Adherium BZT1 Breztri+ Aerosphere Hallie Sensor Instruction Manual
Intended Use
The Breztri+™ sensor is intended for single patient use in the home environment as an electronic data capture accessory for recording actuations of prescribed Inhaler medication. This may be used in the following applications:
- In self-management, where patients need to track their medication use as part of their management plan.
- In clinical practice, where specialists, general practitioners, nurses, and educators need to know if a patient has actuated their prescribed Inhaler medication.
- In clinical trials, where researchers need to know when a patient has actuated their trial Inhaler medication.
The Breztri+™ sensor is compatible only with Breztri Aerosphere™ Inhaler.
The Breztri+™ sensor is not intended to indicate remaining quantity of medication in an Inhaler and does not include a dose counting function.
Important User Information
![]() Warnings identify actions or situations that could lead to personal injury. Take note of all warnings before using the Breztri+™ sensor.
Warnings identify actions or situations that could lead to personal injury. Take note of all warnings before using the Breztri+™ sensor.
![]() Precautions identify actions or situations that could damage the Breztri+™ sensor or other equipment, or affect the accuracy or availability of recorded actuation data.
Precautions identify actions or situations that could damage the Breztri+™ sensor or other equipment, or affect the accuracy or availability of recorded actuation data.
 Notes contain advisory information about some aspect of the Breztri+™ sensor or its use.
Notes contain advisory information about some aspect of the Breztri+™ sensor or its use.
Warnings and Precautions
![]() Warnings
Warnings
- To ensure your Inhaler functions correctly, do not use the Breztri+™ sensor with any other Inhaler or medication than that indicated on the Breztri+™ sensor label.
- Refer to the labelling provided with the Inhaler for instructions on use. Carry out all steps required to use your Inhaler according to the instructions.
- The Breztri+™ sensor is intended to track medication usage. It is not intended to diagnose your condition or to replace the diagnosis of a licensed physician.
- The Breztri+™ sensor does not contain a dose counter. Do not use data collected by the Breztri+™ sensor to determine the number of doses remaining in a medication canister.
- This manual does not provide information on how to use prescription medication, and is not intended to replace the advice provided by a health professional.
- Directions for using prescription medication should be obtained from a health professional and followed accordingly. Any questions related to prescription medication should be referred to a health professional.
- If your prescribed Inhaler changes, do not use it with this Breztri+™ sensor. Only use a Breztri+™ sensor that is labeled as compatible with your prescribed Inhaler.
![]() Precaution
Precaution
- The Breztri+™ sensor is a batterypowered electronic device. Do not immerse the Breztri+™ sensor in water. Do not use the Breztri+™ sensor if it is not in good condition.
- Take care to not spill liquids on the Breztri+™ sensor or immerse it in water. Do not expose the Breztri+™ sensor to excessive perspiration during exercise.
- For hygiene and data integrity reasons, do not use the Breztri+™ sensor with more than one patient.
- Remove the Breztri+™ sensor from the Inhaler before cleaning the Inhaler.
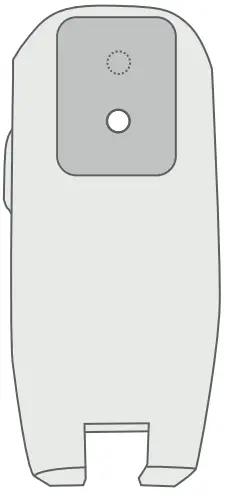
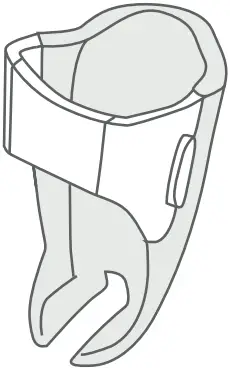
What is the Breztri+™ Sensor?
The Breztri+™ sensor is a companion tool that tracks your prescribed medication use.
The Breztri+™ sensor is a small batterypowered electronic data logger that attaches to an Inhaler. The Breztri+™ sensor contains an electronic clock and calendar that is used to log the date an time of Inhaler actuation. Usage data can be uploaded via a wireless Bluetooth® connection.
![]() Keep the Breztri+™ sensor outside MRI scanner rooms.
Keep the Breztri+™ sensor outside MRI scanner rooms.
