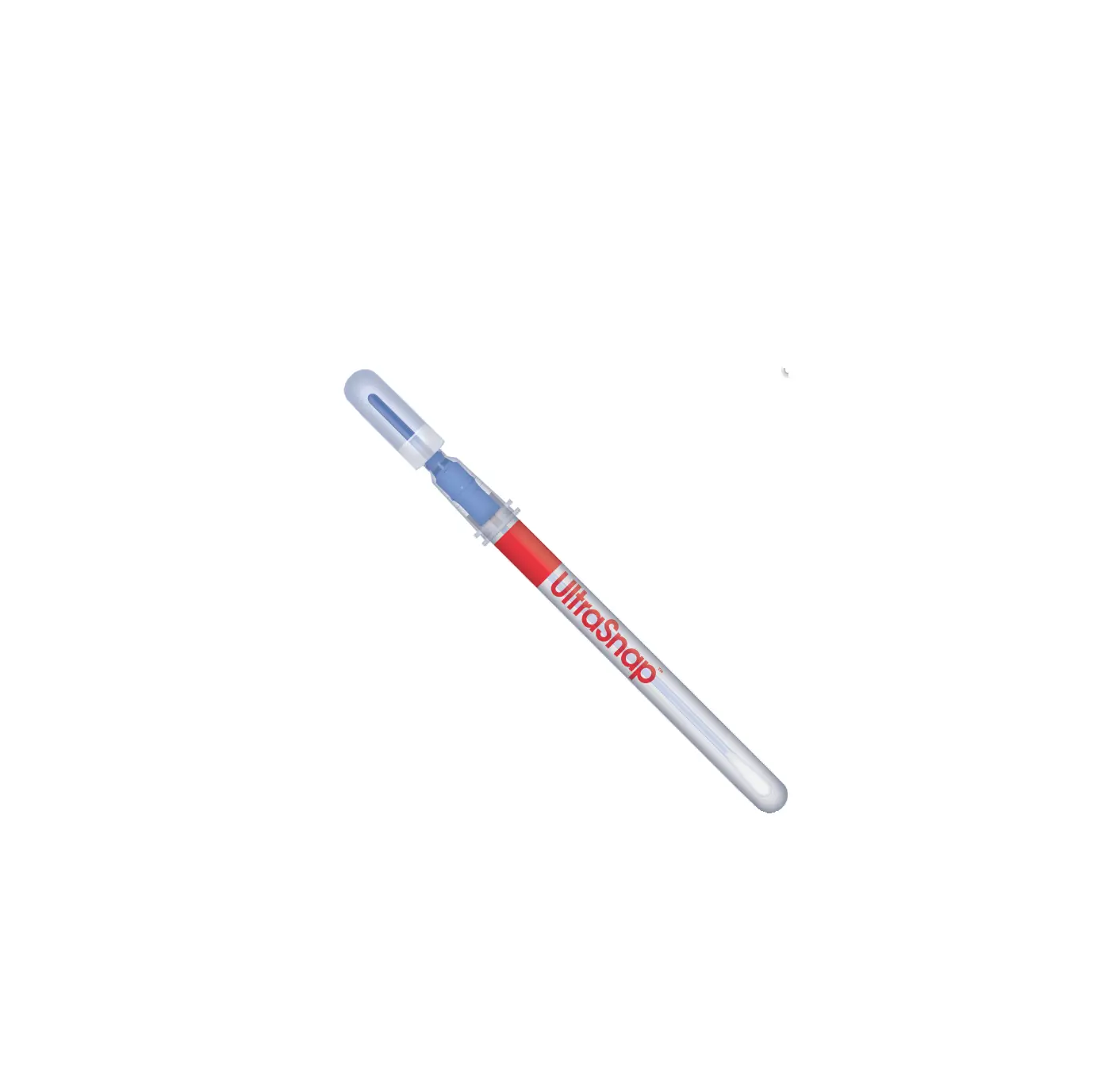
Spectrum DNA SDNA – 1000 2mL Saliva Collection Device User Manual
![]() Do NOT eat, drink, smoke, or chew gum for 30 minutes before giving your saliva sample.
Do NOT eat, drink, smoke, or chew gum for 30 minutes before giving your saliva sample.
Product Overview
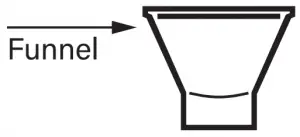
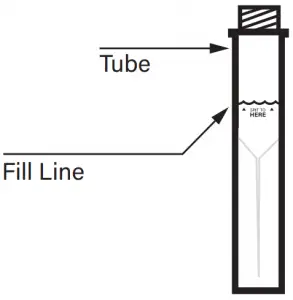
Fill the tube with saliva to the black wavy line. Fill the tube until your saliva (not including bubbles) is at or just above the wavy line. DO NOT OVERFILL.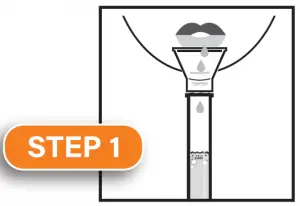
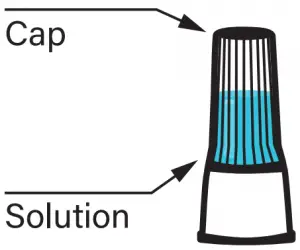
Replace the funnel with the fluid cap. Remove the funnel from the tube. Screw on the enclosed cap TIGHTLY to release the solution that will stabilize the DNA in your saliva.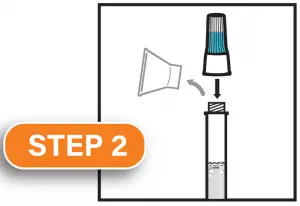
Firmly screw cap down to release solution and seal tube. You will know it works when the blue solution from the cap is released into the tube. Firmly tighten cap to assure the cap and tube is completely sealed.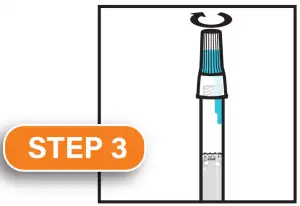
Shake the tube for at least five seconds. This will ensure your sample mixes thoroughly with the stabilizing solution.
- Funnel

