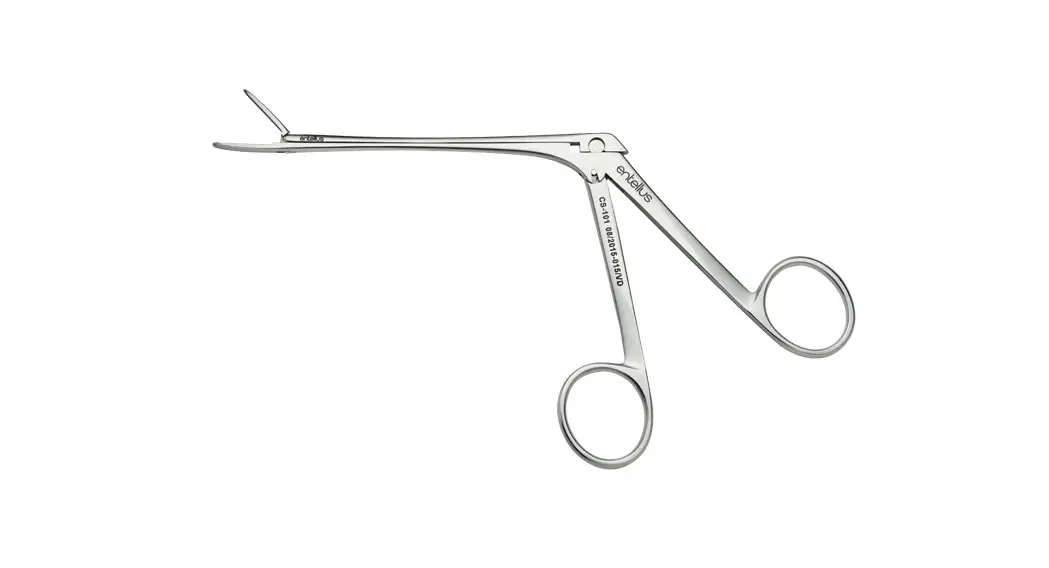
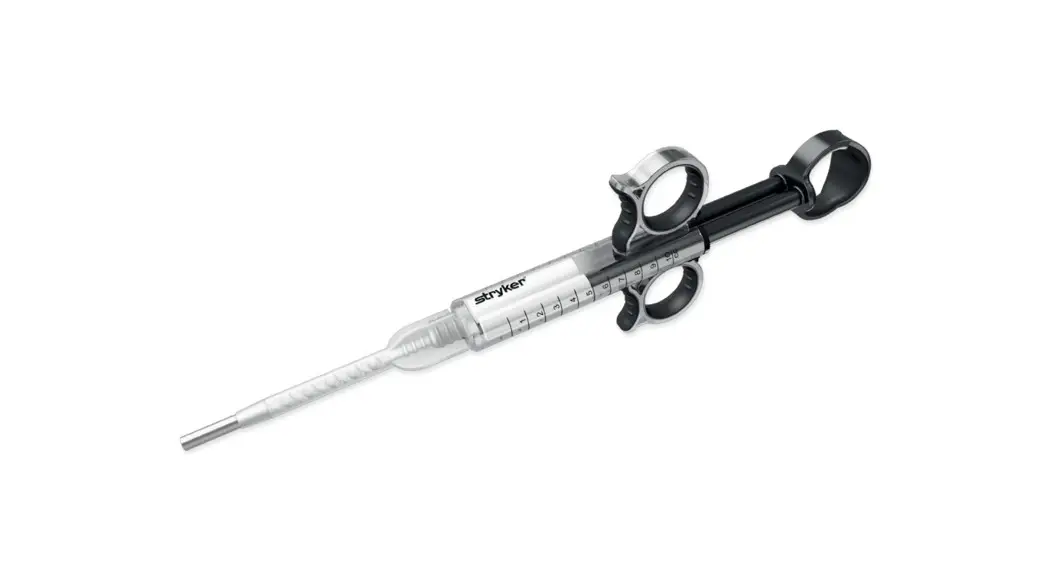
Stryker 0233032105 Endoscope and Camera Sterilization Tray

Introduction
This processing guide provides instructions for the proper cleaning and sterilization of the following sterilization tray:
