DSMAREF DTS Series Tourniquet Cuff User Manual
Intended use
- Tourniquet cuffs intended to occlude the blood flow to obtain hemostasis field during limb surgery.
Cuff descriptio
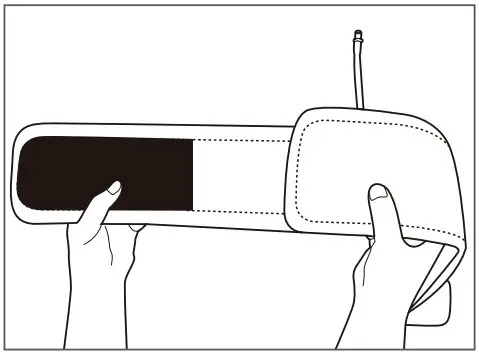
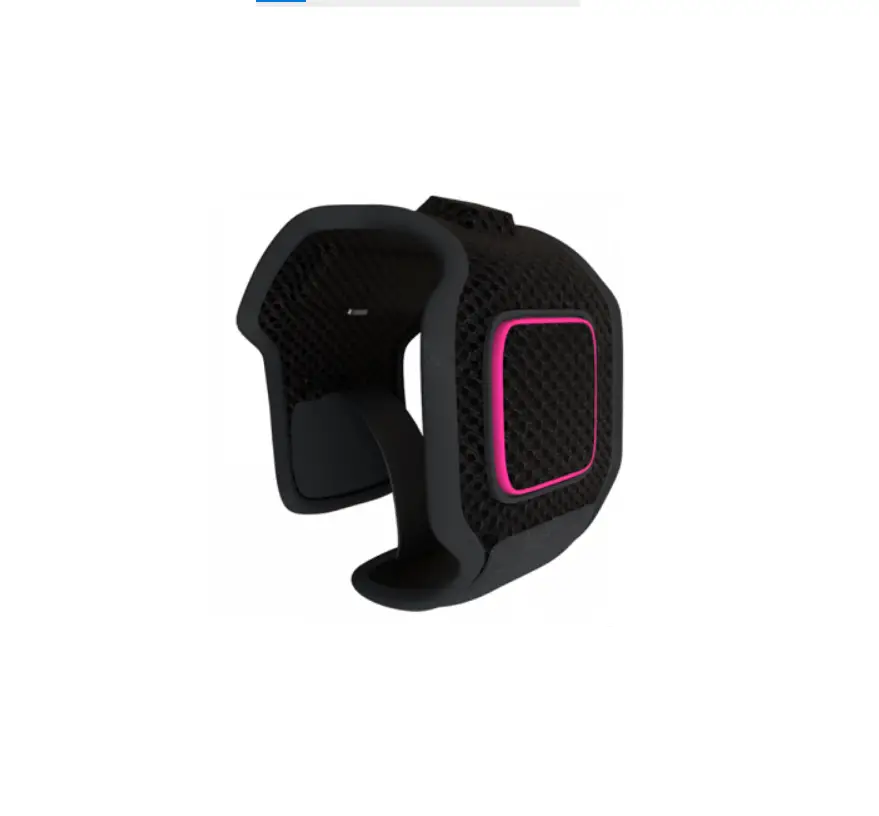
Cuff appearance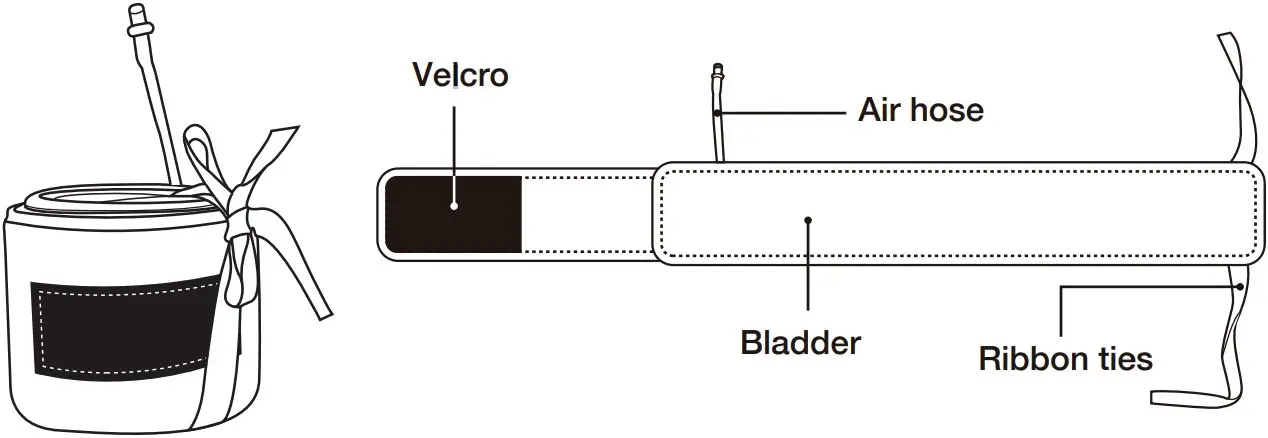
The shape could be different depending on products.
Features of the cuff
- As the elastic bladder guarantees the transmission of pressure to deeper tissue, the bladder can inflated easily by lower pressure. (Please refer to general pressure used normally in hospitals normal max pressure value is 300mmHg for adult’s arm, 400mmHg for adult’s leg, 200mmHg for child’s arm and 300mmHg for a child’s leg but the pressure setting value can be adjusted according to a patient’s age, sex, weight, size… etc..)
- All the cuffs have been tested under 800mmHg under high quality control.
How to use cuff
In accordance with national or regional surgical procedures, sterile drapes are applied to DTS cuff and all parts (except the surgical site).
Cautions before wearing the cuff
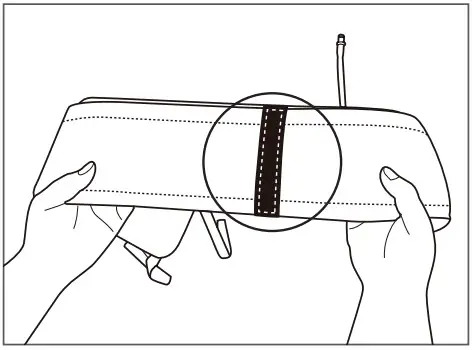
- Check if the Velcro part is inserted properly into the fixing belt.
- Check if the Velcro is attached to the bladder part.

