BLOOD PRESSURE MONITOR
BT106
USER MANUAL
v1.1
Components of the Device
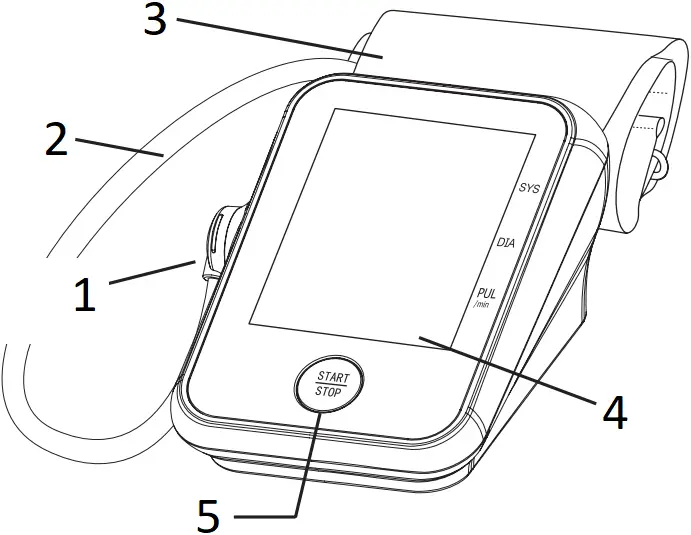
- CUFF TUBE CONNECTOR
- CUFF TUBE
- CUFF
- DISPLAY
- START/STOP BUTTON
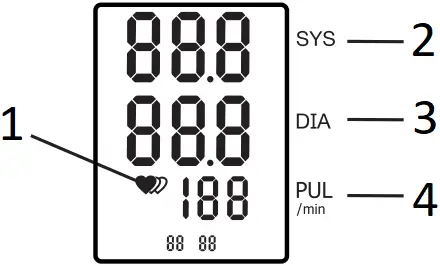
- IRREGULAR HEARTBEAT INDICATOR
- SYSTOLIC MEASUREMENT
- DIASTOLIC MEASUREMENT
- PULSE MEASUREMENT
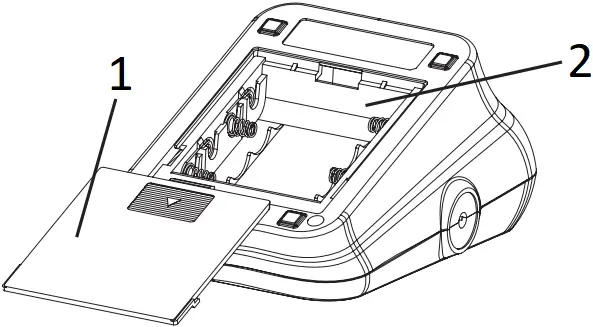
- BATTERY DOOR
- BATTERY COMPARTMENT
Installing the Batteries
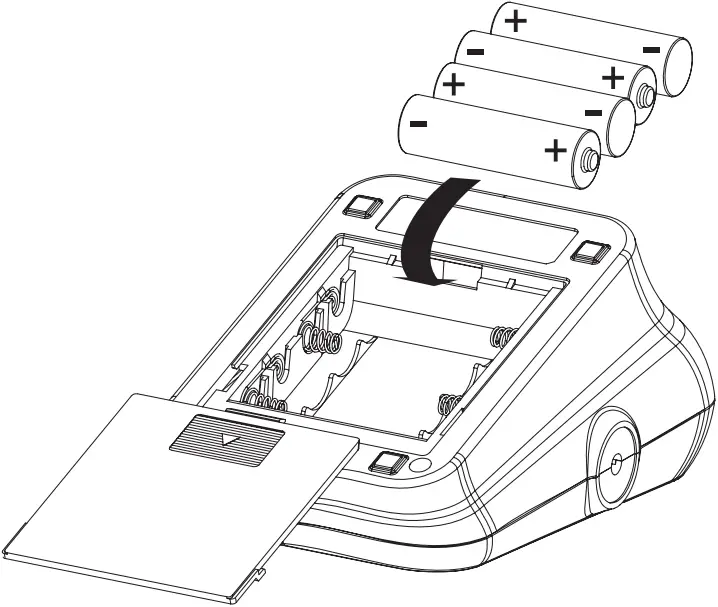
If this is your first time using the device:
1. Slide open the battery door on the back of the device.
2. Install the batteries provided with the device. Follow the diagram inside the battery compartment for correct polarity—the springs should align with the negative sign on the batteries.