Breztri+™ Sensor Components
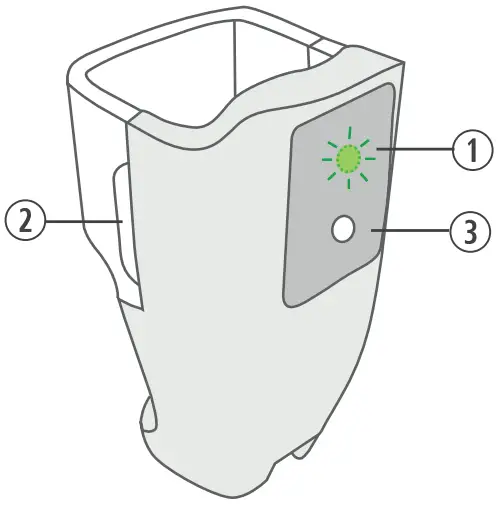
- Door Latch
- Light Emitting Diode (LED)
- Status Button for pairing, battery check and manual upload
Compatible Inhalers
The Breztri+™ sensor is designed to work only with the Breztri Aerosphere™ Inhaler as indicated on the Breztri+™ sensor label.
The Breztri+™ sensor intended population is identical to the intended population of the compatible Inhaler.
Preparation for First Use
Setting Up Bluetooth® Communications
The Breztri+™ sensor must be paired with a compatible Bluetooth® phone or tablet computer in order to sync stored usage data.
Download a compatible mobile app to communicate with your sensor.
Ensure Bluetooth® is enabled on your phone or tablet computer. Open the compatible mobile app and follow the prompts to initiate the pairing process.
- Hold the Breztri+™ sensor in an upright position.

- Press and hold the Status Button for 1 second.

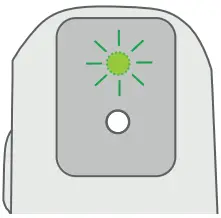
- The Breztri+™ sensor will wake up and the LED will flash green.

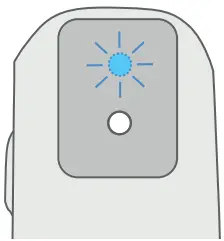
After 5 seconds the Breztri+™ sensor will automatically enter Bluetooth® pairing mode and the LED will flash blue for 60 seconds.
- Follow the pairing prompts on the mobile app to complete pairing.
If the LED is not flashing blue following wake up or the time limit is reached, press the Status Button 3 times to enter pairing mode again. The LED will flash blue when the Breztri+™ sensor is in pairing mode.
The LED will flash green to indicate successful pairing.
 If pairing is not successful within 60 seconds the LED will flash red. The pairing process can be repeated if necessary.
If pairing is not successful within 60 seconds the LED will flash red. The pairing process can be repeated if necessary.
 The Breztri+™ sensor can only be paired with a single phone or tablet computer at one time. Successful pairing with a new phone or tablet computer will remove the current pairing. If pairing on the new phone or tablet computer fails, the existing pairing will be retained.
The Breztri+™ sensor can only be paired with a single phone or tablet computer at one time. Successful pairing with a new phone or tablet computer will remove the current pairing. If pairing on the new phone or tablet computer fails, the existing pairing will be retained.
Installing and Removing an Inhaler
Installation
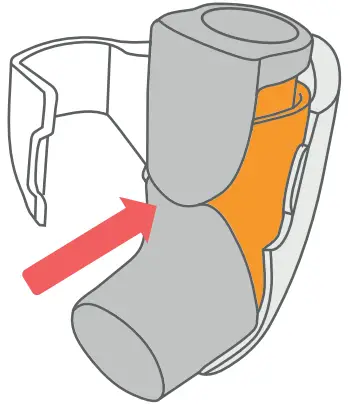
- To install the Inhaler hold the Breztri+™ sensor in an upright position.

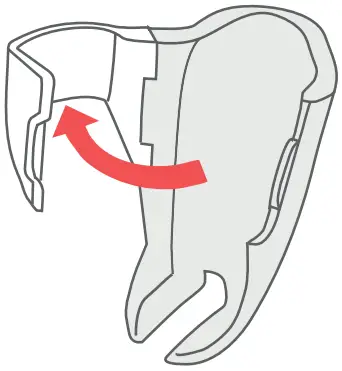
- Open the door of the Breztri+™ sensor by pulling the Door Latch.