0233032105 Endoscope and Camera Sterilization Tray.
This sterilization tray is intended for use only with the devices listed below. For tray/device configuration, see the “Sterilization Tray Setup” section of this guide:
| For steam sterilization, a combination of | |
| Any three | Stryker autoclavable arthroscopes or sinuscopes |
| Any one | Stryker autoclavable camera head |
| Any one | Stryker autoclavable coupler |
| Any one | Stryker light cable |
| Any three | Stryker arthroscope or sinuscope adapters |
| Any two | Stryker light cable adapters |
| Any three | Stryker Sinuscope Sheaths |
| For Sterrad and V-Pro sterilization, a combination of | |
| Any three | Stryker arthroscopes or sinuscopes |
| Any one | Stryker camera head |
| Any one | Stryker coupler |
| Any one | Stryker light cable |
| Any three | Stryker arthroscope or sinuscope adapters |
| Any two | Stryker light cable adapters |
| Any three | Stryker Sinuscope Sheaths |
Intended Use of Sterilization Trays
Sterilization trays are plastic and/or metal containers used to hold and protect surgical devices during the sterilization process. They consist of an interlocking tray and lid, which are both perforated to allow the passage of sterilizing agent from outside the tray to the devices placed inside.
Sterilization trays typically feature a silicon finger mat or group of device holders that secure devices during the sterilization process. Some models feature stacking internal trays to allow the segregation of devices.
Warnings
- These instructions are validated only for sterilization of the tray and device(s) identified herein. Using combinations or parameters not described in this manual may result in incomplete sterilization.
- These instructions do not replace the cleaning instructions provided with individual devices. Prior to sterilization, clean all devices as specified in their respective user manuals.
- Wear appropriate protective equipment (gloves, eye protection, etc.) when processing any medical device.
- Both the device and the tray must be cleaned prior to sterilization, or incomplete sterilization will result.
- The sterilization tray, its lid, and any internal components have been designed and validated for use as a single system. Do not separate components from the system, for use individually or in combination, or incomplete sterilization may result.
Cautions
- Before lifting the tray assembly, verify that the latches connecting the lid to the tray are secure.
- The tray is not designed for use as a shipping container. To avoid damage, remove all devices and pack them separately.
Materials And Equipment
The end user shall supply all materials and equipment required to process the device(s) unless otherwise noted.
| Item | Description or Purpose |
| All Phases | |
| Gloves, eye protection, etc | Wear personal protective equipment (PPE) as required by the medical facility and procedure |
| Cleaning/Thermal Disinfection | |
| Water basin or sink | Large enough to accommodate device(s) without causing excessive bending or other physical stress that could damage the device(s) |
| Detergent1 | To remove surgical debris during automated and manual cleaning |
| Utility Water (Water as it comes from the tap) | To rinse during pre-treatment and to prepare cleaning solutions |
| Soft-bristle brush2 | To clean exterior of device and hard-to-reach areas of the device |
| Critical Water3 (Water that has been extensively treated such as RO/DI or distilled) | For final rinsing following soaking and brushing |
| Clean lint-free cloth or filtered pressurized air (≤40 psi) | For drying |
| Automated Washer | For executing automated cleaning/disinfection procedure |
| Sterilization | |
| Sterilization System |
|
| Sterilization Wrap4,5 | To maintain a sterile barrier |
1The following detergents were validated for cleaning efficacy according to the detergent manufacturer’s instructions. Choose one (1) of the detergents listed below or a substantially equivalent detergent. Do not exceed the concentration and temperature recommended by the detergent manufacturer.
| Detergent | Type | Minimum Concentration | Minimum Soak Time | pH |
| Prolystica™ HP Enzymatic (1C22/1C24) | Enzymatic | 1/4 oz/gallon (2 ml/L) | 1 minute | 8.0 |
| Prolystica HP Neutral (1C21/1C27) | Neutral | 1/4 oz/gallon (2 ml/L) | 1 minute | 8.0 |
| Prolystica HP Alkaline (1C20/1C26) | Alkaline | 1/4 oz/gallon (2 ml/L) | 1 minute | 11.0 |
Cleaning was validated with an M16 soft-bristle brush.
Reverse Osmosis/Deionized (RO/DI) water at ≤30 °C was validated for cleaning efficacy.
Sterilization was validated using 2-ply polypropylene sterilization wrap.