3. Slide the battery door closed.
Securing the Cuff
1. Plug the connector on cuff tube into the device.
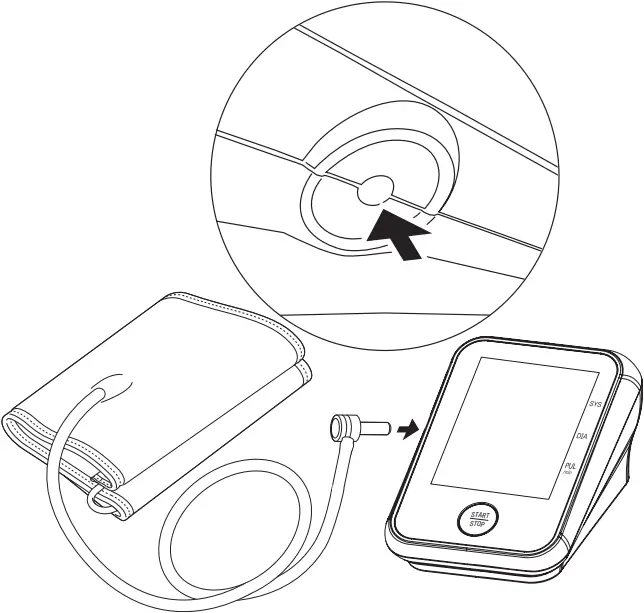
2. Expose your upper arm by removing or adjusting clothing and jewelry. Make sure blood flow is not constricted by a rolled up sleeve.
3. Open the cuff and loosen fully.
4. Orient the cuff so that the tube exits towards the hand.
5. Place your arm through the cuff loop, with your palm facing up.
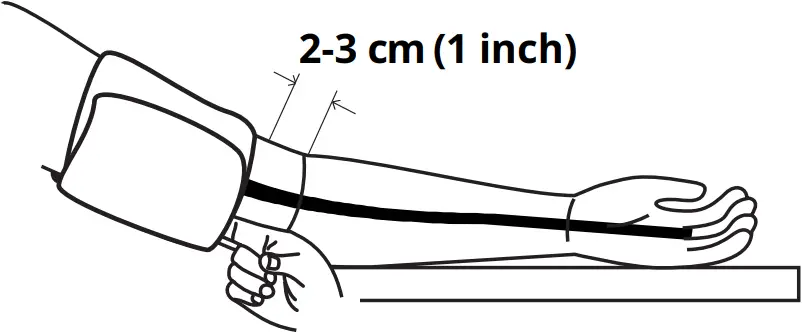
6. Position the cuff’s edge about an inch (2-3 cm) above the elbow.
7. Align the tube with the center of your arm.
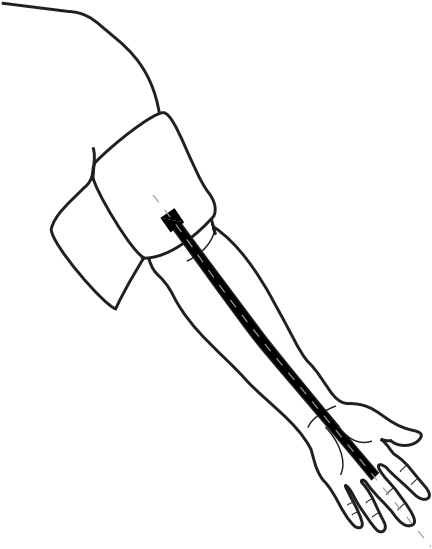
8. Tighten the cuff evenly around your arm by pulling on the end—make sure the tube stays aligned with the center of your arm.
9. Wrap the end of the cuff over your arm to secure it in place. Don’t make it too tight—allow a finger to fit between the cuff and your arm.
Taking a Measurement
10. If possible, relax and rest for at least 5 minutes before taking a measurement.
11. Lay your arm on a table with your palm facing up. The cuff should be at the same height as your heart. Sit up straight and rest your feet flat on the ground. Make sure the tube is not kinked or pinched.
12. Press the START/STOP button on the device to turn it on.
13. Wait for the device to power on and perform the measurement while inflating the cuff.
14. Do not move or talk during the procedure.
15. When the measurement concludes, results are shown on the display and the cuff automatically deflates.
16. Remove the cuff after it deflates.
17. The device automatically transmits measurement results. During this process an indicator is shown in the middle of the display.


