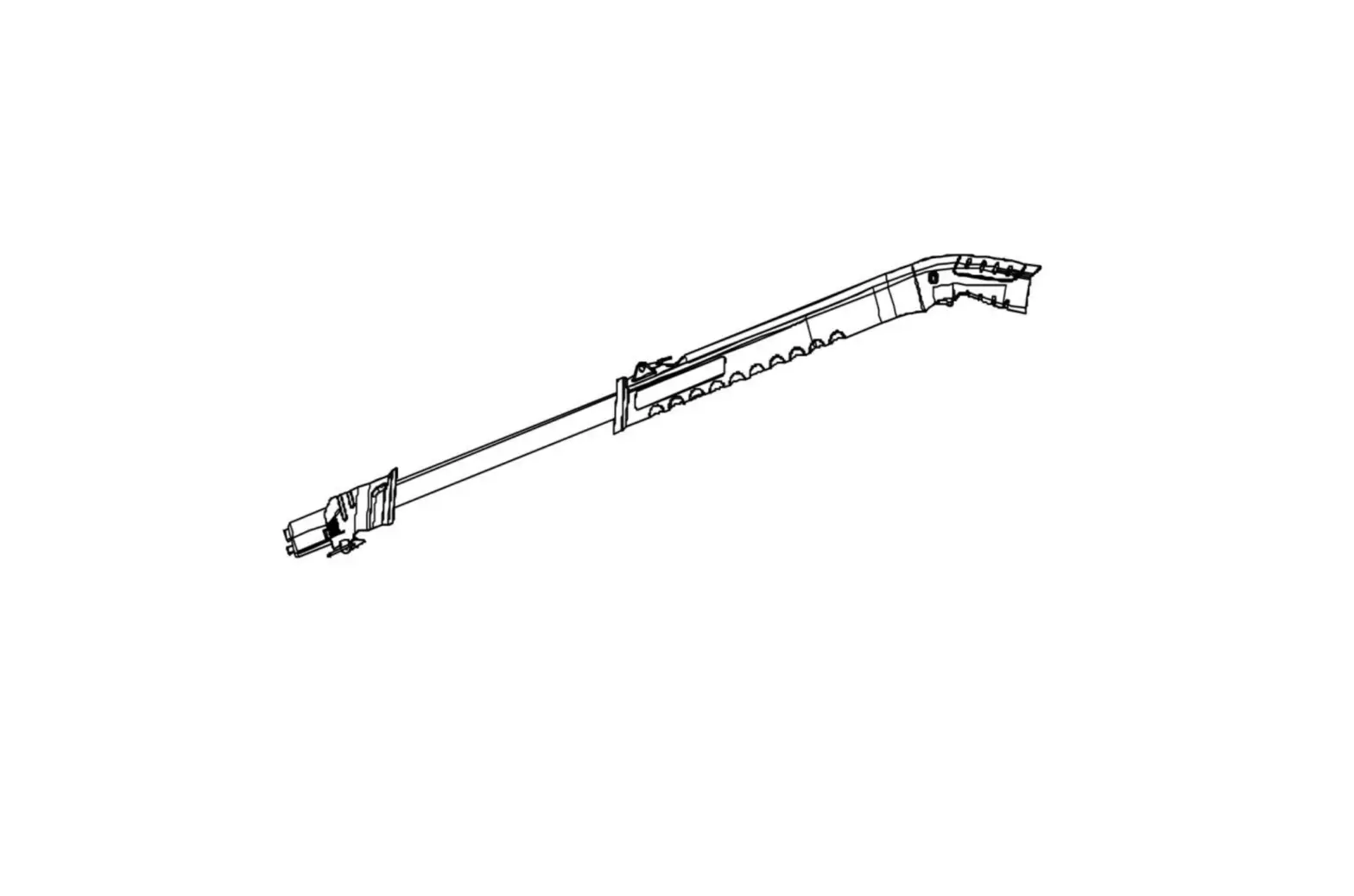
LW10207 Lift Pole
Product Information:
The Asta Di Sollevamento Per Degenti – Tubo Tondo, also known as
Lift Pole – Round Tubing, is a medical device manufactured by
Longway Medical Accessories. It is intended to compensate for a
disability or inability of the bedridden user. The support device
comes with a ground base and is adaptable to any bed. The structure
is made of tubular steel painted that can be positioned according
to the patient’s needs and has a triangle with a strap to adjust
its height. The product has a weight capacity of 75kgs and is
classified as Class I under the European Regulation 745/2017.
