Oral Fluid Sample Collections with Quantisal
Product Information: QuantisalTM Oral Fluid Collection
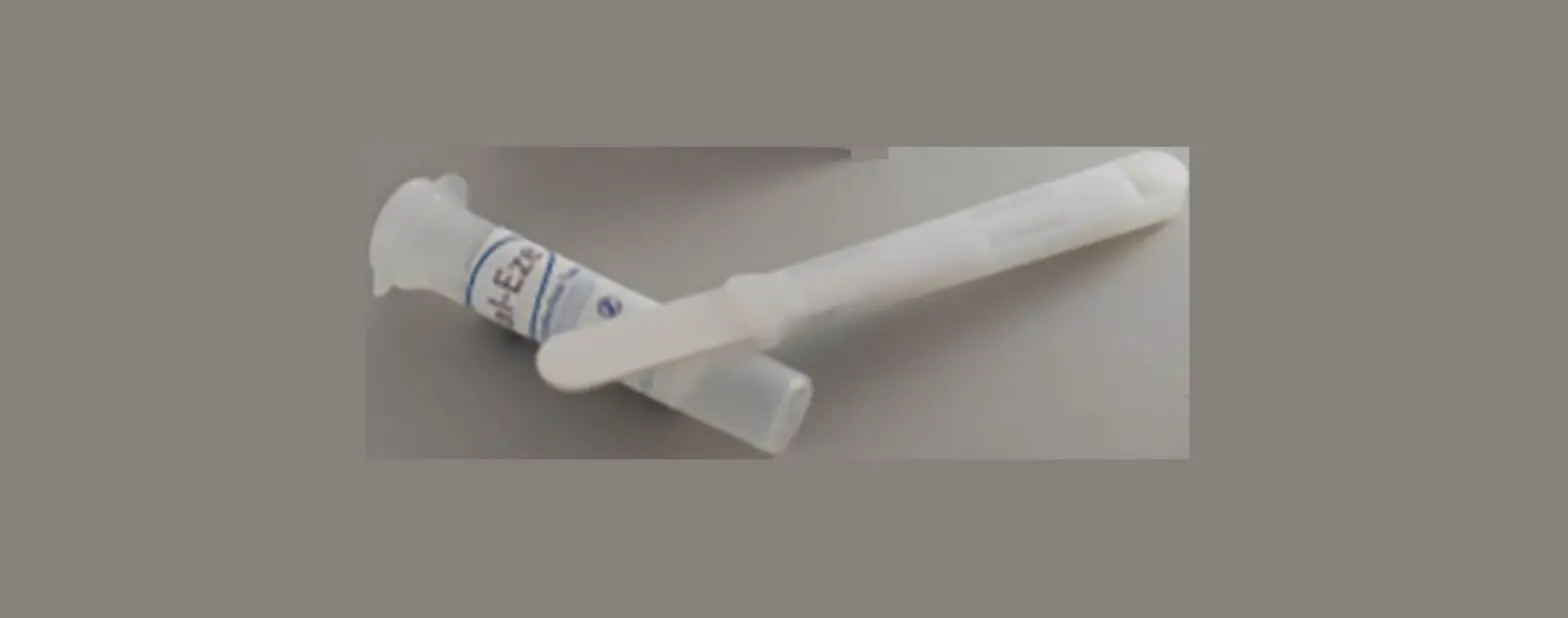
Device
The QuantisalTM Oral Fluid Collection Device is a device
designed for the collection, preservation, and transport of oral
fluid specimens for analytical testing of drugs or drug
metabolites. It is intended for use only under observed collection
and is supplied as part of a test ordered by your healthcare
provider.
These instructions are for provider-observed collections in
instances when in-person collections are not available.
Packing & Shipping Instructions
Follow these steps to properly pack and ship the oral fluid
sample collected using the QuantisalTM device:
- Ensure that the sample collection tube is properly sealed.
- Enter the collection date information onto the provided
requisition form. - Peel the label (detachable lower section of requisition form)
and affix it to the sample collection tube, lengthwise down the
center of the tube. - Place the sample collection tube inside the provided specimen
bag, in the main section of the bag. - Fold the completed requisition form in half and place it inside
the specimen bag, in the smaller, front pouch section of the
bag.
Please ensure that the specimen is collected and shipped on the
same day.
For any questions about the QuantisalTM collection package,
please contact your healthcare provider.
