or expired gases:
Airway Probe A, Chamber Probe B.
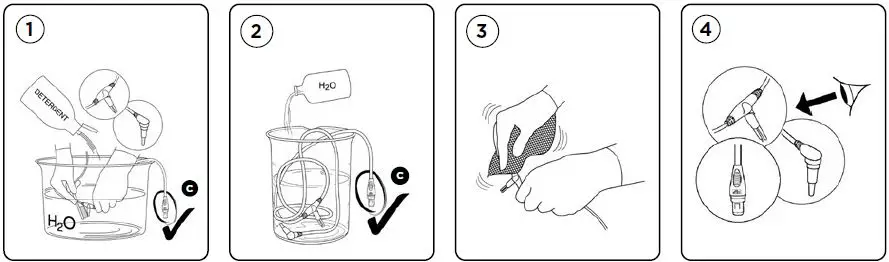
Recommended cleaning procedures between uses (refer to reprocessing diagram):
- Remove any visible soil using a soft bristle brush, water and a mild non-abrasive detergent.
Do not immerse the Temperature Probe Connector C in any liquid. - Rinse in water after cleaning.
- Dry with a non-shedding cloth.
- Check for visible signs of damage. Discard if damaged. Repeat the cleaning steps if parts are not visually clean.
- Proceed to either step A, B, C or D depending on hospital guidelines.
- A. MANUAL DISINFECTION – WIPES
- Follow the wiping instructions supplied by the manufacturer.
- Check for visible signs of damage.
Discard if damaged. - If applicable, product can be packaged for reuse.
- B. MANUAL DISINFECTION – CHEMICAL
- Follow the soaking instructions supplied by the manufacturer of the disinfection solution.
Do not immerse the Temperature
Probe Connector C in any liquid.
Cover container while soaking. - Rinse in water after chemical soaking.
- Dry with a non-shedding cloth.
- Check for visible signs of damage.
Discard if damaged. - If applicable, product can be packaged for reuse.
- Follow the soaking instructions supplied by the manufacturer of the disinfection solution.
- C. VAPORISED HYDROGEN PEROXIDE (VHP) STERILIZATION
- Follow hospital guidelines for using the equipment (Equipment) and settings (Setting).
- D. ETHYLENE OXIDE (ETO) STERILIZATION
- Follow hospital guidelines for process.
- Allow at least 12 hours aeration.
- A. MANUAL DISINFECTION – WIPES
- Store as per hospital guidelines.
Notes:
- Fisher & Paykel Healthcare do not recommend exceeding the number of reprocessing cycles stated (Reprocessing Cycles).
- The product is not designed for autoclave sterilization processes.
- Follow these recommended cleaning procedures and the user instructions of the cleaning agents or disinfection/sterilization equipment. It is the user’s responsibility to qualify any deviations from these procedures, both for disinfecting efficacy and physical effect.
Warnings:
- The use of breathing circuits, chambers, other accessories or parts which are not approved by Fisher & Paykel Healthcare may impair performance or compromise safety.
- Failure to perform routine probe accuracy and visible damage checks may impair performance or compromise safety.
- Ensure that both Temperature Probe sensors are correctly and securely fitted. Failure to do so may result in gas temperatures in excess of 41 °C being delivered to the patient.
- Do not touch the glass tip of the Chamber
Probe B during use; it may cause a skin burn. - Gas mixes, such as helium-oxygen mixtures, that have different physical or thermal properties than air or air-oxygen mixture, may impair system performance or compromise safety.
- No automated method for cleaning and disinfection is recommended.
- California residents please be advised of the following, pursuant to Proposition 65:
This product contains chemicals known to the State of California to cause cancer,birth defects and other reproductive harm.
For more information, please visit: www.fphcare.com/prop65.
Disposal:
Dispose according to standard hospital guidelines for electrical and electronic equipment.
Symbol Definition:
| Operating Instructions | |
| Date of manufacture | |
| Manufacturer | |
| Dispose of product in correct manner | |
| Transportation and storage humidity limitation | |
| Transportation and storage temperature limitation | |
| Lot number | |
| Reference number | |
| European union authorised representative | |
| Prescription only | |
| CE Marking 93/42/EEC | |
| MD symbol |
Manufacturer Fisher & Paykel Healthcare Ltd, 15 Maurice Paykel Place, East Tamaki, Auckland 2013, PO Box 14 348 Panmure, Auckland 1741, New Zealand Tel: +64 9 574 0100
Fax: +64 9 574 0158
Email: [email protected]
Web: www.fphcare.com Importer/ Distributor
REF 617135 REV E 2022-02 © 2022 Fisher & Paykel Healthcare Limited