![]() 453D4, 453D5, 453D7 Derma Seal
453D4, 453D5, 453D7 Derma Seal
Instructions for use
Product description
INFORMATION
Date of last update: 2021-12-16
- Please read this document carefully before using the product and observe the safety notices.
- Instruct the user in the safe use of the product.
- Please contact the manufacturer if you have questions about the product or in case of problems.
- Report each serious incident related to the product to the manu facturer and to the relevant authority in your country. This is par particularly important when there is a decline in the health state.
- Please keep this document for your records.
1.1 Function
The 453D4, 453D5, and 453D7 Derma Seal residual limb socks are made from a polymer gel-coated fabric and are used for transtibial residual limbs. They are used to protect the skin of the residual limb and reduce stress caused by pressure and chafing. The medical-grade mineral oil in the polymer gel cares for the skin of the residual limb when it is worn. The different Derma Seal residual limb socks are designed as follows:
| Reference Number | Design |
| 453D7 Derma Seal | Nylon fabric with an inner polymer gel coating |
| 453D4 Derma Seal forte | Coolmax® fabric with an inner polymer gel coating |
| 453D5 Derma Seal double forte | 2 Coolmax® fabric layers with a polymer gel coating in-between |
Intended use
2.1 Indications for use
The product is intended exclusively for lower limb endoprosthetic fit tings.
2.2 Area of application
The Derma Seal residual limb socks are applied to fittings of Münster supracondylar beddings and PTB prostheses (Patella-Tendon-Bear ing).
Safety
3.1 Explanation of warning symbols![]() CAUTION Warning regarding possible risks of accident or injury.
CAUTION Warning regarding possible risks of accident or injury.
NOTICE Warning regarding possible technical damage.
3.2 General safety instructions![]() WARNING!
WARNING!
Risk of severe injuries
- Store the product out of reach of children. There is a risk of suf location if the product is pulled over the mouth and nose.
![]() CAUTION!
CAUTION!
Risk of injury and risk of product damage
- Keep the product away from open flames, embers, and other sources of heat.
- The product may only be used by one person; reuse on another person is prohibited.
- Observe the cleaning instructions.
- Make sure the product is applied properly and fits correctly. The improper or excessively tight application can cause pressure points and constriction of blood vessels and nerves.
- Do not continue to use the product if it does not fit properly.
Skin irritation, formation of eczema or infections due to contam nation with germs
- Only use the product on intact skin.
Scope of delivery
| Quantity | Designation | Reference number |
| 1 | Instructions for use | – |
| 1 | Residual limb sock | – |
Use
5.1 Donning and doffing the residual limb sock Donning
Requirement: Remove all sharp objects from the hands and wrists (such as rings, bracelets, or watches). Do not allow sharp fingernails to come into contact with the product.
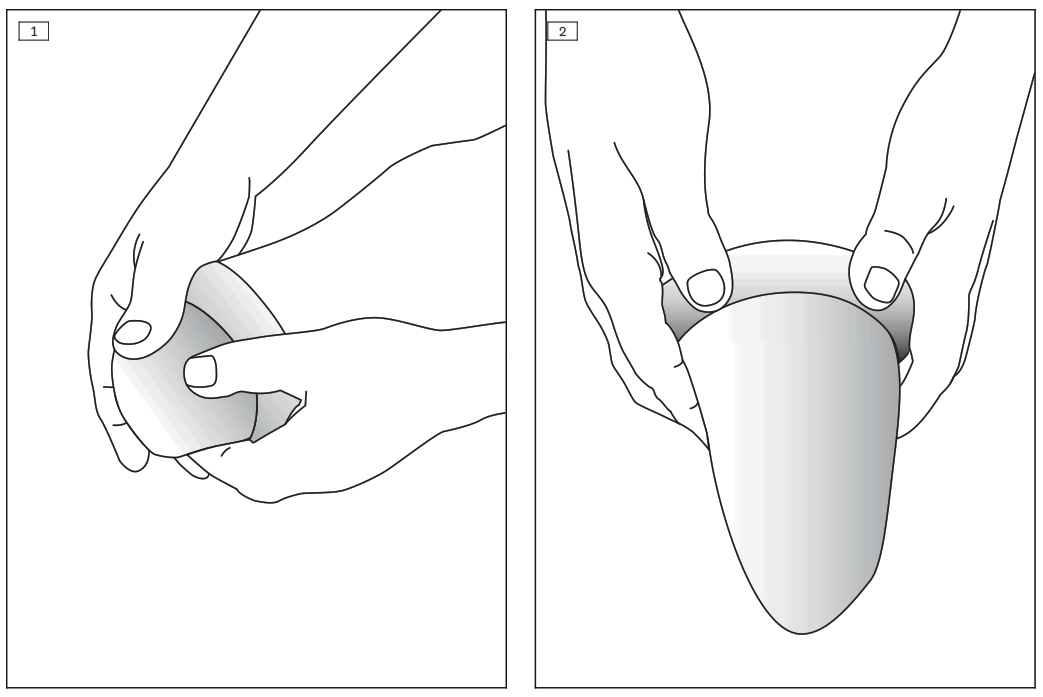
- INFORMATION: Put only dry residual limb socks on. Roll up the residual limb sock, place it on the end of the residual limb, and unroll it onto the residual limb with no wrinkles (see fig. 1).