For additional training information, please visit:
www.immunalysis.com/products/oralfluid/quantisal/collector-training/
Instructions for Oral Fluid Sample Collections with QuantisalTM
Patient Self-Collection Instructions for physician-ordered tests*: For observed collection in instances when in-person collections are not available
Self-Collection Prep–Review the testing procedures with your healthcare provider, select a clean and dry place to conduct the provider-observed collection
· Check device package for integrity and appropriate expiration date. Empty your mouth of gum, food, tobacco prior to test. If mouth is not
empty, rinse with water and wait 10 minutes.
· Peel open the QuantisalTM collection device package just far enough for easy removal of the collector handle. Keep the tip of the device pointed down. (Note: Do not use the collection pad if the package has been previously opened, if the collection pad is wet, or if indicator dye
is visible prior to use.)
· Move tongue side to side to accumulate saliva prior to collection.
· Use the handle to remove the device from the packet. (Note: Do not touch the collection pad with fingers before or after specimen collection.)
Step 1
Position the collector under tongue and close mouth. Keep head tilted downward to allow gravity to help with saliva collection. Do not chew on pad, talk, or remove collector from mouth until indicator turns BLUE or until 10 minutes have passed.
Step 3
When the collection is completed, remove the oral fluid collection transport tube from the package. Hold transport tube in an upright position and uncap by pushing up with thumb(s). Do not stand tube on table. Do not spill or empty the liquid from the tube.
Step 5
Once the collector is inside the transport tube replace the cap. SNAP cap firmly in place.
Snap
Step 2
Start a 10-minute timer. The collection process is complete when the blue color appears in the indicator of the handle (typically 2-10 minutes), or when collection time reaches 10 minutes, whichever occurs first.
Step 4
Remove the Quantisal oral fluid collector from your mouth and guide the collector into the uncapped transport tube and replace cap. (Note: Do not place collector back in mouth after it has been placed in the oral fluid collection transport tube.)
Step 6
Place the specimen vial into the specimen bag and complete the procedures to prepare and ship the specimen to the testing laboratory.
For questions about the QuantisalTM collection package, please contact your healthcare provider.
For additional training information, please visit: www.immunalysis.com/products/oralfluid/quantisal/collector-training/
*Intended Use: The QuantisalTM collection device is being supplied as part of a test ordered by your healthcare provider. This QuantisalTM Oral Fluid Collection Device is intended
for the collection, preservation, and transport of oral fluid specimens for analytical testing of drugs or drug metabolites. This device is for use only under observed collection.
These instructions are for provider-observed collections in instances when in-person collections are not available.
1 Confidential–For Internal Use Only
–Please see reverse side for packing/shipping instructions–
Instructions for Oral Fluid Sample Collections with QuantisalTM
Packing & Shipping Instructions–collect specimen and ship on the same day
Step A Ensure sample collection tube is properly sealed.
Figure A Snap
Step D
Place the sample collection tube inside the provided specimen bag, in the main section of the bag.
Figure D
Tube here
Step B Enter collection date information onto the
provided requisition form.
Figure B
Collection date
Step C
Peel label (detachable lower section of requisition form) and affix to sample collection tube, lengthwise down the center of collection tube.
Step E
Fold completed requisition form in half and place inside the specimen bag, in the smaller, front pouch section of the bag.
Figure E
Fold here
Form here
Step F
Firmly close the adhesive on the specimen bag and insert into the provided shipping box. .
Step G
Close shipping box with provided seal and affix
provided “Return” shipping label to sealed shipping box (over existing label).
Figure G
Step H
Affix return shipping label (over existing)
Place labeled shipping box into a convenient FedEx drop box or drop off at a convenient FedEx collection site.
Figure H
Figure C
Peel label (lower section of form)
Figure F
Insert specimen bag
For questions about the QuantisalTM collection package, please contact your healthcare provider.
For additional training information, please visit: www.immunalysis.com/products/oralfluid/quantisal/collector-training/
QuantisalTM is a trademark of Immunalysis Corporation or their respective owners
QuestDiagnostics.com
Quest, Quest Diagnostics, any associated logos, and all associated Quest Diagnostics registered, or unregistered trademarks are the property of Quest Diagnostics. 2AllCthoinrdf-idpaerntytiaml–arkFso–r In®tearnndaTMl U–searOe nthley property of their respective owners. © 2022 Quest Diagnostics Incorporated. All rights reserved. PP9403 11/2022
–Please see reverse side for self-collection instructions–