KORU MEDICAL SYSTEMS FreedomEdge Syringe Infudion System
Introduction
The FreedomEdge® Syringe Infusion System is portable and easy to use, requiring no batteries or electricity. It starts to operate when the syringe driver is closed. KORU Precision Flow Rate Tubing™ sets are used to control the flow rate.
The FreedomEdge operates at a constant pressure, which automatically decreases the flow rate if there is an increase in resistance during the delivery. The system will find balance, known as Dynamic Equilibrium (or DynEQ®), between the increasing resistance and flow rate. It provides constant flow which tends to inhibit clots, and holds full pressure after an infusion is complete to prevent blood or drug return. The FreedomEdge also eliminates concerns of a bolus, overflow, overdose or runaway infusion.
Indications for Use
The Freedom Edge Syringe Infusion System consists of the following components:
- Freedom Edge Syringe Driver
- Precision Flow Rate Tubing™
- HIgH-Flo Subcutaneous Safety Needle Sets™
The Freedom Edge Syringe Infusion System is indicated for use with the BD® 20 ml syringe (US Reference number: 302830), BD 30 ml syringe (US Reference number: 302832) and Hizentra® 20 ml prefilled syringe (NDC 44206-458-96).
For Immunoglobulin Administration:
The Freedom Integrated Syringe Infusion System is specifically indicated for the subcutaneous infusion of the following human plasma-derived immunoglobulins when used according to the FDA approved biologic labeling: Cuvitru®, Immune Globulin Infusion (Human) 20% (manufactured by Takeda®); Gammagard Liquid®, Immune Globulin Infusion (Human) 10% (manufactured by Takeda®); Hizentra®, Immune Globulin Subcutaneous (Human) 20% Liquid (manufactured by CSL Behring®) and Hizentra®, Immune Globulin Subcutaneous (Human) 20% Liquid (manufactured by CSL Behring®) Single-use pre-filled syringe for subcutaneous administration in the home, hospital, or ambulatory settings when administered according to the approved biologic or drug product labeling.
For EMPAVELI® (pegcetacoplan) Administration:
The Freedom Integrated Syringe Infusion System is specifically indicated for the subcutaneous infusion with EMPAVELI™ (pegcetacoplan) in the home, hospital, or ambulatory setting when administered according to the approved biologic or drug product labeling.
For Intravenous Antibiotic Administration:
The Freedom Integrated Syringe Infusion System with the Freedom Edge Syringe Driver and Precision Flow Rate Tubing™, is specifically indicated for the intravenous infusion of the following antibiotics when used according to the FDA approved drug product labeling: ertapenem, meropenem, oxacillin, and tobramycin.
MRI Safety Information
![]() The Freedom Integrated Syringe Infusion System is MR Unsafe. Do not use the Freedom Infusion Systems or its components, such as the Precision Flow Rate Tubing™, HlgH-Flo Subcutaneous Safety Needle Sets™ or the Low Residual Volume Y-Connector, while undergoing MRI medical diagnostic procedures.
The Freedom Integrated Syringe Infusion System is MR Unsafe. Do not use the Freedom Infusion Systems or its components, such as the Precision Flow Rate Tubing™, HlgH-Flo Subcutaneous Safety Needle Sets™ or the Low Residual Volume Y-Connector, while undergoing MRI medical diagnostic procedures.
![]() Caution
Caution
- Use the FreedomEdge® Syringe Infusion System only for the patient for whom the device is prescribed and only for its intended use.
- Use only Freedom System accessories manufactured by KORU Medical Systems. Use of off-brand products may result in unknown flow rates.
- Patient tolerability may vary. For patients experiencing discomfort, contact your healthcare provider to determine if a flow rate adjustment is necessary.
- Flow rates can be affected by multiple factors such as temperature, patient conditions, height differences between the system and infusion site, and variations in solution viscosity.
- Excessive motion during infusion may cause flow rate variability. Vigorous activity is not recommended.
- It is recommended to perform infusions while stationary or walking. Infusions with movement other than walking may result in faster, slower, or more variable flow rates than specified. Testing has been performed to simulate walking and its effect on flow rates, no other physical activity has been analyzed.
- Directly connecting extension tubing or HIgH-Flo needle sets (without the luer disc) to the syringe may cause internal damage to the syringe driver.
- Use only BD® brand 20/30 ml syringes, or Hizentra® 20 ml prefilled syringes with the FreedomEdge.