- Check a Velcro to be attached to inner part of a cuff when a cuff is spread out.

How to wear cuff
- The selection of the cuff should be considered each patient’s conditions and various assessment like contraindications, complications, infection and any damage with limitation.
- Double cuff can be recommended if operation time is more than 1 hour, it should be considered using the double cuff to minimize any damage like blood vessel, skin and nerve.
- Cone cuff should be selected if difference between the distal and proximal cuff edges of the patient’s wear area is big.

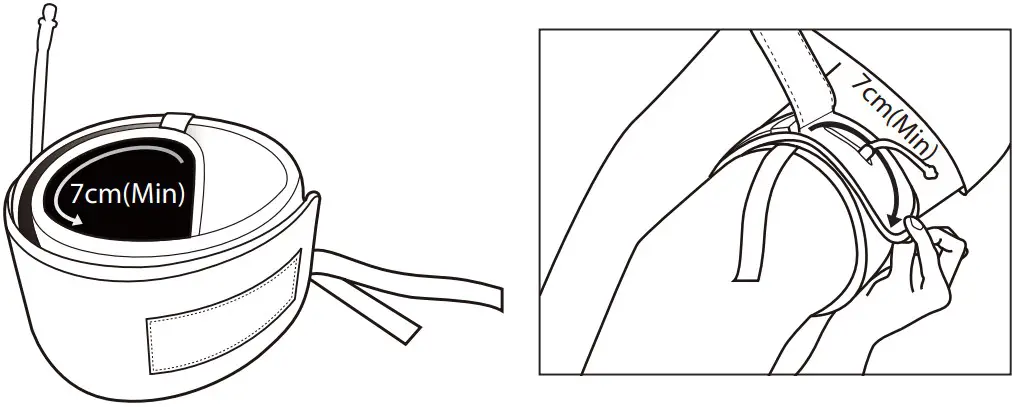
- The cuff should be overlapped at least 7cm (3inches) when a cuff is wrapped on the part requiring homeostasis.
- The width of the cuff should be individualized and wider than half of the limb’s diameter.
- The cuff should be wrapped on the most muscular and wide body part proximally(near the heart) as possible to minimize the damage of blood vessels, nerves and skin.
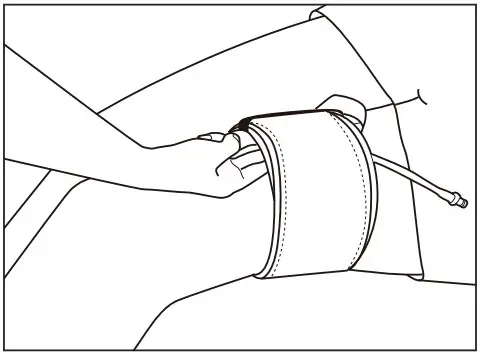
- Position the cuff hose(tube) upwards when a cuff is wrapped on a limb.
Attach the cuff part with a ribbon on the center of the extremity as the starting point as the starting point, and then wind it tightly as far as just two fingers can be input inside of the cuff.
* If there is the gap between the skin and the cuff, pinch or leakage of blood may occur.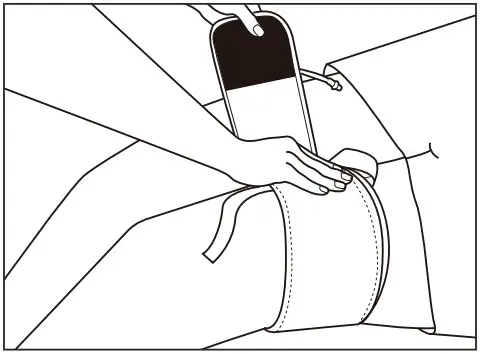
Cautions after wearing calf
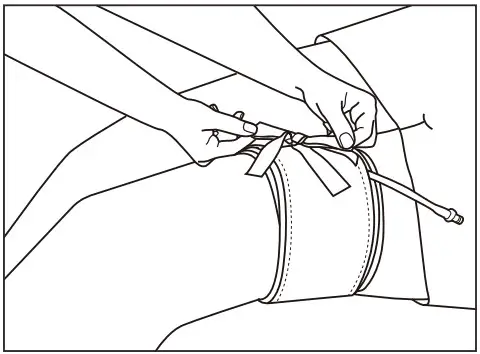
- Cuff should be aligned with ribbon ties not to be twisted or bounced out during inflation.