Doffing
- Roll the residual limb sock up from the open end to the closed end and remove it from the residual limb (see fig. 2).
- Recommendation: clean the residual limb sock according to the section “Cleaning and Care” (see page 7).
5.2 Cleaning and Care
- NOTICE! In order to avoid damage, do not wring out when washing. Hand washes the residual limb sock in warm water (30 °C) with mild detergent.
- Thoroughly rinse the residual limb sock with clear water.
- INFORMATION: Using a hand towel speeds up the drying process. Leave the residual limb sock to dry lying flat on a washing stand or a hand towel.
Disposal
In some jurisdictions, it is not permissible to dispose of the product with unsorted household waste. Improper disposal can be harmful to health and the environment. Observe the information provided by the responsible authorities in your country regarding a return, collection, and disposal procedures.
Legal information
All legal conditions are subject to the respective national laws of the country of use and may vary accordingly.
7.1 Liability
The manufacturer will only assume liability if the product is used in accordance with the descriptions and instructions provided in this document. The manufacturer will not assume liability for damage caused by disregarding the information in this document, particularly due to improper use or unauthorized modification of the product.
7.2 CE conformity
The product meets the requirements of Regulation (EU) 2017/745 on medical devices. The CE declaration of conformity can be down loaded from the manufacturer’s website.
7.3 Warranty
The manufacturer warrants this device from the date of fitting. The the warranty covers defects that can be proven to be a direct result of flaws in the material, production or construction and that are reported to the manufacturer within the warranty period.
Further information on the warranty terms and conditions can be obtained from the competent manufacturer distribution company.
Technical Data
The selection of the sock size depends on the length and circumfer ence of the residual limb. The residual limb length is measured over the condyles. The length of the gel of the residual limb sock should exceed that of the prosthetic socket.
| 453D4 | ||||
| Reference Number | Sock length [cm] | Gel length [cm] | Distal cir cumfer ence [cm] | Proximal circumfer ence [cm] |
| 453D4=1 | 30 | 25 | 15 to 22 | 20 to 32 |
| 453D4=2 | 30 | 25 | 20 to 26 | 28 to 42 |
| 453D4=3 | 40 | 30 | 20 to 26 | 28 to 45 |
| 453D4=10 | 70 | 25 | 15 to 22 | 20 to 32 |
| 453D4=20 | 75 | 30 | 20 to 26 | 28 to 42 |
| 453D5 | ||||
| Reference Number | Sock length [cm] | Gel length [cm] | Distal cir cumference [cm] | Proximal circumfer ence [cm] |
| 453D5=1 | 30 | 25 | 15 to 20 | 20 to 28 |
| 453D5=2 | 30 | 25 | 20 to 24 | 28 to 40 |
| 453D5 | ||||
| Reference Number | Sock length [cm] | Gel length [cm] | Distal cir cumference [cm | Proximal circumfer ence [cm] |
| 453D5=3 | 40 | 30 | 20 to 24 | 28 to 43 |
| 453D5=10 | 70 | 25 | 15 to 20 | 20 to 28 |
| 453D5=20 | 75 | 30 | 20 to 24 | 28 to 40 |
| 453D7 | ||||
| Reference Number | Sock length [cm] | Gel length [cm] | Distal cir cumference [cm | Proximal circumfer ence [cm] |
| 453D7=1 | 30 | 20 | 16 to 22 | 16 to 25 |
| 453D7=2 | 40 | 25 | 18 to 26 | 18 to 30 |
| 453D7=3 | 40 | 25 | 20 to 31 | 20 to 35 |
| 453D7=4 | 45 | 33 | 20 to 31 | 20 to 35 |
| 453D7=5 | 45 | 25 | 23 to 35 | 23 to 40 |
| 453D7=6 | 50 | 33 | 23 to 35 | 23 to 40 |
| 453D7=7 | 50 | 33 | 27 to 40 | 27 to 45 |
| 453D7=8 | 50 | 33 | 30 to 48 | 30 to 53 |
![]() Ottobock SE & Co. KGaA
Ottobock SE & Co. KGaA
Max-Näder-Straße 15 · 37115 Duderstadt · Germany
T +49 5527 848-0 ·
F +49 5527 848-3360
[email protected]
www.ottobock.com