Transmission in progress Transmission successful
18. When the measurement has been successfully transmitted, the device shows the indication above before powering off.
Notes
- If you wish to abort the measurement while the cuff is inflating, press and hold the START / STOP button for 3 seconds.
- If the device detects an irregular heartbeat, the display will show this
 indicator along with the measurement results. For more details, please see Blood Pressure, Pulse, and Irregular Heartbeat below.
indicator along with the measurement results. For more details, please see Blood Pressure, Pulse, and Irregular Heartbeat below.
Contraindications
- Consult a medical professional before using this device if you have an implanted cardiac device, such as a pacemaker or defibrillator.
- Consult a medical professional before using this device during pregnancy.
- Do not use this device on infants.
Precautions
- Only use Duracell alkaline AA batteries with this device as these offer the longest battery life. Other brands may not be able to provide sufficient power for cellular data transmission.
- Only use a cuff that has been approved by the manufacturer for this device model.
- This device is for adults. Do not use it on children unless otherwise instructed by a medical professional.
- Keep the device, cuff, and batteries away from children as their components they may pose a risk of choking or strangulation if used improperly.
- Do not apply the cuff on an arm that has:
- an unhealed wound,
- another medical device attached, or
- an intravenous drip or a blood transfusion attached.
- The accuracy of this device has not been specifically validated for use:
- during pregnancy,
- on users with an implanted cardiac device, such as a pacemaker or defibrillator,
- on users with premature ventricular beats, atrial fibrillation, peripheral arterial disease,
- on users undergoing intravascular therapy, or with arteriovenous (AV) shunt.
Consult a medical professional before use.
- Do not use this device if you have pre-eclampsia, or if you have undergone a mastectomy.
- Do not use this device with high-frequency (HF) surgical equipment.
- Do not use this device if you are allergic to polyester, nylon, or plastic.
- This device is intended for indoor, home use. This device is not intended for public use.
- If in doubt, contact your medical professional before use.
- The device may require up to 30 minutes to warm up / cool down from the minimum / maximum storage temperature before it is ready for use.
- Do not use the device where flammable gases are present, or in environments with high oxygen concentration.
- Measurements may not be accurate if the device is used in close proximity to devices generating strong electromagnetic fields, such as microwave ovens, wireless charging stations, X-ray machines, etc.
- Wireless communication devices, such as Wi-Fi access points, mobile phones, and cordless phones may cause interference that may affect the accuracy of measurements. A minimum distance of 1 foot (30 cm) should be kept from such devices during measurement.
- The device is not intended for use on extremities other than the arm, or for any purpose other than measuring blood pressure and pulse rate.
- Do not confuse self-monitoring with self-diagnosis. This device allows you to monitor blood pressure. Do not begin or alter medical treatment without consulting a medical professional.
- Do not take frequent, consecutive measurements as these may constrict blood flow and lead to injury.
- This unit is not suitable for continuous monitoring during medical emergencies or operations.
- This device is not anesthesia proofed (AP/APG), and it is not suitable for use in the presence of flammable anesthetics.
- Do not touch the batteries during measurement.
- This device is portable, but should not be operated in transit.
- You can use this device to take your own measurement, no third-party operator is required.
- In rare occasions, the device may remain inflated after a measurement. If this were to occur, make sure to disconnect the tube from the device to release pressure. Prolonged pressurization of the cuff may lead to ecchymosis or other injury.
- Do not kink, fold, stretch, compress, or otherwise deform the tube during measurement as it may lead to an inaccurate result, or excessive inflation of the cuff which may cause injury.