- After putting the cuff, make sure that you can insert two fingers between the skin and the bladder.

How to connect the device and cuff
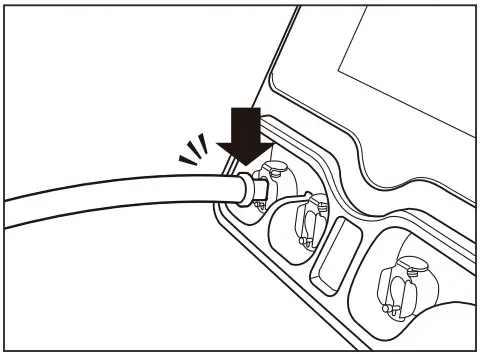
- Connect the hose jacks to the socket of device and the jack of cuff hose until click is sounded.

- Check whether the indicated colors are equal for the air socket of the device, the cuff hose and the air hose.

DISPOSAL
- Used or damaged devices must be disposed of in accordance with the relevant national and international legal regulations.
How to reprocess cuff
※ Please refer to the user manual for more information on reprocessing.
Cuff
| Preparation | Remove all accessories (hose/cuff/IOP sensor). |
| Cleaning | Use a cloth/sponge or brush to wipe off foreign substances on the cuff hose and gently shake off dust from the Velcro part. Wipe until contaminants are no longer visible to the naked eye. |
| Inspection | 1. Roll up the cuff and tie the ribbon, then connect it to the DTS-3000 and the hose.2. Enter the maintenance mode of DTS-3000.3. Enter the Cuff test mode and run the test 3 times.4. During the test, there should be no flaring of the velcro, and check if the Pass is output on the display.* Connect the cleaned Hose and proceed with the inspection. The test is conducted 3 times, andPASS must be issued at least 2 times. For detailed test method, refer to the description of the Cuff test in the user’s manual. |
| Package | Store in a clean cloth or plastic bag. |
| Storage | 1. Store in the hospital storage room according to our instructions.2. Store the cuff according to hospital storage procedures, referring to the environmental conditions for storage in the user’s manual. |
| Transfer | Avoid contact with contaminants when the product is moved other than where it was cleaned. |
Silicone bladder cuff
| Preparation | Remove all accessories (hose/cuff/IOP sensor). |
| Cleaning | After plugging the hose plug of the silicone bladder cuff, Use a brush or cloth/sponge to shake off foreign substances on the tube and cuff.Wipe until no foreign matter can be seen with the naked eye. |
| Inspection | 1. Roll the cuff and tie the ribbon, then connect it to the hose and main device.2. Enter the maintenance mode of DTS-3000.3. Enter the Cuff test mode and run the test 3 times.4. During the test, there should be no flaring of the velcro, and check if the Pass is output on the display.* Connect the cleaned Hose and proceed with the inspection. The test is conducted 3 times, and PASS must appear at least 2 times. For detailed test method, refer to the description of the Cuff test in the main body user’s manual. |