Product Usage Instructions:
Before Use:
- Read the instructions (warnings) carefully.
- Ensure proper environmental protection.
- Refer to the maintenance, cleaning, and disinfection
information.
Assembling:
Note: The illustrations for the assembly are indicative to make
the assembly easier. There may be differences from reality. The
support with LW10207 base is delivered disassembled and requires
the following assembly operations:
- Use 2*M10*115mm screws to fix the straight tube between the
left and right leg tubes. - Insert the lifting pole in the space provided and use a plastic
screw to fix it. - Insert the triangle with an adjustable strap into the space
provided.
Symbol Legend:
- Manufacturer
- Date of manufacture
- Product code
- Lifting pole
- Plastic screw
- The triangle
- Lot number
- Authorized representative in the European community
- Straight tube
- Consult instructions for use
- Medical Device compliant with Regulation (EU) 2017/745
- M10*115 mm screw
- Left tube
- Right tube
- Medical Device Unique device identifier
- WEEE disposal
- Imported by
Warranty:
The Gima 12-month standard B2B warranty applies. Disposal: The
product must not be disposed of along with other domestic waste.
The users must dispose of this equipment by bringing it to a
specific recycling point for electric and electronic equipment.
PROFESSIONAL MEDICAL PRODUCTS
ASTA DI SOLLEVAMENTO PER DEGENTI – TUBO TONDO
LIFT POLE – ROUND TUBING
POTEAU DE LEVAGE – TUBE ROND
SOPORTE DE ELEVACIÓN – TUBO REDONDO
PENDURAL – TUBAGEM REDONDA
BETTGALGEN – RUNDSTANGEN
TIJ TELESCOPIC – EAV ROTUND LYFTPOL – RUNDRÖRSSTRUKTUR WYSIGNIK – Z OKRGLYMI RURKAMI –
È necessario segnalare qualsiasi incidente grave verificatosi in relazione al dispositivo medico da noi fornito al fabbricante e all’autorità competente dello Stato membro in cui si ha sede. All serious accidents concerning the medical device supplied by us must be reported to the manufacturer and competent authority of the member state where your registered office is located. Il est nécessaire de signaler tout accident grave survenu et lié au dispositif médical que nous avons livré au fabricant et à l’autorité compétente de l’état membre où on a le siège social. Es necesario informar al fabricante y a la autoridad competente del Estado miembro en el que se encuentra la sede sobre cualquier incidente grave que haya ocurrido en relación con el producto sanitario que le hemos suministrado. É necessário notificar ao fabricante e às autoridades competentes do Estado-membro onde ele está sediado qualquer acidente grave verificado em relação ao dispositivo médico fornecido por nós. Jeder schwere Unfall im Zusammenhang mit dem von uns gelieferten medizinischen Gerät muss unbedingt dem Hersteller und der zuständigen Behörde des Mitgliedsstaats, in dem das Gerät verwendet wird, gemeldet werden. Orice accident grav produs, privitor la dispozitivul medical fabricat de firma noastr, trebuie semnalat productorului i autoritii competente în statul membru pe teritoriul cruia îi are sediul utilizatorul. Det är nödvändigt att meddela tillverkaren och de behöriga myndigheterna i den berörda medlemsstaten, om alla allvarliga olyckor som inträffat i samband med den medicintekniska utrustning som levererats av oss. Naley poinformowa producenta i kompetentne wladze danego Kraju czlonkowskiego o kadym powanym wypadku zwizanym z wyrobem medycznym naszej produkcji. .
LW10207 (GIMA 27664)
Mingguang Longway Technology Co., Ltd. No. 59 Lingji Rd., Industrial Park, Mingguang, Anhui, China Made in China
Lotus NL B.V. Koningin Julianaplein 10, 1 e Verd, 2595AA, The Hague, Netherlands
Gima S.p.A. Via Marconi, 1 – 20060 Gessate (MI) Italy [email protected] – [email protected] www.gimaitaly.com
M27644-M-Rev.0-04.23
ENGLISH
2
Weight Capacity: 75kgs
INTENDED USE:
Longway Medical Accessories are Medical Devices intended to compensate for a disability or inability of the bedridden user. Support device with ground base, adaptable to any bed. Structure in tubular steel painted that can be positioned according to the patient’s needs and triangle with strap to adjust its height.