For United States users: When sterilizing the cycle, use only sterilization wraps and sterilization trays that have been cleared by the FDA for use with selected sterilization cycle.
| Limitations On Processing |
|
| Processing Instructions apply to the sterilization tray only. For instructions on how to process the devices, consult their respective instructions for use. | |
| Point of Use |
|
| Containment and Transportation |
|
| Preparation for Cleaning | Disassemble the tray into its individual components: base, lid, and (if applicable) silicon mat. |
| Pre-Treatment | For necessary materials and equipment, see Materials and Equipment table.
|
| Caution |
|
| Manual Cleaning | Soak
|
Brush
| |
Rinse
|
| Automated Cleaning And Optional Thermal Disinfection |
The following minimum and maximum washer parameters have been validated for cleaning efficacy and device functionality: | |||||||
| Recirculation Time | Temperature | Detergent type | ||||||
| Pre-Wash | 1 – 2 minutes | Cold water | N/A | |||||
| Enzyme Wash (Optional) | 1 minute | 43 – 50°C (110 – 122 °F) | Enzymatic | |||||
| Main Wash | 2 – 4 minutes | 43 – 66°C (110 – 151°F) | Neutral or Alkaline | |||||
| Rinse 1 | 1 – 2 minutes | 43 – 66°C (110 – 151°F) | N/A | |||||
| Thermal Rinse (optional) | 1 – 5 minutes | 90 – 93°C (194 – 199°F) | N/A | |||||
| Dry Phase | 3 – 15 minutes | High (≤115°C or 239°F) | N/A | |||||
| Drain excess water from the device and dry it using a clean lint-free cloth or pressurized air. Visually inspect each device for cleanliness, paying close attention to hard-to-reach areas. If visible soil remains, repeat steps 1 – 4.Note: Thermal disinfection value A0 ≥3000 may be utilized for compatible devices. Do not exceed the time and temperature specified in the table above. | ||||||||
| Maintenance, Inspection And Testing |
| |||||||
| Preparation For Sterilization |
| |||||||
| Sterilization | Warnings:
V-PRO in a non-lumen cycle may result in incomplete sterilization. | |||||||
| Sterrad | ||||||||
| Sterilization method | Cycle | |||||||
| Sterrad 100S | Standard | |||||||
| Sterrad NX | Standard | |||||||
| Sterrad NX ALLClear | Standard | |||||||
| Sterrad 100NX | Standard | |||||||
| Sterrad 100NX ALLClear | Standard | |||||||
| Steris V-PRO If the tray contains at least one Sinuscope Sheath, the tray should only be processed with the lumen cycle. | ||||||||
| Sterilization method | Cycle | |||||||
| With Sheaths | Without Sheaths | |||||||
| V-PRO 1 Sterilizer | Lumen | Standard | ||||||
| V-PRO 1 Plus Sterilizer | Lumen | Non-Lumen or Lumen | ||||||
| V-PRO maX Sterilizer | Lumen | Non-Lumen or Lumen | ||||||
| V-PRO 60 Sterilizer | Lumen | Non-Lumen or Lumen | ||||||
| V-PRO s2 | Lumen | Non-Lumen or Lumen | ||||||
| V-PRO maX 2 Sterilizer | Lumen | Non-Lumen, Lumen or Fast Non- Lumen1 | ||||||
When using the Fast Non-Lumen cycle, follow the instructions provided with the V-PRO maX 2 system regarding use of rigid containers or sterilization wrap. The cycle is intended to be used with sterilization pouches. Also observe the Fast Non-Lumen cycle weight limit of up to 11 pounds (4.99 kg) of instruments.