- Using the same tubing set, BD 30 ml syringes will have slightly slower flow rates and slightly increased delivery times versus BD 20 ml syringes.
- Before use, carefully inspect the tubing and needle set packaging. Do not use the set if the package is opened or damaged. Inspect tubing and needle sets for damage. If damaged, replace and contact your healthcare provider.
- Do not re-sterilize tubing or needle sets.
- The slide clamp included on the Precision tubing and HIgH-Flo needle sets should only be used in the case of an emergency, to stop flow immediately. Use of the slide clamp may cause damage to the tubing and can affect the intended flow rate.
- Carefully inspect the FreedomEdge before use. Discontinue use of a syringe driver that has been damaged, exposed to severe impact, or which fails to operate properly.
- Do not place fingers inside while opening or closing the FreedomEdge.
- Do not attempt to interfere with the syringe pusher at any time.
- Do not attempt to open the rear housing.
- Avoid placing needles over a mole, tattoo, scar, muscle, hardened or bruised areas, where proper needle insertion could be difficult.
- To obtain maximum accuracy of the pump, verify that the height of the syringe driver relative to the needle site is positioned within ±3’’ both while in a stationary position and while in motion.
- The FreedomEdge should be used at the same height as the user. If positioned higher than the infusion sites, the pressure will increase and can affect the flow rate. If positioned lower, the pressure will decrease and can affect the flow rate.
- Do not attempt to remove the syringe or disconnect the tubing set without first fully opening the top cover of the FreedomEdge.
- The FreedomEdge Syringe Infusion System does not have an alarm, therefore, no alarm will sound if an interruption to flow occurs. There is no display of infusion status.
- The syringe driver is not suitable for use with medication where delay or under-infusion could result in serious injury.
- If the syringe driver is submerged in any fluid, discontinue use and call your healthcare provider for a replacement.
- Do not autoclave the FreedomEdge.
- The Freedom Edge Syringe Infusion System is not intended for blood transfusions.
- The Freedom Edge Syringe Infusion System is not to be used during diagnostic procedures, such as MRI, x-ray, or CT scans.
- Federal law (USA) restricts this device to sale by or on the order of a physician.
Freedom Edge® Diagram
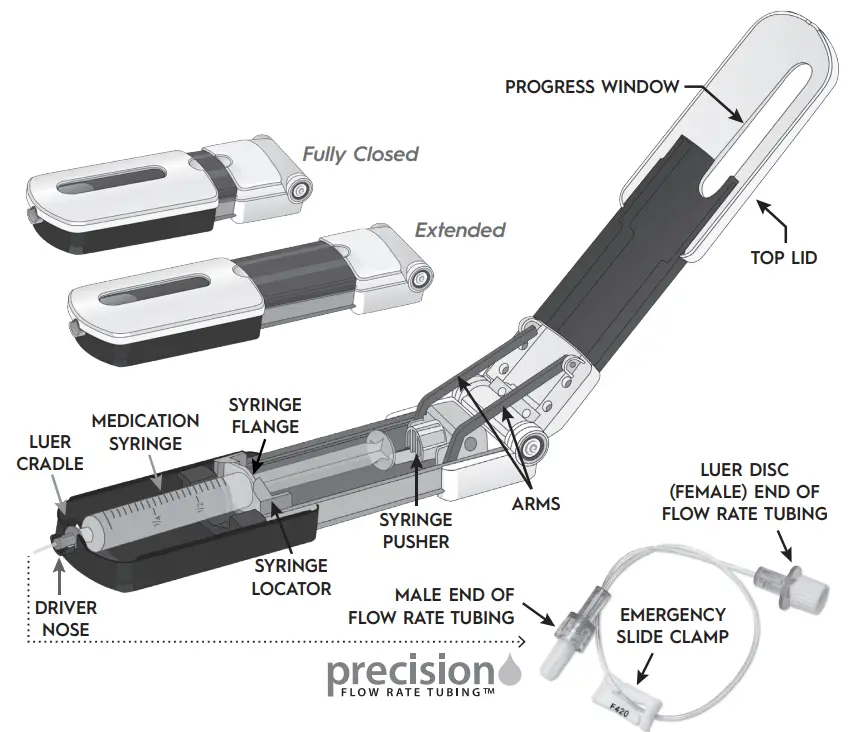
Product Line
Each Freedom Edge includes a travel pouch and user manual.
| Product | Part # |
| Freedom Edge Syringe Driver | F10020 |
| Replacement Travel Pouch | 347400 |
Syringes for use with the FreedomEdge
- Becton Dickinson & Co. BD® Luer-Lok® 20 ml (US Ref. Number: 302830)
- Becton Dickinson & Co. BD® Luer-Lok® 30 ml (US Ref. Number: 302832)
- Hizentra® 20 ml Single-Use Prefilled Syringe (NDC 44206-458-96)
Reoperation Instructions
The Freedom Edge Syringe Driver should be tested prior to any administration.