CAUTION (!): · In case of damage or malfunction of your product DOT NOT use the product and please contact the authorized and trained representative. · Do not use the product for a purpose not indicated in this manual · LONGWAY Medical declines all responsibilities for any consequences resulting from an incorrect use of this product and from unauthorized alteration to the frame of the product. · LONGWAY Medical reserves the right to change the information contained in this document without previous notice · There is a ± 3%. Deviation for the Dimensions of the Products.
GENERAL SAFETY WARNINGS: · If you don’t read this user manual, it is preferable not to use this product or another available part. If you don’t understand the usage or the precautions, please contact with the dealer or the proper technical person before you use the
product because may be caused damage. · Keep away the product from heat sources. · Save this document for future reference. · Be careful when children are nearby and do not allow the
children to play with the product. · Do not exceed the maximum payload. · Special care is always required where there are moving
parts that can cause limb entrapment and injury. · Do not attempt to lift the product by any removable parts. · Do not use the product seat for a different purpose from
the intended one. · The lifting poles are not intended to carry the full weight of an individual.
DECLARATION OF CONFORMITY:
We are solely responsible for declaring that the Medical Devices mentioned in this statement are of Low-Risk Class (Class I) and comply with the requirements of the European Regulation 745/2017 and where appropriate, the standards and legislation referred to.
BEFORE USE:
· Check if the frame is damaged in order to guarantee a safe use of the product. (There are no cracks or fractures in the frame). · In case of damage do not use the product and contact your dealer for further instructions. · Check, if the product is properly assembled and all screws are secure and well screwed on. · Always check the condition of wear for the mechanical parts to ensure the absolute safe use of the product for people and objects
ENVIRONMENTAL PROTECTION:
If one day you find that your product needs to be replaced or it is no longer working for you, consider protecting the environment: 1) Do not dispose your product along with the rest of the public waste (this is also the meaning of the shown recycling sign). 2) Contact your Public Authorities and they will instruct you of the Recycling centers to which your product must be disposed. 3) Correct disposal of your product helps the protection of the environment as well as the recycling of the product’s components.
MAINTENANCE, CLEANING & DISINFECTION
INFORMATION:
To guarantee a safe use and adequate standard of hygiene, the user should perform these procedures before every use. The user has to make certain of the structural integrity of the device and its components. For cleaning and sanitation procedures, follow the steps below: 1. Gloves should be worn. 2. Remove the evident dirt before to proceed with the most accurate disinfection. 3. Use water or mild detergent for the cleaning procedure. 4. Wipe with a dry and clean cloth. 5. DO NOT leave the product wet. 6. Do not use chemical cleaners to clean the frame. This could cause damage to the surface of the product 7. If you need to disinfect the product, use a common, mild disinfectant 8. Protect the item from scratches, cuts and punctures. 9. Lubrication: the product has designed for a minimal maintenance; however, check every six months the lubrication on the moving & mechanical parts, this would ensure an optimal duration.
ASSEMBLING:
NOTE: The illustrations for the assembly are indicative to make the assembly easier. ay will be differences from reality. The support with LW10207 base is delivered disassembled and requires the following assembly operations: 1. Use 2*M10*115mm screws to fix the straight tube between the left and right leg tubes
3
ENGLISH
2. nsert the lifting pole in the space provided, and use plastic screw to fixed; 3. Insert the triangle with adjustable strap into the space provided.
SYMBOL LEGEND
Manufacturer
Date of manufacture
Product code
Lifting pole Plastic screw
The triangle
Lot number Authorized representative in the European community
Caution: read instructions (warnings) carefully
Straight tube
Consult instructions for use
Medical Device compliant with Regulation (EU) 2017/745
M10*115 mm screw Left tube
Right tube
Medical Device Unique device identifier WEEE disposal Imported by
WARRANTY:
The product is guaranteed for a period of one (1) year from the purchasing date. The warranty apply on factory defects & DOES NOT apply on damages resulting from misuse, poor maintenance, modification, overuse, or non-compliance with the product’s operating instructions. Also DOES NOT apply on the parts, that can wear out during use or over time. Damages or defects caused by natural disasters, unauthorized maintenance or repair, power supply problems (where applicable) & carriers are not covered by the warranty. No service or spare part is applied unless it has been inspected by an authorized service department or the manufacturer of the product. Shipping costs for goods of warranty are to be paid by the end user- customer or by dealer.
GIMA WARRANTY TERMS The Gima 12-month standard B2B warranty applies.
Disposal: The product must not be disposed of along with other domestic waste. The users must dispose of this equipment by bringing it to a specific recycling point for electric and electronic equipment.