Standard laryngoscope handles
Standard laryngoscope handles
Directions for use
Intended use
The standard laryngoscope handle is an accessory used with a compatible rigid standard laryngoscope blade which is used to examine and visualize a patient’s airway and aid placement of a tracheal tube.
About this document
These directions for use apply to Welch Allyn reusable standard laryngoscope handles REF:
60200, 60300, 60400, 60305. Welch Allyn reusable standard laryngoscope handles may be used with Welch Allyn standard laryngoscope blades MacIntosh REF: 6904X, English
MacIntosh REF: 6924X, and Miller REF: 6804X and REF 68470.
Warnings and cautions![]() WARNING: Welch Allyn’s reusable standard laryngoscope handles must be reprocessed after each use.
WARNING: Welch Allyn’s reusable standard laryngoscope handles must be reprocessed after each use.![]() WARNING: The reprocessing procedure and the equipment and materials described must be followed and conducted by persons trained and familiar with medical device
WARNING: The reprocessing procedure and the equipment and materials described must be followed and conducted by persons trained and familiar with medical device
reprocessing.![]() WARNING: Consult cleaning and disinfecting agent manufacturer instructions for their proper preparation and use.
WARNING: Consult cleaning and disinfecting agent manufacturer instructions for their proper preparation and use.![]() WARNING: Repeated reprocessing may degrade elements of the handle. Follow inspection procedures to assure damage has not occurred to the handle.
WARNING: Repeated reprocessing may degrade elements of the handle. Follow inspection procedures to assure damage has not occurred to the handle.![]() WARNING: High-level disinfection and/or sterilization are not achieved by these methods.
WARNING: High-level disinfection and/or sterilization are not achieved by these methods.![]() WARNING: Discard any component that shows evidence of damage or deterioration.
WARNING: Discard any component that shows evidence of damage or deterioration.![]() WARNING: Do not modify this equipment. Any modification of this equipment may lead to patient injury. Any modification of this equipment voids the product warranty.
WARNING: Do not modify this equipment. Any modification of this equipment may lead to patient injury. Any modification of this equipment voids the product warranty.![]() WARNING: Personnel shall follow their facility policies and procedures and wear appropriate personal protective equipment when handling potentially contaminated equipment.
WARNING: Personnel shall follow their facility policies and procedures and wear appropriate personal protective equipment when handling potentially contaminated equipment.![]() WARNING: Laryngoscope equipment is not suitable for use in intense magnetic fields
WARNING: Laryngoscope equipment is not suitable for use in intense magnetic fields![]() CAUTION: Failure to follow these instructions may cause damage to this handle.
CAUTION: Failure to follow these instructions may cause damage to this handle.![]() CAUTION: Do not immerse/soak handle, damage to handle may occur.
CAUTION: Do not immerse/soak handle, damage to handle may occur.![]() CAUTION: If the device will be unused for several months or longer, remove the batteries prior to storing the device.
CAUTION: If the device will be unused for several months or longer, remove the batteries prior to storing the device.
Reprocessing instructions
These reprocessing instructions refer to procedures for cleaning and intermediate-level disinfection. Standard laryngoscope handles must be reprocessed prior to first use and between each use using the following method as outlined in this document:
- Cleaning and intermediate-level disinfection
Welch Allyn has validated the above instruction as being capable of preparing these laryngoscope handles for re-use. The user must ensure that the reprocessing as actually
performed by the user’s personnel, with the user’s equipment and materials, achieves the desired result. This may require validation and routine monitoring of the user’s actual process
NOTE: The main handle and bottom cap components of handles marked “AUTOCLAVE” are compatible with the autoclave methods identified which are provided for facilities who wish to autoclave after cleaning and intermediate-level disinfection.