| Steam (Autoclave) | Warning: Only devices marked AUTOCLAVE are compatible with steam sterilization methods. Using steam sterilization on devices that do not bear this marking can cause permanent device damage. Note: The water used in the autoclave process must meet standards for clean steam per AAMI ST79 Annex-Steam Quality. | |||
| Pre-vacuum (dynamic air removal) | ||||
| U.S. | Outside of U.S. | Prion Cycle1 | ||
| Wrapping | Double | |||
| Temperature | 132°C (270°F) | 134 – 137°C (273 – 279°F) | 134°C (273°F) | |
| Exposure Time | 4 minutes | 3 – 5 minutes | 18 minutes | |
| Dry Time (minimum in chamber) | 60 minutes | 50 minutes | 50 minutes | |
| 1Refer to Video Devices Processing Guide P46385 to confirm device compatibility with the Prion cycle. Caution: Longer cycles, such as those recommended for control or elimination of Transmissible Spongiform Encephalopathies, may be utilized. However, a device exposed to longer cycles should be expected to have reduced functional life. Warning: Drying time depends on several variables including altitude, humidity, type of wrap, preconditioning, size of chamber, mass of load, and placement in the chamber. Users must verify that drying time set in their autoclave yields dry surgical equipment. | ||||
| Storage | Store the device in a dry, clean, and dust-free environment at room temperature. | |||
| Expected Service Life | The service life of the tray is largely determined by wear, processing methods, and any damage resulting from use. To extend the time between tray servicing, always follow the care and handling instructions in this user manual. Before each use, test the functionality of the tray and inspect it for any sign of damage per the Inspection section. | |||
| Adverse Event Reporting | Any serious incident that has occurred in relation to this device should be reported to Stryker and, in the European Union, to the competent authority of the Member State in which the affected person resides. | |||
| Safe Disposal | Warning: Device(s) may present a biohazard and should be handled in accordance with accepted medical practice and local and national requirements. It is recommended that devices are decontaminated as per the above Processing section in this Guide. | |||
Immediate-Use Steam Sterilization
Immediate-Use Steam Sterilization (IUSS or “Flash”) is intended only for emergency situations. In the event that immediate-use steam sterilization is required, the following instructions should be used.
Warning: Immediate-use steam sterilization should only be used in carefully selected clinical situations (e.g., an instrument needed for a case falls on the floor and no replacement instrument is available). IUSS should not be used due to inadequate inventory.
| Pre-vacuum (dynamic air removal) | ||
| Region | U.S. | Outside of U.S. |
| Wrapping | None | |
| Temperature | 132°C (270°F) | 134 – 137°C (273 – 279°F) |
| Exposure Time | 4 minutes | 3 minutes |
| Dry Time | None | |
Note: IUSS may be used only if allowed by local regulation.
| Sterilization Tray Setup | |
| Maximum Weight Load (devices and tray combined) | 2.59 kg (5.71 lbs) |
| Internal Stacking | No internal stacking is permitted with this tray. |
| External Stacking | Do not stack other trays or devices on or below this tray. |
| Accessories | There are no accessories available for use with this tray. |
| Device Distribution | Devices should be placed in the tray as illustrated below. |
| Chemical Indicator* | Place the chemical indicator ( |
| Biological Indicator* | Place the biological indicator |
| *Note: Placement of the biological or chemical indicator is not required during sterilization of this tray. If, per hospital procedure, placement of an indicator is desired, the recommended placement sites are as follows. | |

References
These processing instructions are provided in accordance with the below referenced standards. While they have been validated by Stryker as being capable of preparing the device for re-use, the end user is responsible for ensuring that the processing, as actually performed (using equipment, materials, and personnel in the processing facility), achieves the desired result. This normally requires routine monitoring and validation of the facility’s processing procedures. Stryker recommends users observe these standards when processing medical devices.
- AAMI TIR12: Design, testing and labeling reusable medical devices for processing in healthcare facilities: a guide for medical device manufacturers
- AAMI TIR30: A compendium of processes, materials, test methods, and acceptance criteria for cleaning reusable medical devices
- AAMI TIR34: Water for processing of medical devices
- ANSI/AAMI ST58: Chemical sterilization and high-level disinfection in healthcare facilities
- ANSI/AAMI ST77: Containment devices for reusable medical device sterilization
- ANSI/AAMI ST79: Comprehensive guide to steam sterilization and sterility assurance in healthcare facilities
- ISO 15883-1: Washer-disinfectors — Part 1: General requirements, terms and definitions and tests
- ISO 17664-1: Processing of health care products – Information to be provided by the medical device manufacturer for the processing of medical devices – Part 1: Critical and semi-critical medical devices
Symbol Definitions
| Symbol Definitions | |
| The device meets European Union medical device requirements | |
 | Stryker European representative |
| Product catalog number | |
| Batch code | |
 | Consult instructions for use |
 | Device is shipped non-sterile and must be sterilized before use |
| Made in USA | |
| Quantity | |
 | Legal manufacturer |
 | Date of manufacture |
| Federal law (USA) restricts this device to use by, or on order of, a physician | |
 | Medical device in the European Union |
Produced for
Stryker Endoscopy
5900 Optical Court
San Jose, CA 95138 USA
1-800-624-4422
U.S. Patents: www.stryker.com/patents
Stryker or its divisions or other corporate affiliated entities own, use or have applied for the following trademarks or service marks: the Stryker logo. All other trademarks are trademarks of their respective owners or holders.
1000400907M
2022/03