MATIK
1P200 / 1P200-KD Instruction for use
Instruction for use
1P200 / 1P200-KD MATIK
| MATIK – 1P200(-KD) Instructions for use for Prosthetists/Orthotists Please read before using or working on the device | 1P20099 2021-04 |
Pass on § 3, 7, 8, and 9 of these instructions to the Patient
COMPONENTS INCLUDED
| Designation | Ref. | Included/Sold separately |
| Knee | 1P200(-KD) | Included |
| One-part cosmetic cover | 1G18 | Sold separately |
DESCRIPTION, PROPERTIES, AND MECHANISM OF ACTION
A. Description
A 4-axis pneumatic knee is available in two different versions:
- 1P200: Male pyramid proximal adapter, suitable for patients who have had a transfemoral amputation or hip disarticulation.
- 1P200-KD: Threaded proximal adapter (M36×1.5), suitable for patients with a long amputation stump or who have had a knee disarticulation.
For both versions, the distal connection is made via a male pyramid connector.

B. Properties
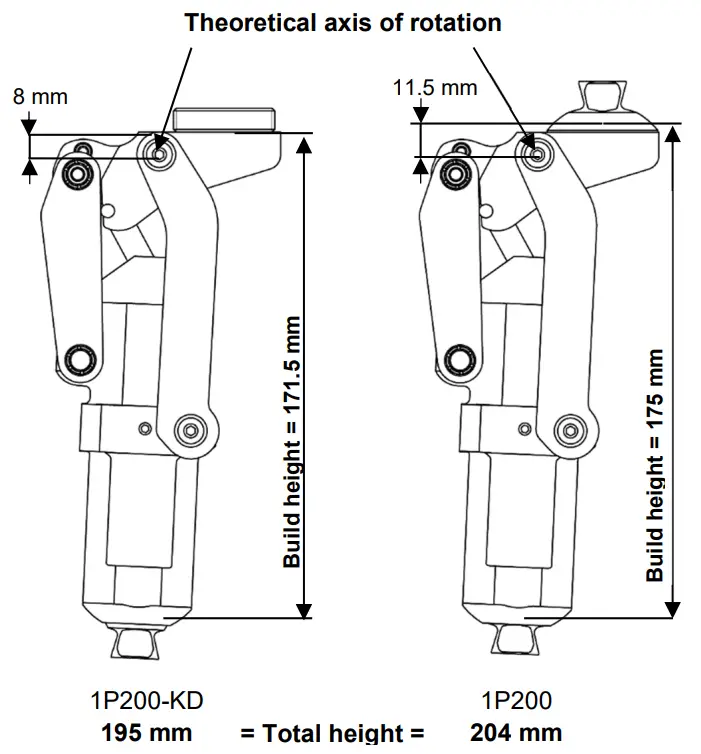
| Ref. | 1P200 | 1P200-KD |
| Weight | 743 g | |
| Maximum flexion | 170° | |
| Maximum patient weight (Including carried load) | 125 kg | |
 This device has undergone a three-million-cycle test, in accordance with standard NF EN ISO 10328 at a P6 load level (i.e. 125 kg), which corresponds to a service life of 4 to 5 years, depending on the patient’s mobility level.
This device has undergone a three-million-cycle test, in accordance with standard NF EN ISO 10328 at a P6 load level (i.e. 125 kg), which corresponds to a service life of 4 to 5 years, depending on the patient’s mobility level.
C. Mechanism of action
When an extension, the system of linkages ensures that the instantaneous center of rotation is to the rear of the load line, which keeps the knee stable during the stance phase. The speed of every flexion and extension movement can be adjusted independently. The extension assist is not adjustable.
INTENDED USERS/INDICATIONS
This medical device is supplied to healthcare professionals (prosthetists/orthotics) who then instruct the patient on how to use it.![]() The prescription is drawn up by a doctor. The doctor works with a prosthetist/orthotist to assess whether the patient is suited to using the device.
The prescription is drawn up by a doctor. The doctor works with a prosthetist/orthotist to assess whether the patient is suited to using the device. This device should be used on ONE PATIENT ONLY. It must not be reused on other patients.
This device should be used on ONE PATIENT ONLY. It must not be reused on other patients.
This device is exclusively intended to be used as prosthetic equipment for patients who have undergone transfemoral amputation, hip disarticulation, or knee disarticulation. It is specifically recommended for active or slightly active patients (L2/L3) and allows them to perform everyday activities.
Maximum weight (including carried load): 125 kg![]() Not suitable for children.
Not suitable for children. ![]() The maximum knee flexion is 170°. This flexion may, however, be limited by the volume of the socket or by the cosmetic covering.
