 Instructions for Use
Instructions for UseREF Pediatric E-Z Lift Stirrups, KIDS 800-0342-KIDSREF Pediatric E-Z Lift Stirrups, PUPS 800-0342-PUPS
Replacement Pads
KIDS Boot Pads 508-1296
PUPS Boot Pads 508-1295
INTENDED USE
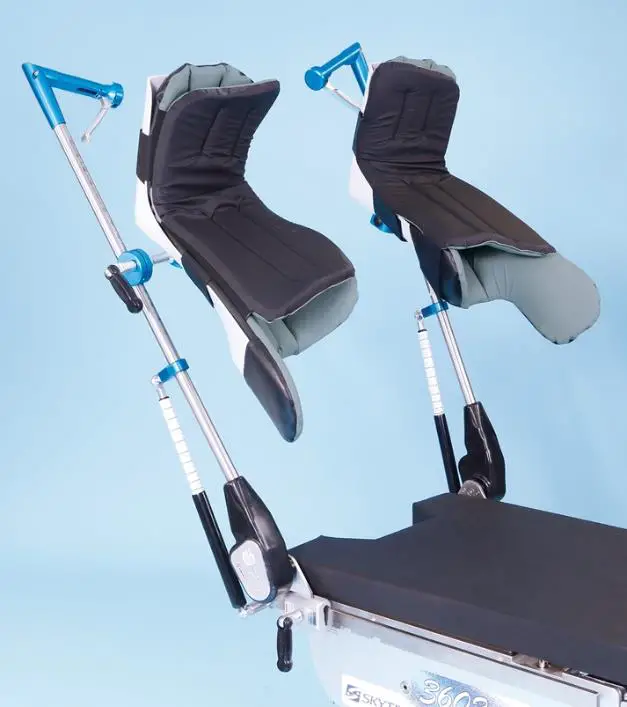
Pediatric E-Z Lift Stirrups are a patient positioning accessory used in Gynecology, Urology, and laparoscopic procedures. They provide a safe system for positioning the legs up to 160 lb. (73 kg) and 250 lb. (113 kg) patients in a variety of lithotomy/abduction positions. The intended users of this device are medical professionals within hospitals and surgery centers.
GENERAL SPECIFICATIONS
Device Dimensions (maximum)
- Height: 35” +/- 0.5” (89 cm +/- 1 cm)
- Width: 9” +/- 0.5” (23 cm +/- 1 cm)
- Depth: 14” +/- 0.5” (36 cm +/- 1 cm)
- Device Weight Per Stirrup: 8 +/- 0.5 lbs. (3.6 +/- .22 kg) (with pad)
- Range of Motion: -30° to 75° lithotomy range, -9° to 25° adduction to abduction
- Single-person installation
- Connects to seat section of surgical table
- Store stirrups on transport/storage dolly (P/N 800-0074-P sold separately)
GENERAL INFORMATION
- Product not made with Natural Rubber Latex
- This device supports a 160 lb. (73 kg) patient for 800-0342-PUPS and 250 lb. (113 kg) patient for 800-0342-KIDS proportional patient load (5’4″ (163 cm) tall patient per 99% human body model)
- Product warranty covers product from manufacturing defects for period of 2 years
- CE marked medical device according to MDR (EU) 2017/745
- Life of device is 5 years under normal use