Troubleshooting
| Problem | Possible Reason | Resolution |
| Device doesn’t power on when START/STOP button is pressed. or Cuff does not inflate. (display remains off) | Batteries completely drained. | Replace batteries with new Duracell AAs. |
| Batteries inserted with incorrect polarity. | Remove batteries and reinstall them making sure the + and − polarity symbols on the batteries match those inside the battery compartment. | |
| Cuff does not inflate. (display turns on) | Cuff not plugged in correctly. | Make sure the connector on cuff tube is plugged all the way into the device. |
| Cuff doesn’t form a loop around arm. | Cuff not assembled. | To form a loop: pass the end of the cuff furthest away from the tubing through the long metal D-ring. The smooth side (without the hook and loop fasteners) should be on the inside of the cuff loop. |
| Readings too high or low. or Readings vary significantly between measurements. | Cuff not positioned correctly. | Review instructions on Securing the Cuff and retry measurement. |
| Movement during measurement. | Place your arm on a table with your palm facing up. The cuff should be at the same level as your heart. Make sure you stay still and don’t talk during measurement. | |
| Measurement results differ from those measured at the doctor’s office. | Measurements taken in a medical / professional environment (white coat effect) may affect your blood pressure. | Measurements taken in a more relaxed, home environment may avoid effects experienced in clinical settings. Continue taking readings as usual. If in doubt, contact a medical professional. |
| Error code shown after measurement. | Measurement accuracy may be affected or transmission may have been unsuccessful. | Consult the error code table below for more information and further instructions. |
Error Codes
| Code | Description | Resolution |
| Er 1 | Battery low | Replace batteries with four Duracell AAs. |
| Er 2 | Movement detected | Avoid moving or talking while taking a measurement. After securing the cuff and assuming the correct posture, consider waiting for five minutes to settle down before starting a measurement. |
| Er 3 | Measurement out of range | Confirm that the cuff is secured correctly on your exposed arm. Remove all clothing and jewelry from your arm. If you feel unwell, contact a medical professional. |
| Er 4 | No cellular coverage | Move to another area, closer to a window. Use the device at a location where you get strong cellular signal with your mobile phone. |
| Er 5 | Cellular connection error | |
| Er 6 | Data transmission error | |
| Er 7 | Internal error | Replace batteries with four Duracell AAs. If the issue persists, contact customer support. |
| Er 9 | Provisioning error | Retake measurement. If the issue persists, contact customer support. |
| Er 10 | Subscription suspended | Contact customer support. |
| Er 11 | Subscription terminated | |
| Er 15–19 | System error | Contact customer support. |
| Er 20 | Cuff inflation error | Make sure the cuff is properly connected to the device. |
| Er 21 | Pulse not detected | Adjust the cuff on the arm, and make sure to assume the correct posture. |
| Er 22 | Measurement error | |
| Er 23–24 | System error | Contact customer support. |
Storage and Maintenance
- Only use Duracell AA batteries with the device.
- Store your device and cuff in a clean and dry place.
- Avoid exposure to direct sunlight, moisture, extreme heat, cold, or humidity.
- Avoid exposure to vibration, shaking, or other mechanical stresses.
- Make sure the rubber tube of the cuff is not squeezed, stretched, or kinked during use and storage.
- Avoid twisting or folding the cuff and tube too tightly as this may cause damage over time.
- Clean both device and cuff with a soft, dry cloth. If necessary use a dampened cloth and natural detergent. Do not use alcohol, benzene, or other harsh chemicals.
- Avoid washing or splashing the device or cuff with water. They are not water resistant.
- Keep the device away from infants and small children.
- Do not try to repair or modify the device at home. Do not attempt to replace components (e.g., tube, connector, etc.) by yourself. If you suspect incorrect operation, contact customer support for further assistance.
- The device must only be serviced and repaired by authorized service personnel.
- At the request of authorized service personnel, circuit diagrams, component part lists, descriptions, and calibration procedures will be made available by the manufacturer or distributor.
- Do not use the device while under maintenance, or being serviced.
- Dispose of the device, its accessories, and batteries according to your local waste disposal regulations and guidelines.
Blood Pressure and Pulse
Blood pressure is represented by two numbers:
1. Systolic pressure: indicates the maximum pressure blood is exerting against the artery walls when the heart beats;
2. Diastolic pressure: indicates the minimum pressure blood is exerting against the artery walls while the heart is resting between beats.
An additional useful metric is the pulse rate, which indicates the number of times the heart beats in a minute.
This device measures blood pressure using the oscillometric method. Before each measurement, the device establishes a zero point based on the atmospheric pressure. The cuff is then inflated, and the device measures the oscillations in pressure created by the user’s heartbeat.
Irregular heartbeat is indicated by the device (![]() ) if there is a 25% deviation from the average pulse rate between three, or a deviation of 15% between five heartbeats.
) if there is a 25% deviation from the average pulse rate between three, or a deviation of 15% between five heartbeats.
While this may not be a reason for immediate concern, contact your medical professional if you see this symbol frequently.