- Cap Solution

- Tube Fill Line

Safety Information
- Children under 12 should be supervised.
- Do not clean with any substance containing bleach.
- Do not use if kit is expired.
- Cap is a choking hazard. Keep out of reach of children. Wash with water if liquid solution comes in contact with eyes or skin. DO NOT INGEST LIQUID.
- Avoid contact with skin and eyes. If contact with body occurs, rinse with water. If irritation persists, seek medical advice.
- This kit is designed for the collection of human saliva samples.
- This product has not been FDA cleared or approved, but, has been authorized by FDA under an EUA;
This product has been authorized only for the collection and maintenance of saliva as an aid in detection of nucleic acid from SARS-CoV-2, not for any other viruses or pathogens;
The emergency use of this product is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of medical devices during the COVID-19 outbreak under Section 564(b)(1) of the Federal Food, Drug, and Cosmetic Act, 21 U.S.C. § 360bbb-3(b)(1), unless the declaration is terminated or authorization is revoked sooner.
Label Legend:
![]() Consult instructions for use
Consult instructions for use![]() Use by date (Collect saliva by)
Use by date (Collect saliva by)![]() In vitro diagnostic medical device
In vitro diagnostic medical device![]() Catalog number
Catalog number![]() Caution, consult instructions for use
Caution, consult instructions for use![]() Temperature limit
Temperature limit![]() Authorized representative
Authorized representative![]() Manufacturer
Manufacturer![]() Batch code
Batch code![]() Prescription Only
Prescription Only
| CUSTOMER: Spectrum Solutions | Black |  | |
| CREATED DATE: 3-19-2021 | PROOF #: 1 | PMS 151 |  |
| SIZE: 0 | DESIGNER: LTB | PMS 7621 |  |
| APPROVED BY: | DATE: | PMS 3115 |  |
| FILE: SDNA 1000 2-Sided IFU Insert | PMS 447 |  | |
| DOC# | PS-2019077 | V. 5.3 | |
Contact us
![]() Spectrum Solutions
Spectrum Solutions
12248 S Lone Peak Parkway, #106 Draper, UT 84020
spectrumsolution.com
Storage 15oC ![]() 30oC
30oC![]() 2019162
2019162![]()
![]()
![]() Quality First International OU Laki 30, 12915 Tallinn, Estonia
Quality First International OU Laki 30, 12915 Tallinn, Estonia
Tel:: +372 61041 96