27403 Examination Couch Aluminium
Product Information
| Product Name | LETTINO VISITA MEDICA TRADIZIONALE EXAMINATION COUCH – ALUMINIUM |
|---|---|
| Manufacturer | Gima S.p.A. |
| Model Number | 27403 |
| Manufacturer Address | Via Marconi, 1 – 20060 Gessate (MI) Italy |
| [email protected] / [email protected] | |
| Website | www.gimaitaly.com |
| Country of Origin | Italy |
Product Usage Instructions
- Read the user manual carefully before using the device.
- Ensure that the product is used only for its intended
purpose. - Do not use the product in a way that is not specified in the
manual. - Inspect the product for any worn or damaged parts before each
use. - Replace any worn or damaged parts immediately for safety
reasons. - If any component parts deteriorate beyond general maintenance,
contact your distributor for appropriate solutions. - Follow any specific instructions provided in the manual for
disposal of the product.
Technical Data Sheet
| Dimension (L x W x H) cm | 176 x 60 x 78 |
|---|---|
| Capacity Kg | 90 |
| Net weight kg | 22.0 |
| Gross weight kg | 27.0 |
| Volume M3 | 0.21 |
| Number of necks | 1 |
| Packaging (L x W x H) cm | 151 x 19 x 74 |
PROFESSIONAL MEDICAL PRODUCTS
LETTINO VISITA MEDICA TRADIZIONALE EXAMINATION COUCH – ALUMINIUM
MANUALE D’USO ISTRUZIONI E MONTAGGIO USER MANUAL ASSEMBLY INSTRUCTIONS È necessario segnalare qualsiasi incidente grave verificatosi in relazione al dispositivo medico da noi fornito al fabbricante e all’autorità competente dello Stato membro in cui si ha sede All serious accidents concerning the medical device supplied by us must be reported to the manufacturer and competent authority of the member state where your registered office is located
27403
Gima S.p.A. Via Marconi, 1 – 20060 Gessate (MI) Italy [email protected] – [email protected] www.gimaitaly.com Made in Italy
M27403-M-rev.1-01.23
ENGLISH
4
INTRODUCTION For the purposes of Regulation (EU) 2017/745, the product falls under the CLASS I MEDICAL DEVICES category. We advise you to read the following manual carefully before using the device. This model has been designed with the aim of limiting any risks to the user in the event of unintentional impact, by eliminating sharp edges, sharp corners, etc.
SAFETY Use for purposes other than what it is intended for is not allowed. The manufacturer declines all responsibility for damage caused by using the product the wrong or improper way, or in a way unforeseen in the current manual. For safety reasons, replace any worn or damaged parts immediately.
MAINTENANCE / CLEANING The 1-joint examination bed does not require any special maintenance; however, before use, it is necessary to check that all the moving parts are tightened correctly. To clean the bed, a slightly damp cloth can be used; normal detergents can also be used, provided that they do not contain chlorine and its compounds. Attention: · Avoid using hydrogen chloride or bleach.
ASSISTENCE Should any of the component parts deteriorate beyond general maintenance, contact your distributor, they will be available to advise you on the most appropriate solution to every kind of problem.
WARNINGS The information contained in this document are subject to modifications without notice having to be given. Disposal: the product described in this manual must be sent to specific companies for scrapping.