- Examine the inside to ensure it is free of debris or damage.
- Make sure the orange syringe locator moves freely by sliding it up and down the track with your finger.

Step-by-Step Instructions for Subcutaneous Administration
Before subcutaneous self-administration, patients and/or caregivers should be properly trained by a qualified healthcare provider.
Infusion Preparation
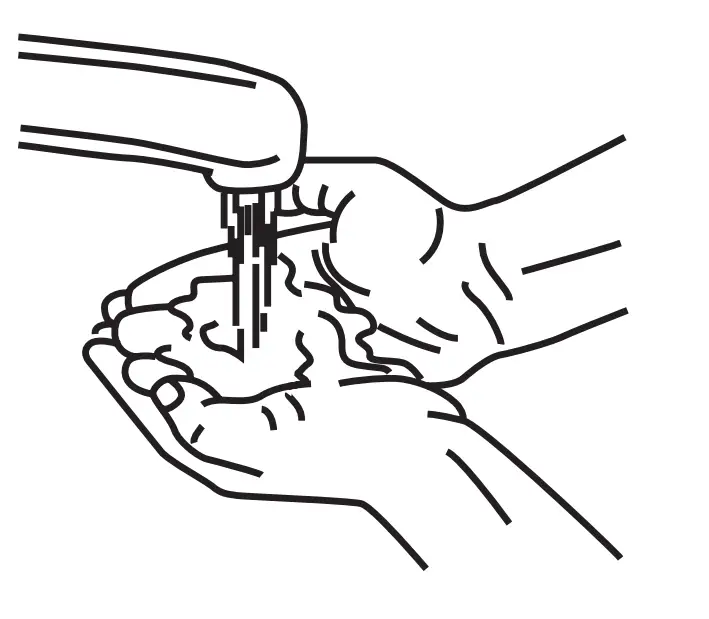
- Gather Supplies & Sanitize
Clean your infusion work surface with antiseptic wipes or disinfecting solution. Wash your hands thoroughly and, if required, put on disposable gloves. Lay out your supplies.
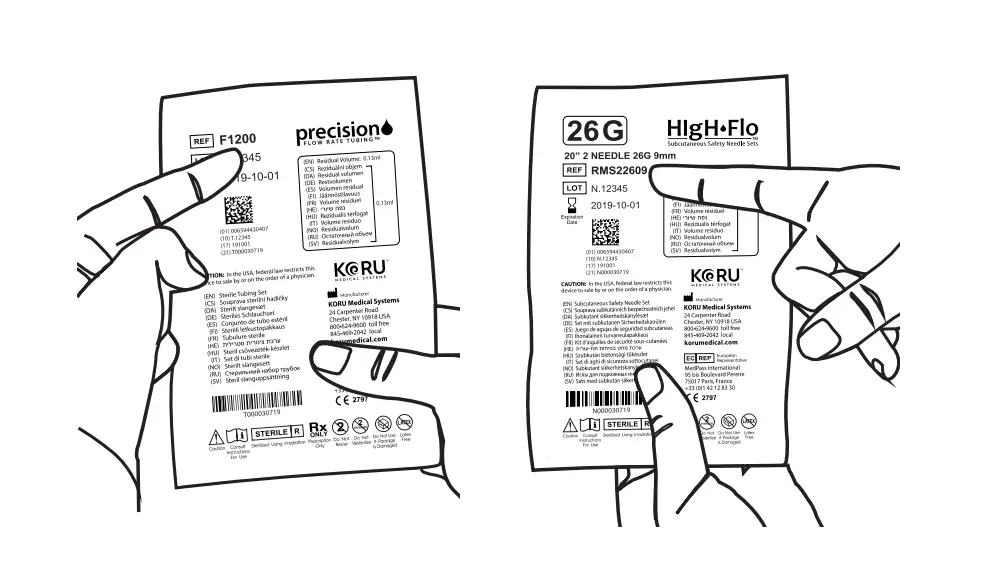
- Verify Flow Rate Tubing & Needles
Verify that you are using the correct Precision Flow Rate Tubing and HIgH-Flo Needle Sets prescribed by your healthcare provider. Inspect tubing and needle sets for damage. If damaged, replace and contact your healthcare provider.