- Push the Inhale into the Breztri+™ sensor.

- Close the door so it clicks into place.

Removal
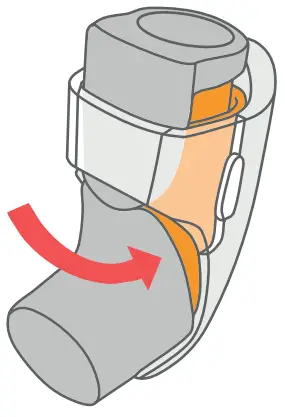
- To remove the Inhaler hold the Breztri+™ sensor in an upright position.
- Pull the Door Latch to open the door of the Breztri+™ sensor.
- Pull the Inhaler to remove it from the Breztri+™ sensor.
- Close the door so it clicks into place.
Delivering a Dose of Medication
![]() Refer to the labelling provided with the Inhaler for instructions on use. Carry out all steps required to use your Inhaler according to the instructions.
Refer to the labelling provided with the Inhaler for instructions on use. Carry out all steps required to use your Inhaler according to the instructions.
The Breztri+™ sensor LED flashes green, orange, or red (to indicate battery level) 3 seconds after detecting the medication actuation.
If the LED flashes red or there is no flash, the Breztri+™ sensor is not monitoring actuations. Check the battery level according to Reviewing the Battery Level.
![]() This manual does not provide information on how to use prescription medication, and is not intended to replace the advice provided by a health professional.
This manual does not provide information on how to use prescription medication, and is not intended to replace the advice provided by a health professional.
Directions for using prescription medication should be obtained from a health professional and followed accordingly. Any questions related to prescription medication should be referred to a health professional.
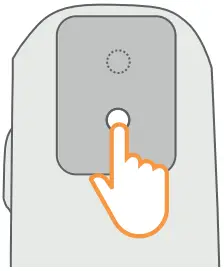
![]() Ensure your thumb is positioned at the base of the Breztri+™ sensor so that the Inhaler actuation is recorded.
Ensure your thumb is positioned at the base of the Breztri+™ sensor so that the Inhaler actuation is recorded.
 The Breztri+™ sensor does not detect or record the quantity of medication delivered by the Inhaler.
The Breztri+™ sensor does not detect or record the quantity of medication delivered by the Inhaler.
To use the Breztri+™ sensor with a spacer, hold the Breztri+ sensor in one hand with the thumb at the base of the Breztri+™ sensor and the index finger and second finger on top of the medication canister. Use the other hand to support the spacer chamber.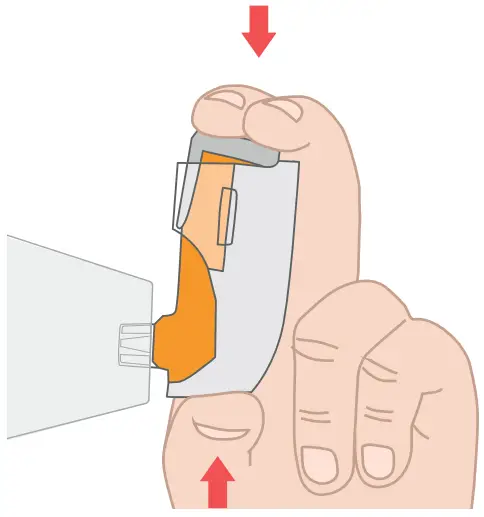
Reviewing Inhaler Usage Data
The Inhaler usage history can be automatically uploaded via a compatible paired phone or tablet computer.
The upload of new information will occur automatically as long as the Breztri+™ sensor is within range (10 meters or 32 feet) of the phone or tablet computer with Bluetooth® enabled.
![]() When travelling ensure your phone or tablet computer is set to the local time zone for accuracy of Inhaler actuation data.
When travelling ensure your phone or tablet computer is set to the local time zone for accuracy of Inhaler actuation data.
Manually Uploading Stored Information
The Breztri+™ sensor can manually upload data to a paired Bluetooth® phone or tablet computer.
- Hold down the Status Button for 3 seconds until the LED shows white, then release.
- The LED will flash white while the Breztri+™ sensor attempts to upload, then flash green if the upload is successful.