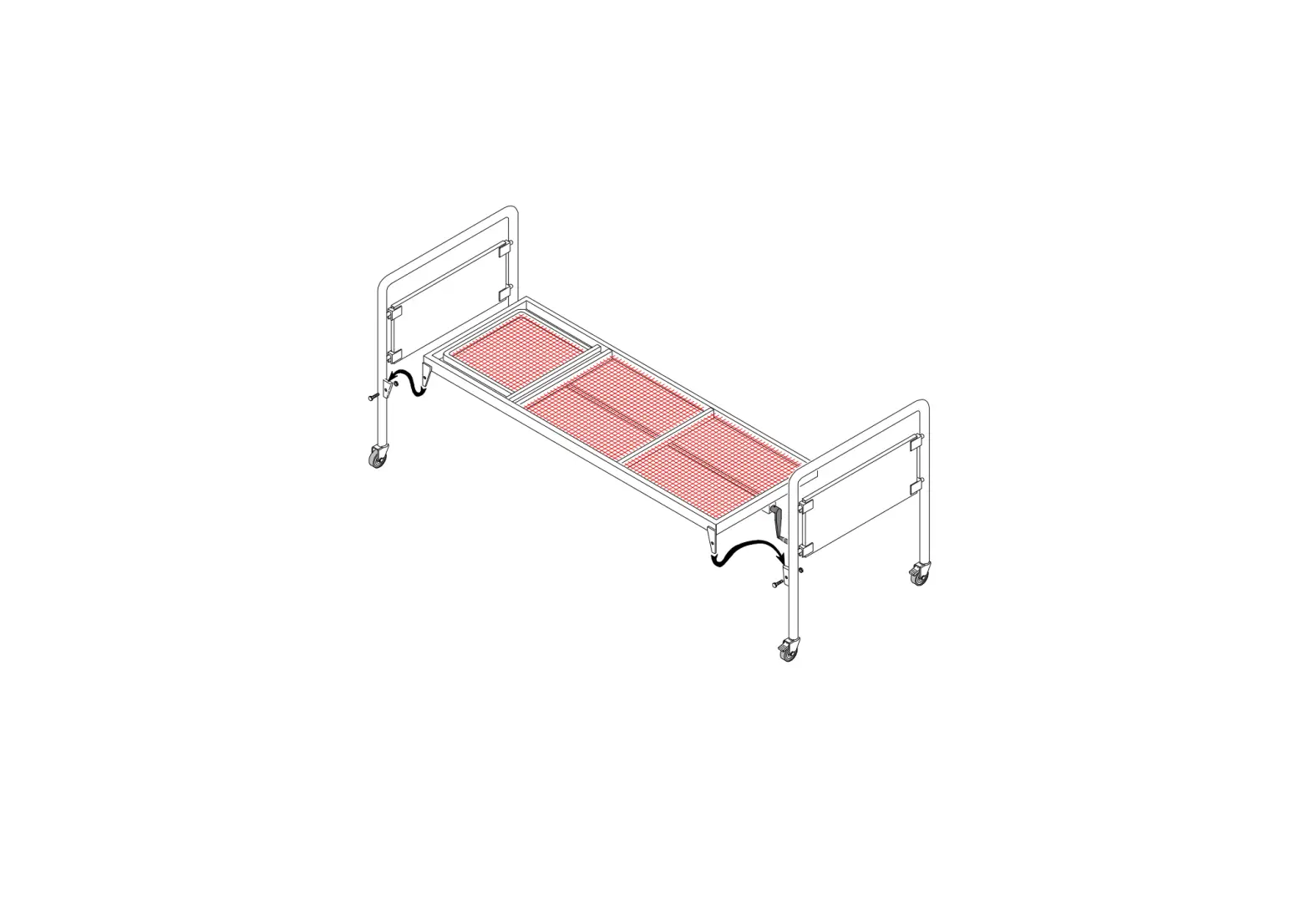
GENERAL INFORMATION Examination bed with a frame made of round polished aluminium tubes Ø 30 x 2 mm thick; fitted with an elevating, manually adjustable headrest with 1 joint and 2 sections, and upholstered in soft, easily washable faux leather. The upholstery consists of: chipboard base with low formaldehyde content, heliocell sponge with a density of 25 kg/sq m, 4 cm thick, convenient and comfortable. Unassembled model supplied as a kit
TECHNICAL DATA SHEET
Dimension (L x P x H) cm
176 x 60 x 78
Capacity Kg
90
Net weight kg
22.0
Gross weight kg
27.0
Volume M3
0.21
Number of necks
1
Packaging (LxDxH) Cm
151 x 19 x 74
5 ASSEMBLING INSTRUCTIONS
ENGLISH
Symbols:
Caution: read instructions (warnings) carefully
Keep in a cool, dry place
Manufacturer
Lot number
Medical Device compliant with Regulation (EU) 2017/745
GIMA WARRANTY TERMS The Gima 12-month standard B2B warranty applies
Keep away from sunlight Date of manufacture Product code Consult instructions for use Medical Device