Components of the measurement system are:
- Cuff with tube
- Control electronics & display
- Pump
- Pressure release valve
Symbols
Symbol | Description |
 | Follow instructions for use |
 | Caution: consult instructions for use |
| Cuff circumference | |
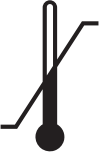 | Temperature range |
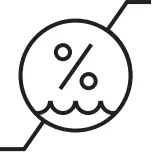 | Relative humidity range |
 | Model number |
 | Serial number |
 | Date of manufacture |
| Ingress protection: against solid objects of 12.5 mm or greater and vertically falling water drops | |
 | Type BF applied part |
 | Complies with FCC regulations |
| Complies with European Community directives Notify body: TÜV SÜD Product Service GmbH | |
 | European Authorised Representative (EC Rep) |
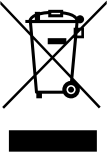 | Do not dispose of this product as unsorted municipal waste, take it for recycling. |
Technical Specification
| Model | BT106 |
| Measurement Method | Oscillometric measurement |
| Measurement Range | Systolic pressure: 60–230 mmHg 8.0–30.7 kPa Diastolic 40–130 mmHg pressure: 5.3–17.3 kPa Pulse: 40–199 beats/min |
| Rated Cuff Pressure | 0–299 mmHg 0.0–39.9 kPa |
| Accuracy | Pressure: ±3 mmHg / 0.4 kPa Pulse: ±5 % |
| Expected Lifetime | 5 years or 10,000 measurements (may vary based on usage conditions) |
| Battery Life | 50–60 measurements (may vary based on usage conditions) |
| Operating Conditions | Temperature: 5–40 °C Relative humidity: 15–90 % (non‑condensing) Pressure: 70–106 kPa |
| Storage Conditions | Temperature: −20–60 °C Relative humidity: 0–93 % (non‑condensing) |
| Cuff Size | 22–42 cm |
| Device Weight | 274 g (excl. batteries) |
| Device Dimensions | 102×143×73 mm |
| Device Ingress Protection | IP21 |
| Applied Part | Type BF |
| Software Version | A01 |
Complied Standards List
| Risk Management | EN ISO 14971:2012 / ISO 14971:2007 Medical devices—Application of risk management to medical devices |
| Labeling | EN ISO 15223-1:2016 / ISO 15223-1:2016 Medical devices. Symbols to be used with medical device labels, labelling and information to be supplied. Part 1: General requirements |
| User Manual | EN 1041:2008+A1:2013 Information supplied by the manufacturer of medical devices |
| General Requirements for Safety | EN 60601-1:2006+A1:2013+A12:2014 / IEC 60601-1:2005+A1:2012 Medical electrical equipment—Part 1: General requirements for basic safety and essential performance EN 60601-1-11:2015/ IEC 60601-1-11:2015 |
| Electromagnetic Compatibility | EN 60601-1-2:2015/ IEC 60601-1-2:2014 Medical electrical equipment—Part 1-2: General requirements for basic safety and essential performance—Collateral standard: Electromagnetic disturbances—Requirements and tests |
| Performance Requirement | EN ISO 81060-1:2012 Non-invasive sphygmomanometers—Part 1: Requirements and test methods for non-automated measurement typeEN 1060-3:1997+A2:2009 Non-invasive sphygmomanometers—Part 3: Supplementary requirements for electro-mechanical blood pressure measuring systemsIEC 80601-2-30:2018 Medical electrical equipment—Part 2-30: Particular requirements for the basic safety and essential performance of automated non-invasive sphygmomanometers |
| Clinical Investigation | EN 1060-4:2004 Non-invasive sphygmomanometers—Part 4: Test procedures to determine the overall system accuracy of automated non-invasive sphygmomanometers ISO 81060-2:2013 Non-invasive sphygmomanometers—Part 2: Clinical validation of automated measurement type |
| Usability | EN 60601-1-6:2010+A1:2015/IEC 60601-1-6:2010+A1:2013 Medical electrical equipment—Part 1-6: General requirements for basic safety and essential performance—Collateral standard: Usability IEC 62366-1:2015 Medical devices—Part 1: Application of usability engineering to medical devices |
| Software Life-cycle Processes | EN 62304:2006/AC: 2008 / IEC 62304: 2006+A1:2015 Medical device software—Software life-cycle processes |
| Biocompatibility | ISO 10993-1:2009 Biological evaluation of medical devices—Part 1: Evaluation and testing within a risk management process ISO 10993-5:2009 Biological evaluation of medical devices—Part 5: Tests for in vitro cytotoxicity ISO 10993-10:2010 Biological evaluation of medical devices—Part 10: Tests for irritation and skin sensitization |
| Unique Device Identification (UDI) | HIBC Supplier Labeling Standard (SLS) 2.4 (contains serial number and date of manufacture) |
Manufacturer’s Declaration
Electromagnetic Emissions
| Emissions Test | Compliance |
| RF emissions CISPR 11 | Group 1 |
| RF emissions CISPR 11 | Class B |
Electromagnetic Immunity
| Immunity Test | IEC 60601-1-2 Compliance Level |
| Electrostatic discharge (ESD) IEC 61000-4-2 | ±8 kV contact ±2 kV, ±4kV, ±8 kV, ±15 kV |
| Power frequency magnetic field IEC 61000-4-8 | 30 A/m 50Hz/60Hz |
| Radiated RF IEC 61000-4-3 | 10 V/m 80 MHz–2.7 GHz 80 % AM at 1 kHz |
Radiated RF Immunity
Test specifications for ENCLOSURE PORT IMMUNITY to RF wireless communications equipment as defined in IEC 61000-4-3.
