
INTENDED USE
The intended use is to hold the patient’s head during the operative procedure. The intended users of this device are medical professionals within hospitals and surgery centers.
INSTRUCTIONS
Become familiar with the features of the patient positioning device before use with a patient. Always practice use on a nurse, physician or appropriate volunteer prior to using clinically.
- Place the patient on the surgical table/shoulder chair in the supine position
- Once the patient is intubated, hold the patient’s head/neck and raise the shoulder chair up so the patient is in Fowler (sitting) position
- Loosen ball joint – adjust the head positioner around the patient’s head making sure the patient’s ears are uncovered – tighten the ball joint
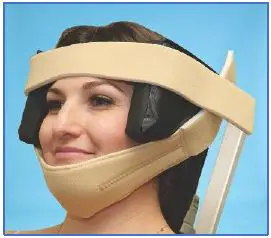
- Hold head positioner flaps alongside the patient’s head, placing the Forehead Strap across the forehead as shown. Attach securely and avoid covering the patient’s eyebrows.
- Center Chin Strap over the chin, secure the strap to one side of the head positioner. Make sure the head is properly positioned, and secure the strap’s other end to the opposite side of the head positioner.
NOTE:
The head should be securely positioned. If not, make the straps a little tighter.
COMPONENT OVERVIEW
Premium Head and Chin Straps hold a patient’s head securely during the operative procedure.
GENERAL SPECIFICATIONS
Device Dimensions (maximum)
- Length: 21”+/- 0.5” (53 cm +/- 1 cm) (Head)
- Length: 22”+/- 0.5” (56 cm +/- 1 cm) (Chin)
- Width: 2”+/- 0.5 (5 cm +/- 1 cm) (Both Head and Chin)
- Device Weight: .066 lbs. +/- .02 lbs. (.03 kg +/- .009 kg)
- Attaches to the head section of SchureMed Shoulder Chairs
- Single-person installation
GENERAL INFORMATION
- Product not made with Natural Rubber Latex
- CE marked medical device according to MDR (EU) 2017/745
- Store device between –4°F to +86°F (-20°C to 30°C)
SINGLE PATIENT USE—Please dispose after each use
USER NOTICE
Any serious incident that has occurred in relation to the device should be reported to the manufacturer and the competent authority of the Member State in which the user and/or patient is established.
 Basic UDI-DI: 081001460F0012DP
Basic UDI-DI: 081001460F0012DP
Symbol Glossary
| Symbol | Title | Symbol Description |
| Manufacturer | Indicates the medical device manufacturer. | |
| Authorized Representative in the European Community | Indicates the authorized representative in the european community. | |
| Use-by Date | Indicates the date after which the medical device is not to be used. | |
| Batch Code | Indicates the manufacturer’s batch code so that the batch or lot can be identified. | |
| Catalogue or Item Number | Indicates the manufacturer’s catalogue or item number so that the medical device can be identified. | |
| Serial Number | Indicates the manufacturer’s serial number so that a specific medical device can be identified. | |
| Caution | To indicate that caution is necessary when operating the device or control close to where the symbol is placed, or to indicate that the current situation needs operator awareness or operator action in order to avoid undesirable consequences. | |
| Medical Device | Indicates the item is a medical device. | |
| Unique Device Identifier | Indicates a barcode as containing unique device identifier information. | |
| CE Marking | European Conformity. | |
| Single Patient Use | Indicates the item is a single patient-use medical device. |
Authorized Representative
Emergo Europe, Prinsessegracht 20, 2514 AP, The Hague, The Netherlands.




