- Store device between -4°F to +86°F (-20°C to 30°C)
IFU-800-0342-PUPS-KIDS REV 3.06 Latest Revision: 2022-01
INSTRUCTIONS
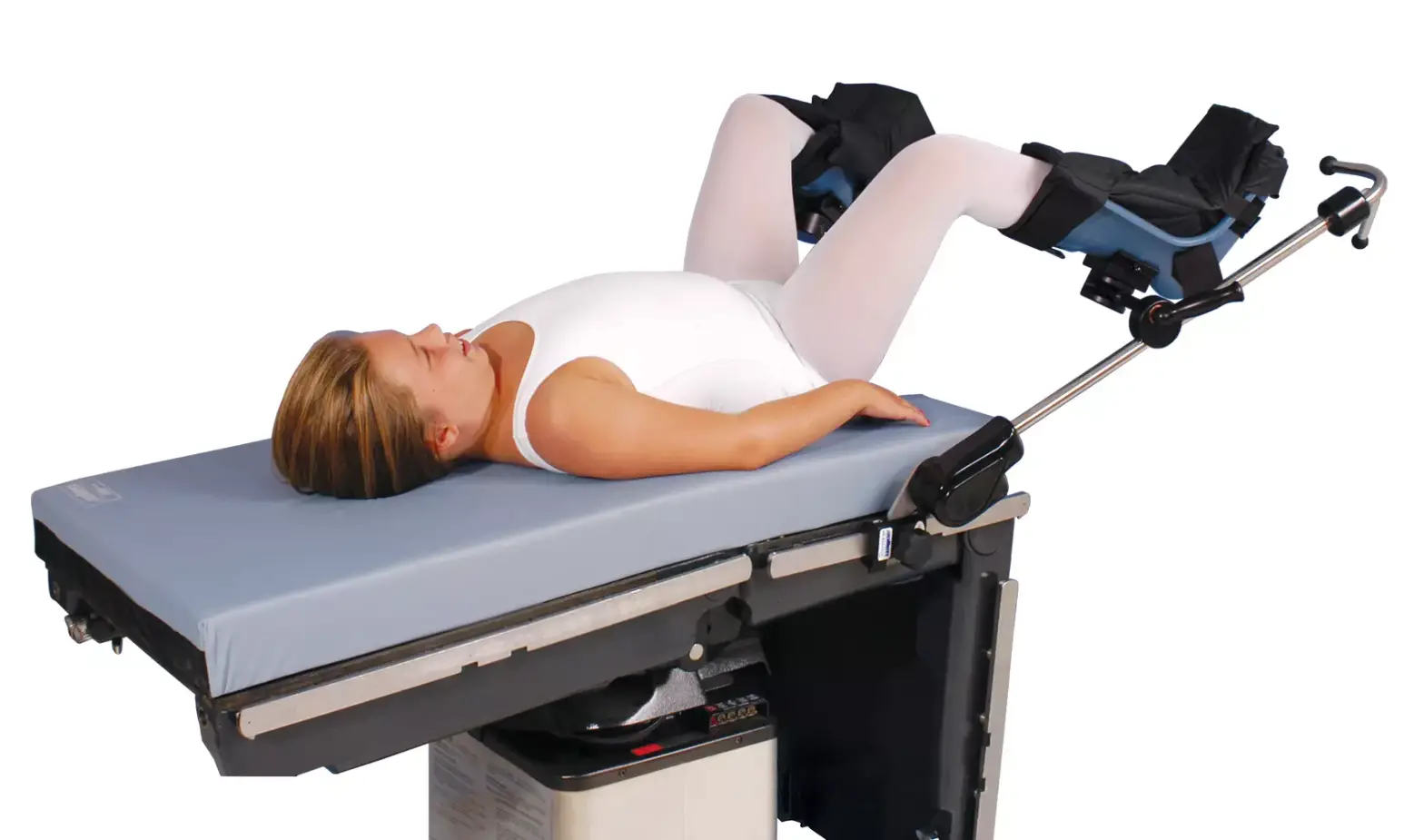
Become familiar with patient positioning device’s features before use with patient. Always practice on a nurse, physician or appropriate volunteer prior to using clinically.
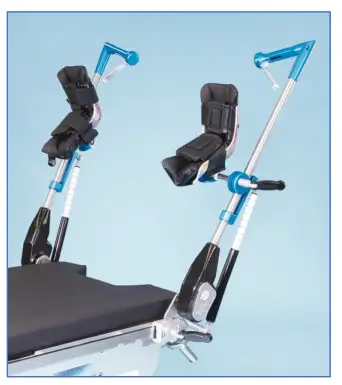
- Attach SpringLoc Clamps P/N 800-0338 on accessory rails in same locations on opposite sides of surgical table at patient’s hip joints.
- Prior to placing device into SpringLoc Clamp, identify patient’s left and right side stirrup indicated on boot label.

- Insert stirrup blade into clamp. Tighten clamp by turning handle to the right.

- Ensure patient is positioned on surgical table in accordance with procedure and surgeon requirements.
WARNING!! To prevent patient or operator injury from inadvertent stirrup movement, securely tighten accessory clamp and boot clamp.
- Support patient’s leg by grasping heel in one hand and underside of knee with other hand. Gently flex knees and transfer leg into boot, then secure boot straps.
- To achieve appropriate leg/foot position, loosen boot clamp. Adjust boot to desired position and re-tighten clamp. Ensure patient’s heels are securely seated in boot heels.
- To achieve appropriate lithotomy and abduction positions, squeeze trigger, adjust to desired position and release to lock.
Note: Free-floating boot is designed to rotate about pivot shown above through-out the full range of motion, reducing risk of superficial nerve injury.