| Test Freq. (MHz) | Freq. Band (MHz) | Service | Modulation | Power (W) | Distance (m) | Immunity Test Level (V/m) |
| 385 | 380–390 | Tetra 400 | Pulse 18 Hz | 1.8 | 0.3 | 27 |
| 450 | 430–470 | GMRS / FRS 460 | Freq. 5 kHz, dev. 1kHz sine | 2 | 0.3 | 28 |
| 710 | 704–787 | LTE Bands 13, 17 | Pulse 217 Hz | 0.2 | 0.3 | 9 |
| 745 | ||||||
| 780 | ||||||
| 810 | 800–960 | GSM 800/900; TETRA 800; iDEN 820; CDMA 850; LTE Band 5 | Pulse 18 Hz | 2 | 0.3 | 28 |
| 870 | ||||||
| 930 | ||||||
| 1720 | 1700–1990 | GSM 1800; CDMA 1900; GSM 1900; DECT; LTE Bands 1, 3, 4, 25; UMTS | Pulse 217 Hz | 2 | 0.3 | 28 |
| 1845 | ||||||
| 1970 | ||||||
| 2450 | 2400–2570 | Bluetooth; 802.11 b/ g/n; RFID 2450; LTE Band 7 | Pulse 217 Hz | 2 | 0.3 | 28 |
| 5240 | 5100–5800 | 802.11 a/n | Pulse 217 Hz | 0.2 | 0.3 | 9 |
| 5500 | ||||||
| 5785 |
Out-of-Range Pressure Alarm
If systolic blood pressure below 60 mmHg or above 230 mmHg, or diastolic blood pressure below 30 mmHg or above 130 mmHg is detected, the device will indicate a measurement error showing Er 3 on the display.
For further details and instructions on what to do when this indication is shown, refer to the Error Codes section.
Configuration of this alarm condition does not change after replacing the batteries.
| Alarm Priority | Low |
| Indication Delay | ≈10 ms |
| Operator Position | 30–100 cm from display |
FCC Compliance Statement
This device complies with part 15 of the FCC Rules. Operation is subject to the following two conditions:
1. This device may not cause harmful interference, and
2. this device must accept any interference received, including interference that may cause undesired operation.
EU Declaration of Conformity
This device complies with the Radio Equipment Directive 2014/53/EU (RED). The declaration of conformity is available at: https://www.bodytrace.com/product/bt106/red_doc.pdf
Intended Use
This digital blood pressure monitor was designed to measure the blood pressure and heartbeat rate of adults with an arm circumference of 22-42 cm (9-17 inches).
This device is intended for indoor, home use.
Contents Included
- Blood pressure monitor
- BT196: Regular Adult Cuff for 22-42 cm (9-17 inch) arm circumference (applied part)
- 4 x AA batteries
Optional Accessories
- BT195: Extended Adult Cuff for 22-45 cm (9-18 inch) arm circumference
Contact Information
Please contact your program’s customer support. BodyTrace will not be able to directly assist customers.
Manufactured for:
BodyTrace, Inc.
2150 N 1st St, Unit 469
San Jose, CA 95131
USA
Manufactured by:
Guangdong Transtek Medical Electronics Co., Ltd.
Zone A, No. 105, Dongli Road
Torch Development District, 528437 Zhongshan, Guangdong
China
European Authorized Representative: Medical Device Safety Service GmbH
Medical Device Safety Service GmbH
Schiffgraben 41, 30175 Hannover, Germany
BT106-UM-101