- Prepare Syringe(s)
Ensure the medication is at room temperature (68-77°F or 20-25°C). Refer to the drug manufacturer’s instructions or ask your healthcare provider for detailed filling instructions. If using a prefilled syringe, go to Step 4. If using vials, fill the BD 20 or 30 ml syringe(s) with the required dose.
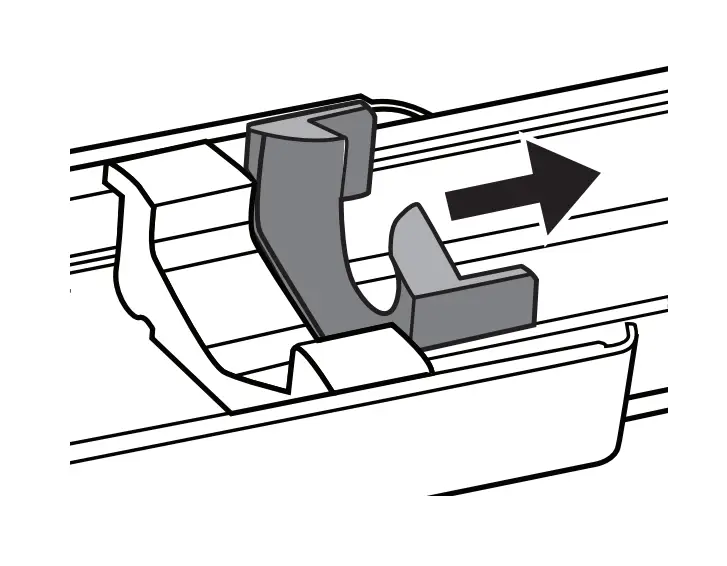
- Attach Tubing & Needles
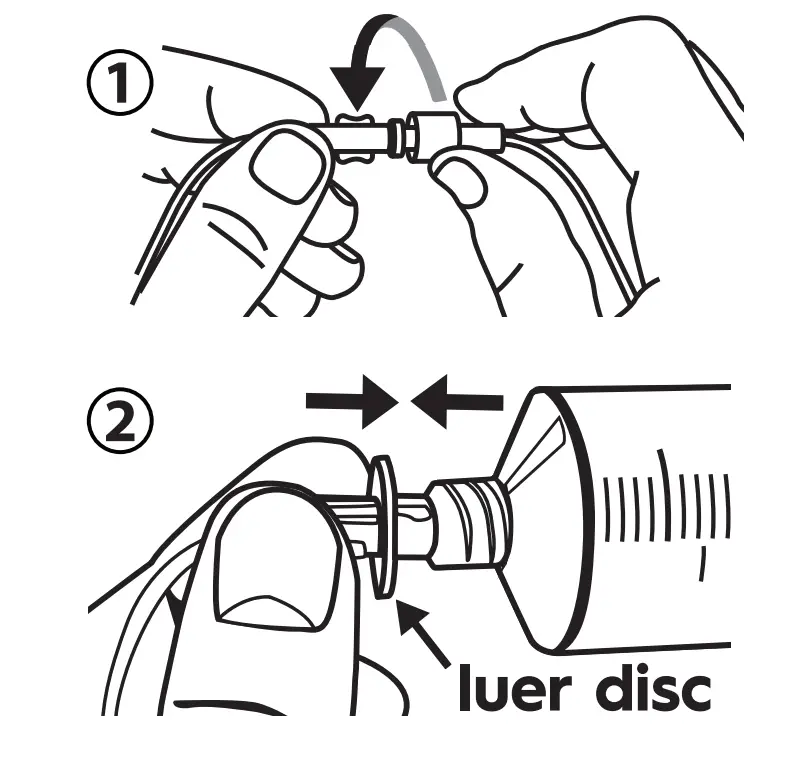
Remove sterile caps from ends of the Precision Flow Rate Tubing set and HIgH-Flo Subcutaneous Needle set and connect, using care not to contaminate the ends. Remove the cap from the luer disc end of the flow rate tubing set with aseptic technique and connect to the syringe.