| Package | Pack the Silicone bladder cuff by sterilization-wraps approved as a medical device with the two layers. |
| Sterilization | Put the packaged CUFF into the Auto Clave and sterilize it under the following conditions. Temperature: 134 degreesAtmospheric pressure: 2 atm Sterilization time: 5 minutes Drying time: 15 minutesAfter sterilization according to hospital policy, check whether sterilization is done properly with MI / CI, BI, etc. |
| Storage | 1. Store in the hospital storage room according to our instructions.2. Store the cuff according to hospital storage procedures, referring to the environmental conditions for storage in the user’s manual. |
| Transfer | Avoid contact with contaminants when the product is moved other than where it was cleaned. |
Cuff specifications
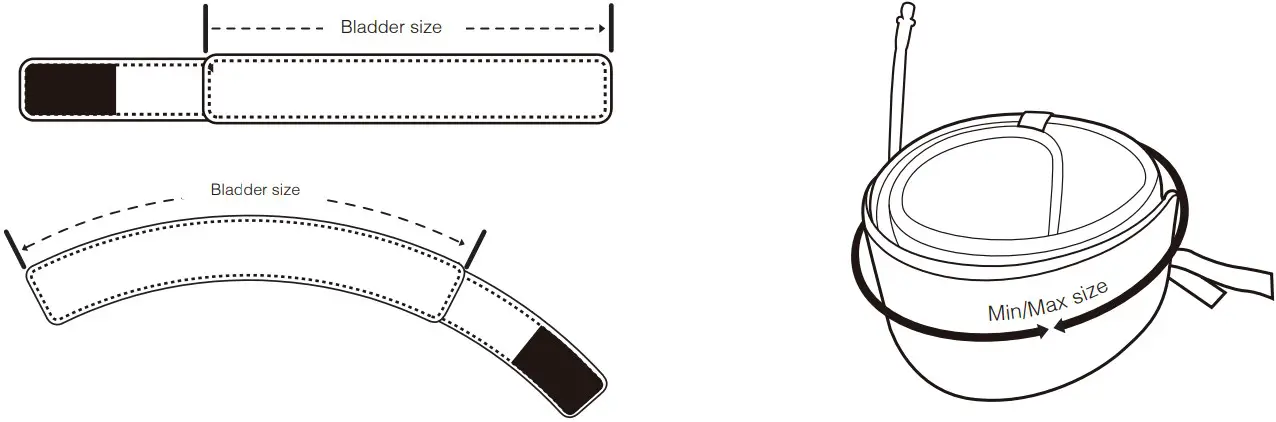
Cuff
| Ref.No | Bladder size | Minimum recommended size | Maximum recommended size | Cleaning | Sterilization possible | Shape |
| DTC-S02 | 25 cm < | < 33 cm | O | X | SINGLE | |
| DTC-S04 | 30 cm < | < 45 cm | O | X | ||
| DTC-S05 | 39 cm < | < 54 cm | O | X | ||
| DTC-S06 | 53 cm < | < 73 cm | O | X | ||
| DTC-S07 | 59 cm < | < 79 cm | O | X | ||
| DTC-S08 | 80 cm < | < 100 cm | O | X | ||
| DTC-D04 | 35 cm < | < 50 cm | O | X | DOUBLE | |
| DTC-D05 | 53 cm < | < 73 cm | O | X | ||
| DTC-D06 | 80 cm < | < 100 cm | O | X | ||
| DTC-D07 | 35 cm < | < 50 cm | O | X | ||
| DTC-C25 | 48cm < | < 63 cm | O | X | CONE SINGLE | |
| DTC-C26 | 63cm < | < 83cm | O | X | ||
| DTC-C27 | 80cm < | < 100cm | O | X | ||
| DTC-CD25 | 48cm < | < 63cm | O | X | CONE DOUBLE | |
| DTC-CD26 | 63cm < | < 83cm | O | X | ||
| DTC-CD27 | 80cm < | < 100cm | O | X |
Silicone bladder cuff
| Ref.No | Bladder size | Minimum recommended size | Maximum recommended size | Cleaning | Sterilization possible | Shape |
| DTC-SA01 | 10 cm < | < 23 cm | O | O | SINGLE | |
| DTC-SA02 | 23 cm < | < 39 cm | O | O | ||
| DTC-SA05 | 39 cm < | < 54 cm | O | O | ||
| DTC-SA06 | 54 cm < | < 69 cm | O | O | ||
| DTC-SA07 | 64cm < | < 79 cm | O | O | ||
| DTC-SA15 | 30cm < | < 45cm | O | O |
※The dimensions of above table can be occurred deviation of 4~5cm according to production condition.
Number of reuses : Cuff : 100 times, Silicone bladder cuff : 40 times
298-24, Gongdan-ro, Gunpo-si, Gyeonggi-do, Korea
Tel : +82-31-459-7211
Fax : +82-31-459-7215
E-mail : [email protected]
KTR Europe GmbH
Mergenthalerallee 77, Eschborn, Hessen, 65760, Germany
Tel : +49(0) 6196-887170
Fax : +49(0) 6196-887-1728