 The Breztri+™ sensor LED will flash red if the upload is not successful.
The Breztri+™ sensor LED will flash red if the upload is not successful.
Check the Breztri+™ sensor is within range of the paired phone or tablet computer, and ensure that Bluetooth® communications are enabled
Reviewing the Battery Level
The LED on the Breztri+™ sensor indicates the battery level after detecting a medication actuation, or when the Status Button is pressed once.
| LED Colour | Battery Status |
| Green | The battery level is good. The Breztri+™ sensor is monitoring Inhaler actuations. |
| Orange | The battery level is low. The Breztri+™ sensor is still monitoring Inhaler actuations. |
| Red | The battery is depleted. The Breztri+™ sensor has stopped monitoring Inhaler actuations. |
| No Flash | The battery is depleted, or the Breztri+™ sensor has failed, e.g. due to liquid immersion or mechanical stress. |
![]() The battery is not rechargeable.
The battery is not rechargeable.
Once depleted, a new Breztri+™ sensor will be required.
Cleaning the Breztri+™ Sensor and Inhaler
Check the instructions from the Inhaler manufacturer for keeping the Inhaler clean and functional.
![]() Remove the Breztri+™ sensor from the Inhaler before cleaning the Inhaler. Replace the Breztri+ sensor when the Inhaler is dry.
Remove the Breztri+™ sensor from the Inhaler before cleaning the Inhaler. Replace the Breztri+ sensor when the Inhaler is dry.
Keep the Breztri+™ sensor clean and free of chemicals, steam, water and dust.
Clean the outside plastic enclosure with a lightly dampened cloth. Check the Breztri+™ sensor is clean and repeat if necessary.
Leave it to dry in a warm place that is less than 30°C (86°F).
![]() The Breztri+™ sensor is a batterypowered electronic device. Do not immerse the Breztri+™ sensor in water. Do not use the Breztri+™ sensor if it is not in good condition.
The Breztri+™ sensor is a batterypowered electronic device. Do not immerse the Breztri+™ sensor in water. Do not use the Breztri+™ sensor if it is not in good condition.
The cleaning method is intended to support single patient use. If the sensor is contaminated by another individual, it is recommended that the Breztri+™ sensor be discarded and replaced with a new sensor.
Storage
To help maintain battery capacity, store your Breztri+™ sensor below 30°C (86°F).
Keep out of direct sunlight and avoid extreme temperatures.
The Inhaler medication has its own storage recommendations. Refer to the labelling provided with the Inhaler medication.
Troubleshooting
If the Breztri+™ sensor is not responding to Bluetooth® communications, check the Breztri+™ sensor battery level as per Reviewing the Battery Level in this manual.
Ensure Bluetooth® is enable in your phone or tablet computer.
Other wireless communications equipment such as wireless home network devices, mobile phones, cordless telephones and their base stations, walkie-talkies, and equipment such as contactless payment or anti-theft systems, can affect the Breztri+™ sensor, and should be kept at least 30cm (12in) away.
Increase the separation distance between the Breztri+™ sensor and any such devices if this could be causing problems.
EU only: Any serious incident that has occurred in relation to the Breztri+™ sensor should be reported to Adherium and the health authority of the Member State where it occurred.
Servicing
Contact the supplier for Breztri+™ sensor servicing. Do not attempt to open or service the Breztri+™ sensor. Tampering with the Breztri+™ sensor voids the warranty.
Disposal
Dispose of or recycle the Breztri+™ sensor in accordance with regulations for your country, as applicable for electronic devices containing a lithium coin-cell battery. Ensure that the Inhaler is removed from the Breztri+™ sensor prior to disposal.
EU only: Do not dispose of the Breztri+™ sensor as unsorted municipal waste. The Breztri+™ sensor must be recycled in accordance with Directives 2012/19/EU and 2006/66/EC. To arrange for return or disposal of the Breztri+™ sensor contact the supplier.
Warranty
The Breztri+™ sensor includes a 12 month warranty against manufacturing defects from date of first use. This warranty may be voided under the following circumstances: damage to the Breztri+™ sensor including dropping, water damage resulting from condensation or immersion, tampering, attempts to service, or other forms of abuse.
The Breztri+™ sensor warranty expires 4 years from date of manufacture.