Cleaning and intermediate-level disinfection instructions
| Point of use: |
|
| Preparation for decontamination: |
|
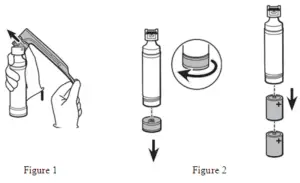
| Cleaning and intermediate-level disinfection: |
|
| Drying: | Allow components to air dry. |
| Maintenance, Inspection, and Testing |
|
| Storage: | Store handle per facility practice to allow the device to remain clean, dry, and ready for service. |
End of reprocessing instructions for intermediate-level disinfection.
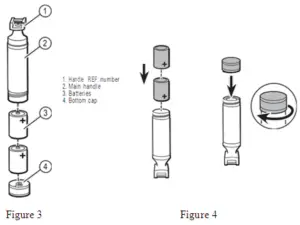
Autoclave instructions
NOTE: The main handle and bottom cap components of handles marked “AUTOCLAVE” are
compatible with the autoclave methods identified which are provided for facilities who wish to
autoclave after cleaning and intermediate-level disinfection.
| Disassembly: | Remove batteries per figure 2 and set aside. |
| After battery removal, select ONE of the following autoclave methods below for the main handle and bottom cap (only): | |
Gravity autoclave: Follow equipment manufacturer and facility procedures in the set-up and operation of autoclave equipment. Gravity autoclave settings are as follows:
| |
Pre-vacuum autoclave: Follow equipment manufacturer and facility procedures in the set-up and operation of autoclave equipment. Pre-vacuum autoclave settings are as follows:
| |
| Maintenance, Inspection, and Testing |
|
| Storage: Store | |
Maintenance instructions
Replace the batteries
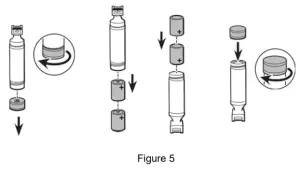
- Unscrew bottom cap of the handle per figure 5 and remove batteries.
- Alkaline batteries are supplied with your handle for maximum performance and are recommended as replacements; however carbon-zinc batteries may also be used.
• Large handle, REF 60200 uses two “D” size
• Medium handle, REF 60300 uses two “C” size
• Penlight handle, REF 60400 uses two “AA” size
• Stubby handle, REF 60305 uses two “AA” size - Insert batteries and reinstall bottom cap per figure 5.
- Reprocess repaired assembly as appropriate per these instructions
Specifications
Electrical:
For information about electromagnetic compatibility (EMC) see Welch Allyn website: http://www.welchallyn.com
Operating: | Storage/Transport: |
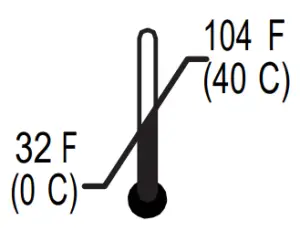 | 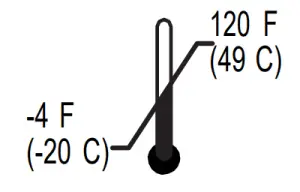 |
Approvals:
Conforms to ASTM F 965 and ISO-7376-1, IEC/EN 60601-1, IEC/EN 60601-1-2![]() The CE mark on this product indicates that it has been tested to and conforms with the provisions noted within the 93/42/EEC Medical Device Directive.
The CE mark on this product indicates that it has been tested to and conforms with the provisions noted within the 93/42/EEC Medical Device Directive.![]() Complies with EMC Framework of Australia
Complies with EMC Framework of Australia
Warranty:
One year
Service Information:
For Technical Support or to obtain information about any Welch Allyn product, contact
Welch Allyn Technical Support: www.welchallyn.com/support.
 ©2014 Welch Allyn, Inc.
©2014 Welch Allyn, Inc.
MM 721357 Ver. C![]() Welch Allyn, Inc.
Welch Allyn, Inc.
4341 State Street Road
Skaneateles Falls, NY 13153-0220 the USA
www.welchallyn.com![]() Authorized European Representative Address:
Authorized European Representative Address:
Regulatory Affairs Representative
Welch Allyn, Limited
Navan Business Park
Dublin Road
Navan, County Meath, Republic of Ireland