Temporary Crown and Teeth
Instructions
Sprint Ray EU Temporary Crown & Teeth Instruction for use
Indication
Sprint Ray EU Temporary Crown & Teeth is a resin for the generative production of long term temporary dental restorations based on image projection systems.
Contraindication
Sprint Ray EU Temporary Crown & Teeth restorations are contraindicated .
- … if a patient is known to be allergic to any of the ingredients.
- … for bridges with more than one pontin.
- … for every application that is not part of the indication (see above).
Requirements
Software:
– Ray Ware (CAD)
Hardware:
– Pro Desktop 3D (3D Printing)
– Pro Wash / Dry (Cleaning)
– Pro Cure (Post Curing)
Sprint Ray, Inc., 3577 North Figueroa St., Los Angeles, CA 90065
Material
Sprint Ray EU Temporary Crown & Teeth consists of functional (meth)acrylic resins and inorganic fillers with particle sizes from 0.4 to 3 microns.
Geometric presetting
Connector areas (minimum):
anterior bridges 12 mm²
posterior bridges 14 mm ²
Minimum wall thickness:
occlusal 1.5 mm (central fissure)
circumferential 1 mm
Material parameters
Radiation penetration depth controllable by exposure time
≥ 100 μm (4 mils)
≥ 125 μm (5 mils)
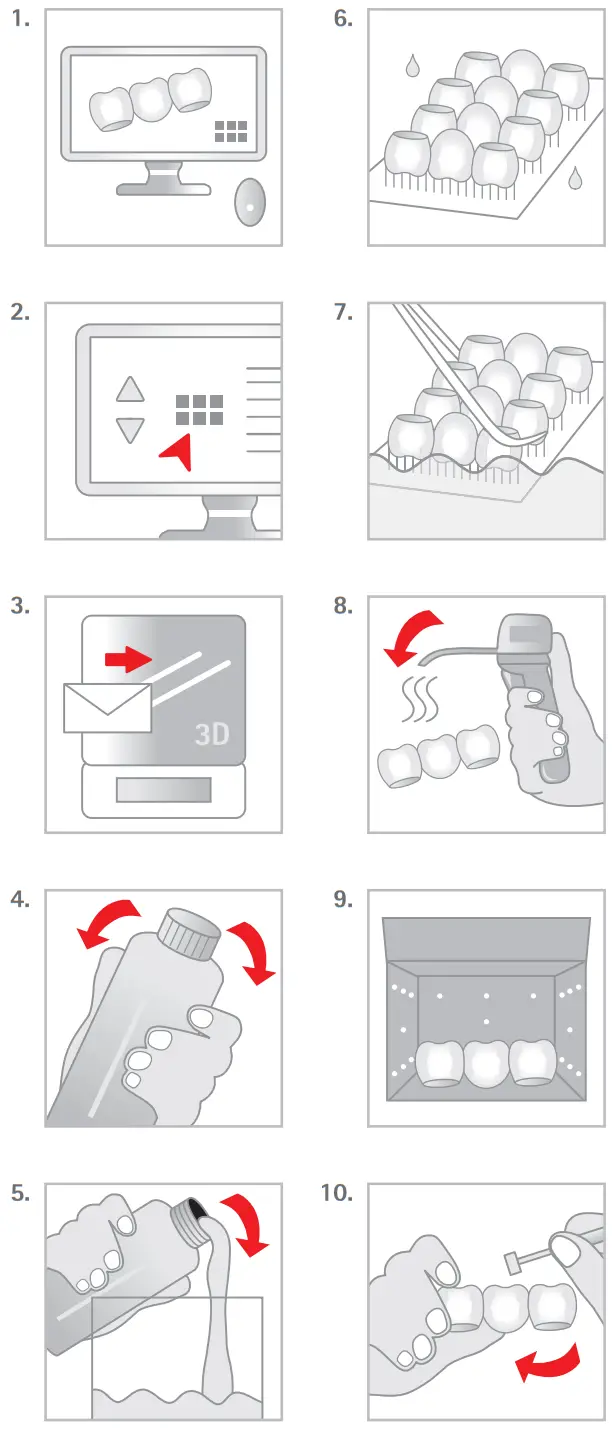
Manufacturing process (fi g. 1-10)
- Prepare data (Ray Ware).