Specifications
| Model Number | NF0103 |
| Actuation Log Precision | 1 second |
| Actuation Log Capacity | 5120 actuations and sensor status records |
| Internal Clock Accuracy | ± 1 hour after 12 months Note: the Breztri+™ sensor clock is updated every time data is uploaded to a compatible mobile app. |
| Compatible with | iOS or Android phones and tablet computers. |
| Wireless Communication | Bluetooth® 4.0: 2.40 – 2.48 GHz, 1.0 mW Low Energy |
| Battery Type | Lithium coin cell, Non-rechargeable |
| Shelf Life | 3 years |
| Battery Life/ Service Life | 1 year |
| Operating Temperature | 0 to 40ºC (32 to 104ºF) |
| Storage Temperature | -20 to 60ºC (-4 to 140ºF) |
| Operating / Storage Humidity | 15 to 90% RH (non- condensing) at water vapour pressure ≤ 50 hPa |
Symbols
 | Manufacturer: Adherium (NZ) Limited |
 | Serial Number |
 | Part Number |
 | Date of Manufacture |
 | Keep dry |
 | Non-rechargeable Sensor |
 | EU only: Do not dispose Breztri+™ sensor as unsorted municipal waste |
 | EU only: Medical Device |
 | EU only: European conformity mark |
 | Temperature limit |
 | Humidity limitation |
 | China RoHS conformity |
Electromagnetic Compatibility
The Breztri+™ sensor does not perform any clinical function where loss or degradation would result in unacceptable risk.
| Emissions / Immunity Test and Standard | Compliance Level |
| Radiated EMI CISPR 11 | Group 1 Class B |
| Electrostatic Discharge IEC 61000-4-2 | ± 8 kV contact, ± 2/4/8/15 kV air |
| Radiated RF EM Fields IEC 61000-4-3 | 10 V/m: 80 – 2700 MHz |
| Proximity fields from RF wireless communications equipment IEC 61000-4-3 | 9 V/m: 710, 745, 780, 5240, 5500, 5785 MHz 27 V/m: 385 MHz 28 V/m: 450, 810, 870, 930, 1720, 1845, 1970, 2450 MHz |
| Power frequency magnetic fields IEC 61000-4-8 | 30 A/m |
FCC Statement
This device complies with Part 15 of the FCC Rules. Operation is subject to the following two conditions:
- This device may not cause harmful interference, and
- This device must accept any interference received, including interference that may cause undesired operation.
“Harmful interference” is defined by FCC as any emission, radiation or induction that endangers the functioning of a radio navigation service or of other safety services or seriously degrades, obstructs or repeatedly interrupts a radio communications service operating in accordance with FCC rules.
The Grantee is not responsible for any changes or modifications not expressly approved by the party responsible for compliance. Such modifications could void the user’s authority to operate the equipment.
Declaration of Conformity
Adherium (NZ) Ltd declares that this Breztri+™ sensor is in compliance with the general safety and performance requirements and other relevant provisions of Regulation EU 2017/745.
Adherium (NZ) Ltd declares that the radio equipment type Breztri+™ sensor is in compliance with Directive 2014/53/EU.
Declarations of Conformity are available at: www.adherium.com/EUDoC.
No part of this document may be reproduced or transmitted in any form or by any means, electronic, mechanical, photocopying, recording, or otherwise, without the prior written permission of Adherium (NZ) Ltd.
Product specifications may change without notice.

Emergo Europe
Prinsessegracht 20
2514 AP The Hague
The Netherlands Breztri+™ sensor is distributed by:
AstraZeneca US
1800 Concord Pike Wilmington, DE 19803, USA
Breztri+™ sensor is manufactured by:
Adherium (NZ) Ltd
Level 11, 16 Kingston Street, Auckland 1010, New Zealand
Contact: [email protected]
www.adherium.com
Breztri+™ Sensor Product Manual
Part Number: ND0515
Version: 1
Issue Date: 12 May 2020
![]() 93/42/EEC, 2014/53/EU, 2011/65/EU
93/42/EEC, 2014/53/EU, 2011/65/EU
© 2020 Adherium (NZ) Ltd. All rights reserved.
For patent and trade mark information, including third-party trademarks, refer to www.adherium/ip.
Breztri Aerosphere is a trade mark of AstraZeneca AB.