The maximum knee flexion is 170°. This flexion may, however, be limited by the volume of the socket or by the cosmetic covering.
CLINICAL BENEFITS
The device allows:
- Everyday mobility with minimum maintenance
- The patient chose a lighter and smaller knee.
- Flexion of up to 170° for sitting or kneeling.
- Good clearance between the foot and the ground in the swing phase, to avoid contact.
- High levels of stability and safety in the stance phase thanks to the 4-axis joint geometry.
ACCESSORIES AND COMPATIBILITIES
| Ref. | 1P200 | 1P200-KD | |
| Proximal adapter | Anchor | 1K179+ 1K183 1K173 or 1K176 1K177 (max. weight 100 kg) | 1K179 |
| Connectors | 1K172 or 1K207-HD | 1K184 | |
| Distal adapter | Tube and connector | 1D41-HD and 1G01-HD 1D52-P6 | |
ASSEMBLY AND FITTING TO THE PATIENT
A. Aligning
Static alignment:
In the sagittal plane, the load line passes through the greater trochanter and between 0 and 10 mm in front of the axis of the knee.
Align the foot in compliance with the manufacturer’s instructions.
In the frontal plane, the load line passes through the middle of the socket, through the middle of the knee, and between the first and second toes. 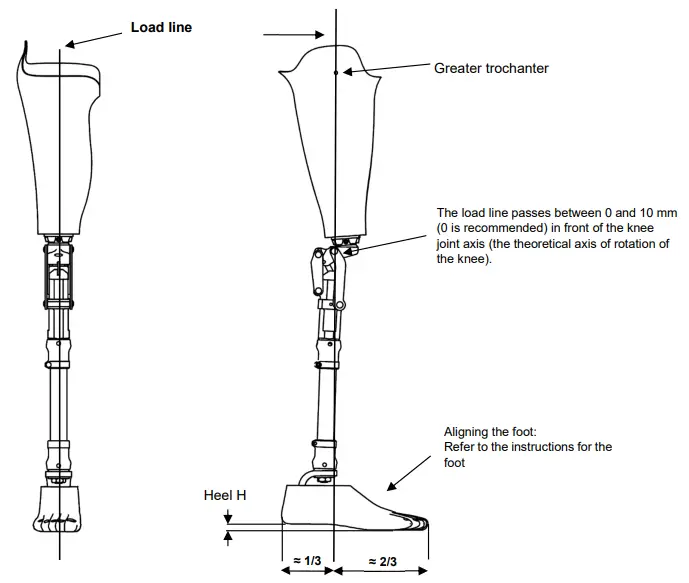
Dynamic alignment: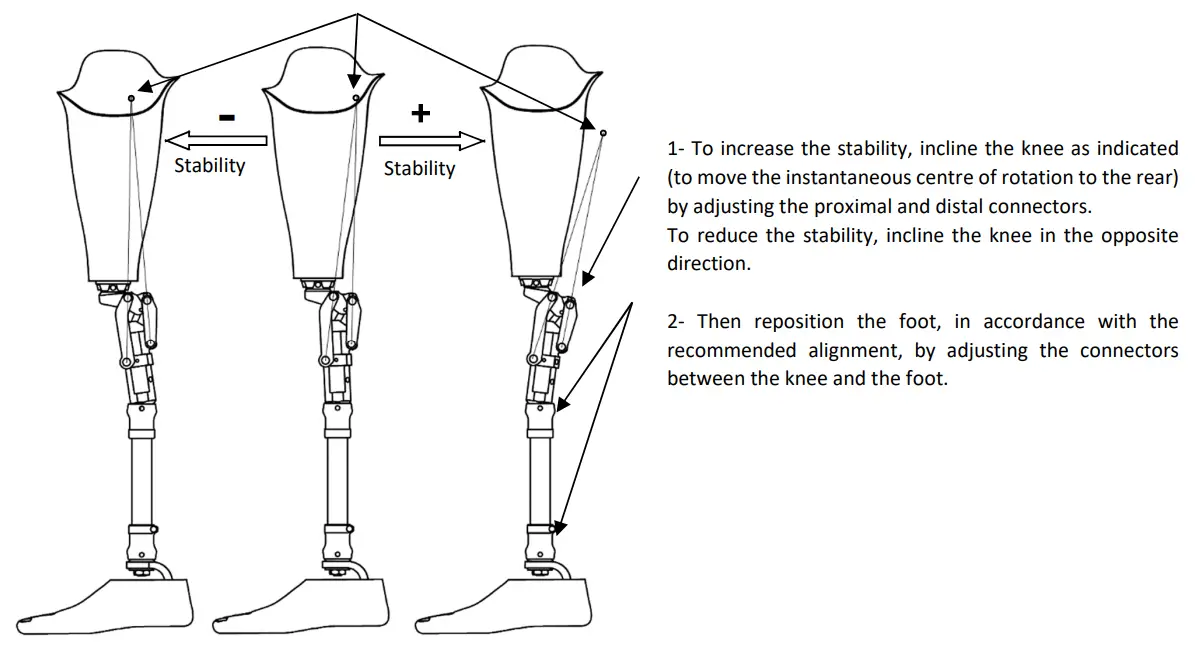
The threaded M36×1.5 adapter for the KD version does not accommodate any alignment adjustment. The socket must therefore be made accordingly.
