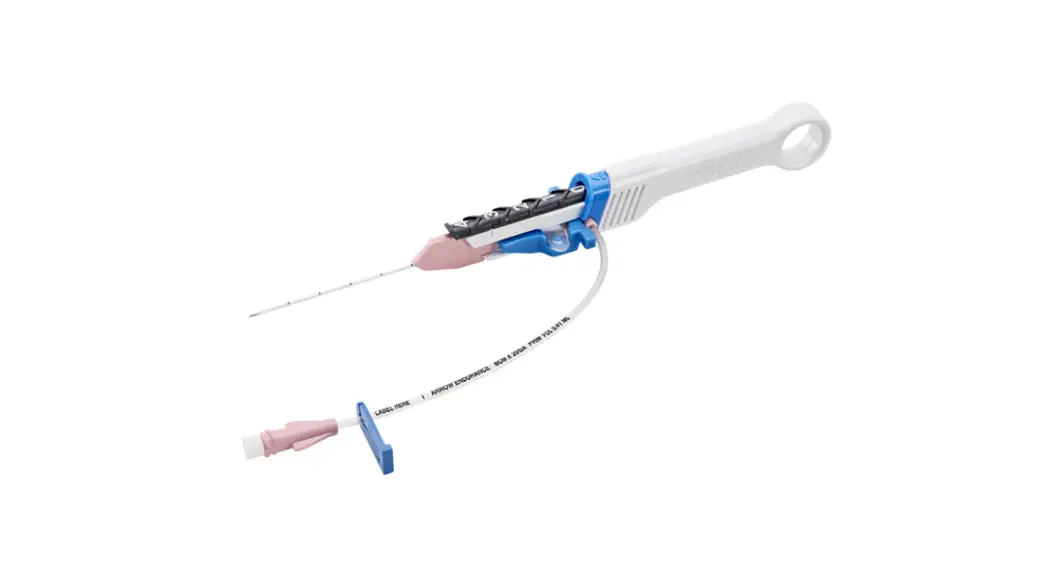
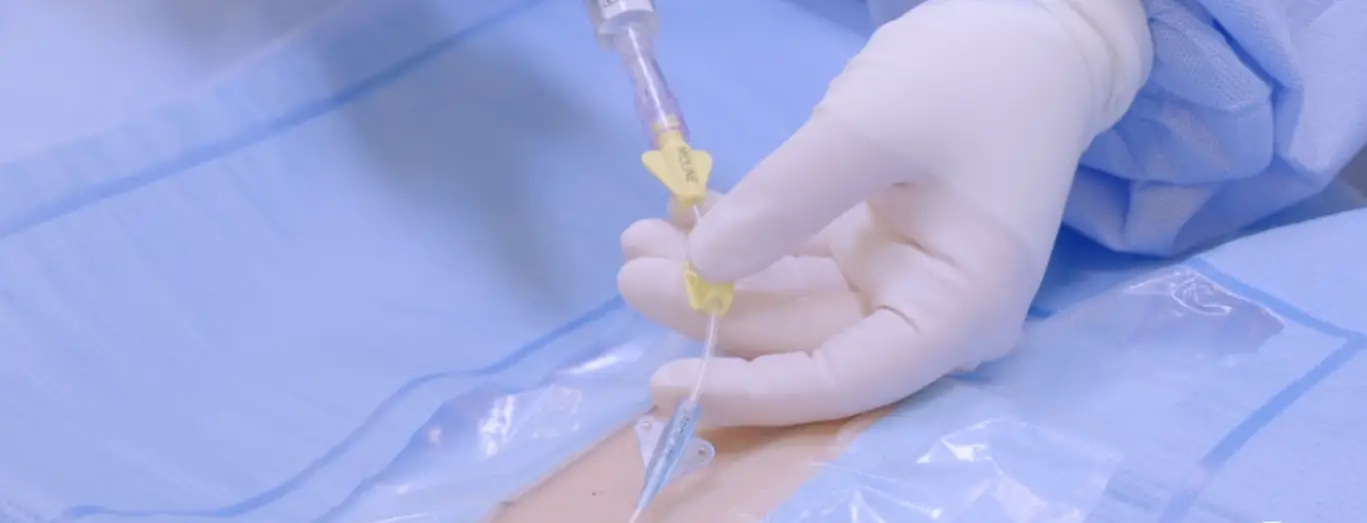
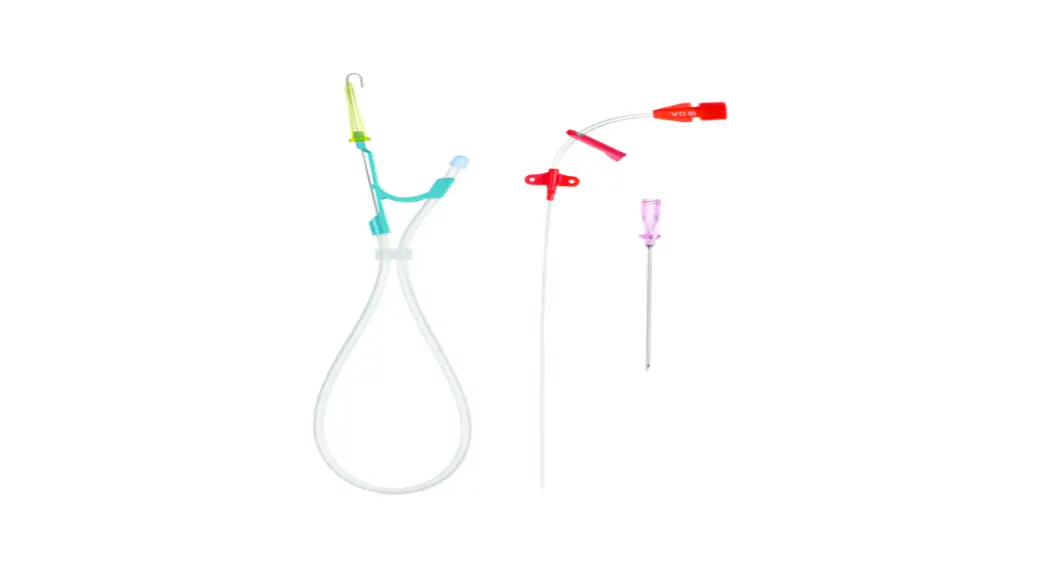
AXS Vecta 46 Intermediate Catheter
Product Information:
The AXS Vecta 46 Intermediate Catheter is a medical device that
is used to facilitate the insertion and guidance of appropriately
sized interventional devices into a selected blood vessel in the
neurovascular system. It is also used as a conduit for retrieval
devices. The catheter is intended for use in patients who are
ineligible for intravenous tissue plasminogen activator (IV t-PA)
or who have failed IV t-PA therapy. The product is for single-use
only and is available for use by healthcare professionals only.
The product is not indicated for use in the coronary or
peripheral vasculature. Automated high-pressure contrast injection
equipment should not be used with the AXS Vecta 46 Intermediate
Catheter as it may damage the device. Potential adverse events
associated with the use of catheters or with the endovascular
procedures include, but are not limited to:
Product Usage Instructions:
The AXS Vecta 46 Intermediate Catheter should only be used by
trained healthcare professionals who are familiar with the
product’s indications, contraindications, warnings, and
instructions for use. Prior to use, carefully review the electronic
instructions for use (eIFU) provided with the product.
The catheter should be inserted through an appropriate access
site and advanced under fluoroscopic guidance to the target
location. The appropriately sized interventional device can then be