- Choose process parameters (build style, etc.).
- Transfer prepared data to 3D printing device.
- Prepare 3D printing – shake bottle.
- Fill resin tank of 3D printing device.
- Build the parts.
- Clean parts (Propwash / Dry):
– blow surplus resin out of cavities with pressured air before washing
– wash 5 min. / 5 min. - Dry parts (Propwash / Dry):
– 3 min.
– additional: dry with pressured air as required - Post curing (Procure): 5 min. / 60°C.
- Finish parts.
Individualization and cementation
The temporary restorations can be individualized by light curing composite materials. The reduction of the restoration should not exceed 0.3 mm (incisal and vestibular area). Please follow the instructions of the light curing composite manufacturers. For cementation common provisional dental cements (free of Eugenol) and adhesives are suitable. The inner areas of the crown should be conditioned by sandblasting or other commonly used techniques. Please follow the instructions and indications of the corresponding manufacturers.
Notice
Please follow the instructions provided by the software manufacturer in terms of parameter settings and design recommendations. Please follow the instructions provided by the hardware manufacturer in terms of parameter settings/ printing & post curing recommendations. To avoid detrimental effects on material quality do not expose the liquid material to irradiation under any circumstances. Deviations from the described manufacturing processes or storing conditions may lead to different mechanical and optical properties of the material. Ensure personal protective gear during processing.
Caution: Polymerized resins are chemically resistant – avoid stains on clothing! Avoid any contact with skin and eyes! In case of accidental contact, rinse with adequate running water, consult a doctor if necessary. The lot number and the best before date are indicated on each packaging. In case of claims please always indicate the lot number of the product. Do not use the product after expiry of the best before date. Dispose of contents/container according to official regulations.
Hazard statements
Causes skin irritation. May cause an allergic skin reaction.
Causes serious eye damage. May cause respiratory irritation.
Harmful to aquatic life with long lasting effects.
* These data come from measurements of a representative sample that was determined as part of our quality assurance.
** According to internal design and requirements specifications
| Physical properties* | Ordering information |
| Sprint Ray EU Temporary Crown & Teeth | Sprint Ray EU Temporary Crown & Teeth |
| Flexural strength MP (ISO 4049): > 100 | 1 kg: ≤ 405 nm A1 REF: SRE-1001441 |
| Modulus of elasticity MP (ISO 4049): > 2000 | A2 REF: SRE-1001442 A3 REF: SRE-1001443 A3.5 REF: SRE-1001617 |
| Water absorption crumple con ISO 10477 | B1 REF: SRE-1001618 B2 REF: SRE-1001623 bleach REF: SRE-1001624 |
| Solubility conformed a ISO 10477 | 500 g: ≤ 405 nm A1 REF: SRE-1001612 A2 REF: SRE-1001613 A3 REF: SRE-1001614 |
| Inorganic filler Riempitivo inorganic: 40.0 m-% | A3.5 REF: SRE-1001620 B1 REF: SRE-1001622 B2 REF: SRE-1001625 bleach REF: SRE-1001626 |
| This side up. | |
| Temperature limit | |
| CE mark | |
| Danger | |
| Do not use if package is damaged. | |
| Consult instructions for use. | |
| Keep away from sunlight. Rx only QTY: 1EA | |
| Medical Device | |
| Use by date | |
| Catalogue number | |
| Lot number | |
| Date of manufacture |
![]() Manufacturer:
Manufacturer:
pro3dure medical GmbH
Am Burger 13 · 58642 Iserlohn, Germany
Phone: +49 (0)2374 920050-0 · Fax +49 (0)2374 920050-50
[email protected] · www.pro3dure.com ·
Made in Germany
Distributor:
Sprint Ray Europe GmbH
Brunnenweg 11 · 64331 Weiterstadt · Germany