Check the alignment using suitable equipment (line laser, plumb line, etc.)
B. Adjusting the walking pace
On delivery, the knee is set up for walking at a medium pace.
For patient safety reasons, the prosthetist/orthotist must begin the adjustment process with the knee set to its factory setting and with the patient walking between parallel bars.![]() The model of the foot used may have an effect on the adjustment of the knee. The knee should be readjusted whenever the foot is changed.
The model of the foot used may have an effect on the adjustment of the knee. The knee should be readjusted whenever the foot is changed.
Prioritize patient safety in all circumstances, particularly to ensure that slopes and stairs can be negotiated safely.![]() After performing any maintenance the knee must be readjusted.
After performing any maintenance the knee must be readjusted.
To adapt the knee to the patient’s pace of walking, adjust hex set screws E and F using an Allen (hex) key.
It is advisable to perform these adjustments in the following order:
- Adjusting the Flexion (F)
Turn hex set screw F: Screw in the set screw to reduce the angle of flexion, and unscrew to increase it. - Adjusting the Extension (E)
Turn hex set screw E: Screw in to increase the cushioning of the impact when the knee reaches full extension.
![]() Once these adjustments have been made, ensure that the knee returns to the full extension when walking slowly to ensure that there is no risk of the patient falling.
Once these adjustments have been made, ensure that the knee returns to the full extension when walking slowly to ensure that there is no risk of the patient falling.
C. Finishing
It is advisable to reinforce the cosmetic cover in front of the joint to prolong its service life.
Maximum flexion stop:
When the patient is crouching or kneeling, the socket must come into contact with the rear flexion limit on the rear linkage. It must not touch the rear linkage, or the plate, since this might damage or destroy the knee.![]() The cosmetic cover may modify the initial knee settings and limit certain movements. It is advisable to always check the operation of the knee, once the cosmetic cover has been fitted.
The cosmetic cover may modify the initial knee settings and limit certain movements. It is advisable to always check the operation of the knee, once the cosmetic cover has been fitted.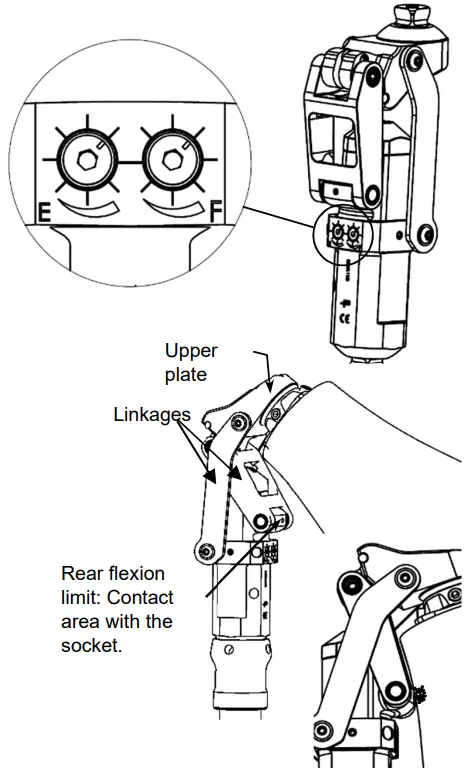
DETECTING ANY MALFUNCTIONING
If you notice that the device is behaving abnormally or if its characteristics feel different, or if it has been badly knocked, consult your prosthetist/orthotist.
WARNINGS, CONTRAINDICATIONS, SIDE EFFECTS
A. Warnings![]() Caution, the patient may need to take time to get used to a new knee.
Caution, the patient may need to take time to get used to a new knee.![]() To reduce the risk of falling when going downstairs, it is advisable to hold on to the handrail.