inserted through the catheter for further treatment. The catheter
can also be used as a conduit for retrieval devices.
It is important to follow all contraindications and warnings
provided with the product to avoid potential adverse events. The
product is for single-use only and should be disposed of properly
after use.
AXS Vecta 46 Intermediate Catheter
RX ONLY
See eIFU for complete indications, contraindications, warnings and instructions for use.
Indications for use as a conduit The AXS Vecta 46 Intermediate Catheter is indicated for use in facilitating the insertion and guidance of appropriately sized interventional devices into a selected blood vessel in the neurovascular system. The AXS Vecta 46 Intermediate Catheter is also indicated for use as a conduit for retrieval devices.
Indications for use as a revascularization device The AXS Vecta 46 Intermediate Catheter, as part of the AXS Vecta Aspiration System, is indicated in the revascularization of patients with acute ischemic stroke secondary to intracranial large vessel occlusive disease (within the internal carotid, middle cerebral M1 and M2 segments, basilar, and vertebral arteries) within 8 hours of symptom onset.
Patients who are ineligible for intravenous tissue plasminogen activator (IV t-PA) or who failed IV t-PA therapy are candidates for treatment.
Contraindications Do not use the AXS Vecta 46 Intermediate Catheter in the coronary or peripheral vasculature.
Do not use automated high-pressure contrast injection equipment with the AXS Vecta 46 Intermediate Catheter because it may damage the device.
