ottobock 17B70=12 System Positioning Joint

Foreword
INFORMATION
Date of last update: 2021-05-10
- Please read this document carefully before using the product and observe the safety notices.
- Instruct the user in the safe use of the product.
- Please contact the manufacturer if you have questions about the product or in case of problems.
- Report each serious incident related to the product to the manufacturer and to the relevant authority in your country. This is particularly important when there is a decline in the health state.
- Please keep this document for your records.
These instructions for use provide you with important information on the processing of the 17B70=12 orthotic joints.
Product description
Available sizes
The 17B70=12 orthotic joint made of stainless steel is available in one size.
Components/design
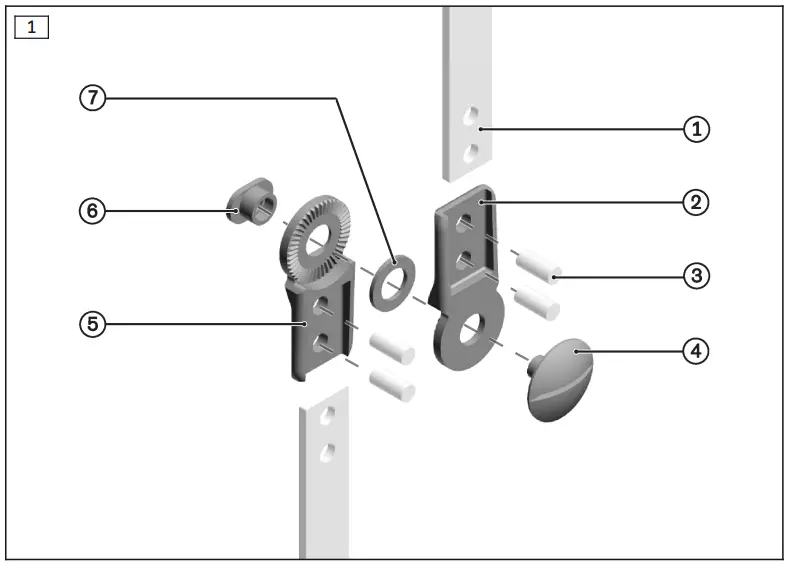
| Scope of delivery (see fig. 1) | |||
| Item | Quantity [Piece (s)] | Designation | Article number |
| 2 | 1 | Upper joint section | |
| 4 | 1 | Joint screw | 501T17=7.5x9xM5 |
| 5 | 1 | Lower joint section | |
| 6 | 1 | Bearing nut | 17Y93=7×4.8xM5 |
| 7 | 1 | Ring | 18Z3 |
| Not included in the scope of delivery (see fig. 1) | |||
| Item | Quantity [Piece (s)] | Designation | Article number |
| 1 | 1 | Side bar | 651B1=12 |
| 3 | 4 | Rivet | 506A12=4×9 |
Design
The orthotic joint angle can be adjusted in 8° increments by repositioning the joint sections.
Intended use
Indications for use
The orthotic joints are to be used exclusively in positioning orthoses for the unilateral or bilateral orthotic treatment of the lower and upper limbs on one patient.
Indications
- Functional joint contractures
- Neurological and orthopaedic disorders
Indications must be determined by the physician.
Contraindications
- Absolute Contraindications
Structural joint contractures - Relative Contraindications
The following accompanying symptoms require consultation with a physician: skin diseases and injuries, especially in case of inflammatory changes, prominent scars with swelling, reddening and hyperthermia in the area treated; lymphatic flow disorders, including unclear soft tissue swelling distal to the device; sensory and circulatory disorders.
Lifetime
The product is designed for a lifetime of 3 years when used as intended and assembled professionally.
Qualification
Patients may be fitted with the product only by trained qualified personnel.
The qualified personnel must be familiar with the handling of the various techniques, materials, machines and tools.
Safety
Explanation of warning symbols
- CAUTION: Warning regarding possible risks of accident or injury.
- NOTICE: Warning regarding possible technical damage.
Safety instructions
- CAUTION
- Excessive strain on load-bearing components
Injuries due to changes in or loss of functionality- Only use the product for the defined area of application.
- If the product has been exposed to extreme strain (e.g. due to falling), take any necessary measures (e.g. repair, replacement, inspection by the manufacturer’s customer service, etc.).
- Excessive strain on load-bearing components
- CAUTION
- Incorrect alignment or assembly
Injuries due to changes in or loss of functionality- The product may only be installed by trained, qualified personnel.
- Observe the alignment and assembly instructions.
- Incorrect alignment or assembly
- CAUTION
- Mechanical damage to the product
Injuries due to changes in or loss of functionality- Use caution when working with the product.
- Check the product for proper function and readiness for use.
- In case of changes in or loss of functionality, discontinue use of the product and have it checked by authorised, qualified personnel.
- Mechanical damage to the product
- CAUTION
- Excessive strain due to use on more than one patient
Risk of injury and loss of functionality as well as damage to the product- Use the product on only one patient.
- Observe the maintenance recommendations.
- Excessive strain due to use on more than one patient
- NOTICE
- Thermal overloading of the product
Damage due to improper thermal treatment- Do not carry out any heat treatment at temperatures above 300 °C (570 °F).
- Prior to thermal treatment, remove all temperature-critical components (such as plastic parts).
- Thermal overloading of the product
- NOTICE
- Exposure of the product to unsuitable environmental conditions
Damage, brittleness or destruction due to improper handling- Avoid storage in condensing ambient humidity.
- Avoid contact with abrasive substances (e.g. sand, dust).
- Do not expose the product to temperatures below -10 °C (14 °F) or above +60 °C (140 °F) (e.g. sauna, excessive sunlight, drying on a radiator).
- Exposure of the product to unsuitable environmental conditions
Preparing the product for use
Mounting the bar

- Required tools: 624S14=4 drill, 726S9=90×11.5 counterbore, hammer, anvil, file.
- Insert the bar into the upper/lower joint section and mark the position of the bore holes.
- Use the 624S14* drill to make the bore holes.
- Countersink the bore holes with the 726S9* counterbore.
- Position the bar in the insertion zone of the joint section and insert the rivets.
- Position the joint with the bar on the anvil and set the rivets using the hammer (see fig. 2).
- File down the projecting rivet heads on both sides.
Adjusting the joint angle
- Loosen the joint screw and adjust the joint angle.
- For the final assembly, secure the screw connection with Loctite 241.
Improving corrosion protection
Polish or sinter the product for improved corrosion protection. Ottobock recommends the 618T40* sintering powder for surface coating.
- Do not heat the material longer than 5 minutes at max. 150 °C for surface coating.
Cleaning
Promptly clean the product after contact with water containing salt, chlorine or soap, or if it gets dirty.
- Rinse the orthotic joint with clean, fresh water and remove dirt.
- Dry with a cloth or allow to air dry.
Maintenance
INFORMATION
The product may be exposed to increased loads by the patient.
- Shorten the maintenance intervals according to the expected loads.
The manufacturer requires at least a semi-annual inspection of the product to verify functionality and check for wear.
Spare parts are listed in the section “Components/design”.
Disposal
Dispose of the product in accordance with national regulations.
Legal information
All legal conditions are subject to the respective national laws of the country of use and may vary accordingly.
Liability
The manufacturer will only assume liability if the product is used in accordance with the descriptions and instructions provided in this document. The manufacturer will not assume liability for damage caused by disregarding the information in this document, particularly due to improper use or unauthorised modification of the product.
CE conformity
The product meets the requirements of Regulation (EU) 2017/745 on medical devices. The CE declaration of conformity can be downloaded from the manufacturer’s website.