To reduce the risk of falling when going downstairs, it is advisable to hold on to the handrail.![]() So as not to damage the knee, do not apply talcum powder to eliminate rubbing noises – use silicon spray instead. Talc degrades the mechanical components, which might cause a malfunction and possibly a fall.
So as not to damage the knee, do not apply talcum powder to eliminate rubbing noises – use silicon spray instead. Talc degrades the mechanical components, which might cause a malfunction and possibly a fall.![]() PROTECTOR disclaims all liability if talcum powder is used.
PROTECTOR disclaims all liability if talcum powder is used.![]() It is possible to trap a finger or pinch clothing in the knee joint. To avoid any injury caused by the movement of the joint, make sure that no one’s hand or fingers are near the mechanism.
It is possible to trap a finger or pinch clothing in the knee joint. To avoid any injury caused by the movement of the joint, make sure that no one’s hand or fingers are near the mechanism.![]() The knee is resistant to bad weather but needs to be dried immediately if it does get wet. Any water that gets into the pneumatic mechanism may lead to the deterioration of the extension assist spring and thus the destruction of the knee. It must, therefore, be protected against any ingress of water
The knee is resistant to bad weather but needs to be dried immediately if it does get wet. Any water that gets into the pneumatic mechanism may lead to the deterioration of the extension assist spring and thus the destruction of the knee. It must, therefore, be protected against any ingress of water
B. Contraindications![]() The following are strictly prohibited: removing the covers, screwing in, or unscrewing any screws on the knee with the exception of the two adjustment screws E and F.
The following are strictly prohibited: removing the covers, screwing in, or unscrewing any screws on the knee with the exception of the two adjustment screws E and F.![]() Never lubricate the knee pins; this can cause them to degrade rapidly.
Never lubricate the knee pins; this can cause them to degrade rapidly.![]() The knee has been designed for patients with a maximum weight of 125 kg (including carried load).
The knee has been designed for patients with a maximum weight of 125 kg (including carried load).![]() The warranty does not cover damage or degradation caused by misuse, unsuitable alignment, use without suitable protection in a very dusty environment, or any other inappropriate use.
The warranty does not cover damage or degradation caused by misuse, unsuitable alignment, use without suitable protection in a very dusty environment, or any other inappropriate use.![]() The knee must not be exposed to environments that might induce corrosion of the metal components (freshwater, seawater, chlorinated water, acids, etc.).
The knee must not be exposed to environments that might induce corrosion of the metal components (freshwater, seawater, chlorinated water, acids, etc.).![]() Showering or taking a bath while wearing the prosthesis is prohibited; this might impair its properties and its correct operation.
Showering or taking a bath while wearing the prosthesis is prohibited; this might impair its properties and its correct operation.![]() Never leave this device near a source of heat since it might burn or release toxic fumes.
Never leave this device near a source of heat since it might burn or release toxic fumes.![]() The use of solvents is prohibited.
The use of solvents is prohibited.
C. Side effects
There are no side effects directly associated with the device.
Any serious incident that occurs which relates to the device must be reported to the manufacturer and to the competent authority of the member state in which the incident occurred.
MAINTENANCE, STORAGE, DISPOSAL, AND SERVICE LIFE
A. Maintenance/cleaning![]() You can clean the knee using a damp sponge
You can clean the knee using a damp sponge![]() Do not immerse the knee or hold it under running water
Do not immerse the knee or hold it under running water![]() Dry your knee immediately if it is exposed to bad weather or accidental splashing.
Dry your knee immediately if it is exposed to bad weather or accidental splashing.
The stops may need to be replaced:
• Spare stop: EE014 (2 are required)
B. Storage![]() Service and storage temperature: -10°C to +40°C
Service and storage temperature: -10°C to +40°C
Relative humidity of the air: no restrictions
C. Disposal
The various components of this device are considered special waste: elastomer, plastic, aluminum, titanium, steel, and brass. They must be treated in accordance with current, applicable legislation.
D. Service life
It is advisable to arrange for a prosthetist/orthotist to check the product once a year.
DESCRIPTION OF THE SYMBOLS
 | Manufacturer |  | Identified risk | CE marking and year of first declaration of conformity |
REGULATORY INFORMATION
![]() This product is a CE-marked medical device that is certified as conforming with regulation (EU) 2017/745
This product is a CE-marked medical device that is certified as conforming with regulation (EU) 2017/745
NAME AND ADDRESS OF MANUFACTURER
 PROTEOR SAS
PROTEOR SAS
6 rue de la Redoute – 21850 Saint-Apollinaire – France
Tel.: +33 3 80 78 42 42 –
Fax: +33 3 80 78 42 15
[email protected] –
www.proteor.com