Adverse events Potential adverse events associated with the use of catheters or with the endovascular procedures include, but are not limited to:
· Acute vessel occlusion
· Air embolism
· Allergic reaction and anaphylaxis from contrast media · Arteriovenous fistula
· Death
· Device malfunction
· Distal embolization
· Emboli
· False aneurysm formation
· Hematoma or hemorrhage at the puncture site
· Inability to completely remove thrombus
· Infection
· Intracranial hemorrhage
· Ischemia
· Kidney damage from contrast media · Neurological deficit including stroke · Risks associated with angiographic and fluoroscopic
radiation including but not limited to: alopecia, burns ranging in severity from skin reddening to ulcers, cataracts, and delayed neoplasia · Sterile inflammation or granulomas at the access site
· Tissue necrosis
· Vessel spasm, thrombosis, dissection or perforation
Warnings
· Do not use kinked, damaged, or opened devices.
· Exposure to temperatures above 54°C (130°F) may damage device. Do not autoclave.
· Torqueing or moving the device against resistance may result in damage to the vessel or device.
· The AXS Vecta 46 Intermediate Catheter has not been evaluated for more than one (1) clot retrieval attempt.
· The AXS Vecta 46 Intermediate Catheter was evaluated for an average duration of direct aspiration of 4 minutes.
· This product is intended for single use only, do not re-sterilize or reuse. Re-sterilization and/or reuse may result in cross contamination and/or reduced performance.
· When the catheter is exposed to the vascular system, it should be manipulated while under high-quality fluoroscopic observation. Do not advance or retract the catheter if resistance is met during manipulation; determine the cause of the resistance before proceeding.
· If flow through the device becomes restricted, do not attempt to clear the lumen by infusion. Remove and replace the device.
· This device is coated with a hydrophilic coating at the distal end of the device for a length of 25 cm. Please refer to the Device Preparation Section for further information on how to prepare and use this device to ensure it performs as intended. Failure to abide by the warnings in this labeling might result in damage to the device coating, which may necessitate intervention or result in serious adverse events.
· Verify aspiration pump is appropriate before use if using a vacuum pump other than the Medela Dominant Flex Pump. The AXS Vecta 46 Intermediate Catheters have been verified for use with the Medela Dominant Flex Pump and AXS Universal Aspiration Tubing. The Medela Dominant Flex Pump is capable of delivering vacuum pressures between -20.08 inHg and -28 inHg [[-68 kPa to -95 kPa] [-510 mmHg to -713 mmHg]] during use and is characterized by the pressure-flow performance curve below. If using another vacuum pump other than the Medela Dominant Flex Pump, carefully review the vacuum pump performance parameters to ensure it is equivalent and can achieve the same operating vacuum pressures between -20.08 inHg and -28 inHg [[-68 kPa to -95 kPa] [-510 mmHg to -713 mmHg]] and corresponds to the same flow rate ranges (see pump pressure-flow performance graph). The vacuum pump should also be verified to be compatible with the AXS Universal Aspiration Tubing.
· Limit the usage of the AXS Vecta 46 Intermediate Catheter to arteries greater than the catheter’s outer diameter.
· Do not exceed the maximum permissible pressure of 70 psi.
Precautions · Use the device prior to the “Use By” date specified on
the package. · Maintain a constant infusion of appropriate flush
solution.
· Examine the device to verify functionality and to ensure that its size and shape are suitable for the specific procedure for which it is to be used.
· The AXS Vecta 46 Intermediate Catheter should be used only by physicians trained in percutaneous procedures and/or interventional techniques.
· The AXS Vecta Aspiration System should be used only by physicians trained in percutaneous procedures and/or interventional techniques.
· If using the AXS Vecta Aspiration System for Thrombectomy, monitor the canister fluid level and replace the canister if the fill level reaches 75% of the canister volume.
· Administration of anticoagulants and antiplatelets should be suspended until 24 hours posttreatment. Medical management and acute post stroke care should follow the ASA guidelines.
· Any neurological deterioration should be evaluated by urgent CT scan and other evaluations as indicated according to investigator/hospital best practice.
· As in all surgical interventions, monitoring of intra-procedural blood loss is recommended so that appropriate management may be instituted.
· Excessive aspiration with the distal tip of the AXS Vecta 46 Intermediate Catheter covered by the vessel wall may cause vessel injury. Carefully investigate location of the distal tip under fluoroscopy prior to aspiration.
· There is an inherent risk with the use of angiography and fluoroscopy.
· Operators should take all the necessary precautions to limit X-Radiation doses to patients and themselves by using sufficient shielding, reducing fluoroscopy times, and modifying X-Ray technical factors where possible.
· Ensure the RHV is fully open before inserting the AXS Vecta 46 Intermediate Catheter. Avoid over- or undertightening the RHV. Do not insert or advance the AXS Vecta 46 Intermediate Catheter if resistance is encountered without careful assessment of the cause.
· When transporting the Medela Dominant Flex Pump, utilize the pump handle.