TAKE DOWN
- Loosen clamps and remove stirrups by lifting them out of clamps
- Remove accessory clamps from side rails
COMPONENT OVERVIEW
Pediatric E-Z Lift Stirrups provide a safe system for positioning legs of up to 160 lbs. (73 kg) per patients for 800-0342-PUPS and 250 lbs. (113 kg) per patient for 800-0342-KIDS in a variety of lithotomy/abduction positions.
Other required products for use:
800-0338 SpringLoc Clamp
800-0338-EU SpringLoc Clamp
800-0338-JPN SpringLoc Clamp
800-0338-SWISS SpringLoc Clamp
Replacement Pads
508-1296 KIDS Boot Pads
508-1295 PUPS Boot Pads
DISPOSAL
- General – Prevent infection by cleaning and disinfecting product before disposal
- Packaging – Dispose packaging material via household waste according to national requirements
- SchureMed accepts back used or retired products – or dispose of product in accordance with national requirements
PRODUCT USE WARNINGS![]() WARNING! Maximum load should not exceed appropriate proportion of a patient weighing 160 lbs. (73 kg) for 800-0342-PUPS and 250 lbs. (113 kg) for 800-0342-KIDS. Use care with low-maximum load capacity surgical tables that accessory rails are not overloaded.
WARNING! Maximum load should not exceed appropriate proportion of a patient weighing 160 lbs. (73 kg) for 800-0342-PUPS and 250 lbs. (113 kg) for 800-0342-KIDS. Use care with low-maximum load capacity surgical tables that accessory rails are not overloaded.![]() WARNING! Hazards result from incorrect use. Strictly follow instructions for use with your Operating Table system.
WARNING! Hazards result from incorrect use. Strictly follow instructions for use with your Operating Table system.![]() WARNING! Do not reuse device if there are obvious signs of damage or functional issues. Consult manufacturer before reusing.
WARNING! Do not reuse device if there are obvious signs of damage or functional issues. Consult manufacturer before reusing.![]() WARNING! Surgical table load capacities may be less. Never overload a surgical table. Device is intended for mounting on surgical table side rail only.
WARNING! Surgical table load capacities may be less. Never overload a surgical table. Device is intended for mounting on surgical table side rail only.
CLEANING RECOMMENDATION
Follow current Association of periOperative Registered Nurses Journal Guidelines for proper cleaning and disinfection procedure.![]() WARNING! Adhere to standards for blood-borne pathogens from the Occupational Safety and Health Administration.
WARNING! Adhere to standards for blood-borne pathogens from the Occupational Safety and Health Administration.
Use recommended protective clothing, gloves, masks and eye protection to clean accessory.
CAUTION Strictly read/follow manufacturer’s directions for cleaning fluids. DO NOT use cleaners containing phenolics.
- Remove major contaminants from accessory with disposable materials. Follow appropriate bio-hazard waste disposal procedures.
- Apply cleaning fluid liberally to entire accessory and wipe with clean, lint-free cloth until all moisture and cleaning fluid is removed from accessory
- Let accessory dry
USER NOTICE
Any serious incident that has occurred in relation to the device should be reported to the manufacturer and the competent authority of the Member State in which the user and/or patient is established.
UDI Basic UDI-DI: 081001460F0040DU
Symbol Glossary
| Symbol | Title | Symbol Description |
| Manufacturer | Indicates the medical device manufacturer. | |
| Authorized Representative in the European Community | Indicates the authorized representative in the european community. | |
| Use-by Date | Indicates the date after which the medical device is not to be used. | |
| LOT | Batch Code | Indicates the manufacturer’s batch code so that the batch or lot can be identified. |
| REF | Catalogue or Item Number | Indicates the manufacturer’s catalogue or item number so that the medical device can be identified. |
| SN | Serial Number | Indicates the manufacturer’s serial number so that a specific medical device can be identified. |
| Caution | To indicate that caution is necessary when operating the device or control close to where the symbol is placed, or to indicate that the current situation needs operator awareness or operator action in order to avoid undesirable consequences. | |
| MD | Medical Device | Indicates the item is a medical device. |
| UID | Unique Device Identifier | Indicates a barcode as containing unique device identifier information. |
 | CE Marking | European Conformity. |
| Single Patient Use | Indicates the item is a single patient use medical device. |
![]() Manufacturer
Manufacturer SchureMed (081001460)
SchureMed (081001460)![]() Authorized Representative
Authorized Representative
Emergo Europe, Prinsessegracht 20, 2514 AP The Hague, The Netherlands
IFU-800-0342-PUPS-KIDS REV 3.06
Latest Revision: 2022-01