- Prime (Fill) Tubing
Always follow your healthcare provider’s protocol. Focus on a single needle and try to stop the flow when the fluid approaches the needle. Be careful not to prime to the needle tip. Push the syringe plunger and follow the medication as it flows through the tube. Release pressure from the plunger to stop the flow.
NOTE:
It is recommended to insert the needles dry to minimize site irritation.
To best see the medication, we suggest priming the tubing against a dark, solid-colored surface in a well-lit area.
Insert Needles and Check for Blood Return:
NOTE: Always refer to the medicial product manufacturer’s prescribing information and recommendations from your healthcare provider for infusion site location(s). The most common areas for subcutaneous infusion include the abdomen, thighs, side of the upper hips and back of the arms.*
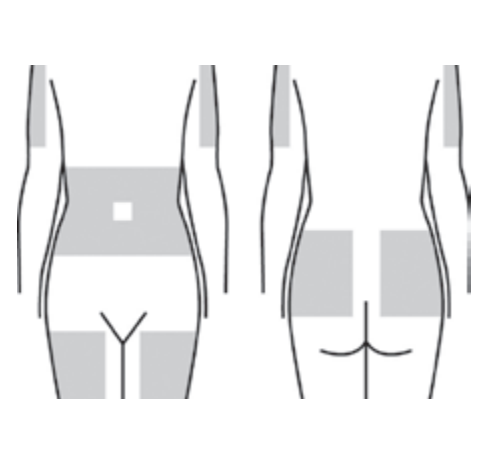
Prepare Sites
Select and clean site(s) before inserting needles. Carefully remove the shield from the needle tip, with care not to touch the needle.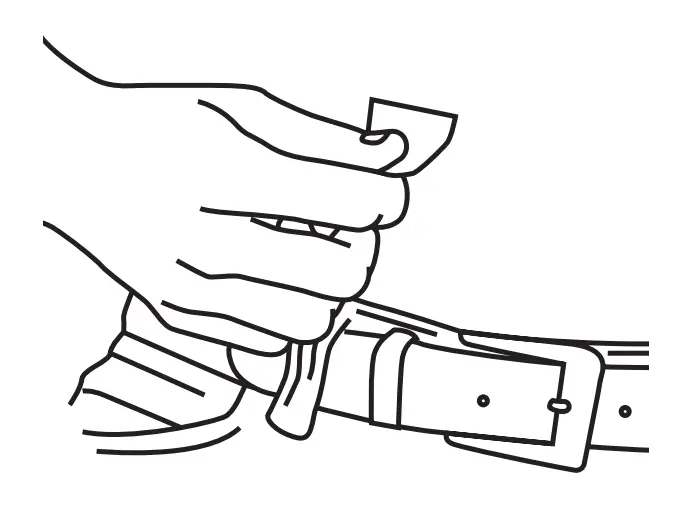
Insert Needles
Pinch the skin and insert each needle into the subcutaneous tissue at a 90˚ angle.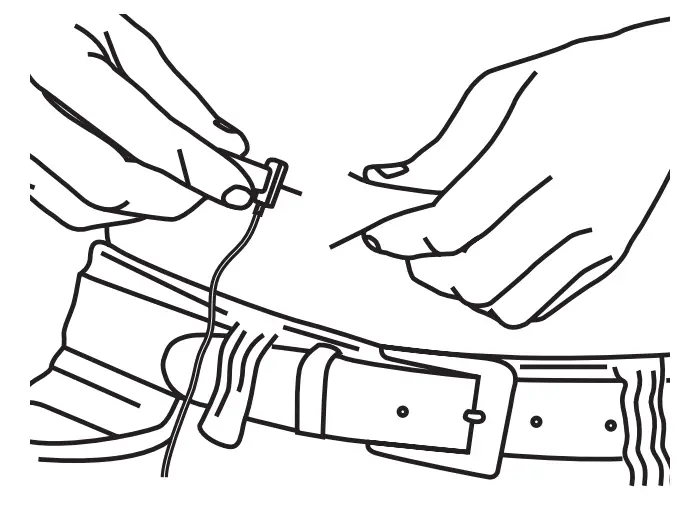
Secure Needles
Peel the printed side from the dressing to expose adhesive. Secure the needle by placing the adhesive dressing in the center of the needle butterfly. Smooth it outward over skin.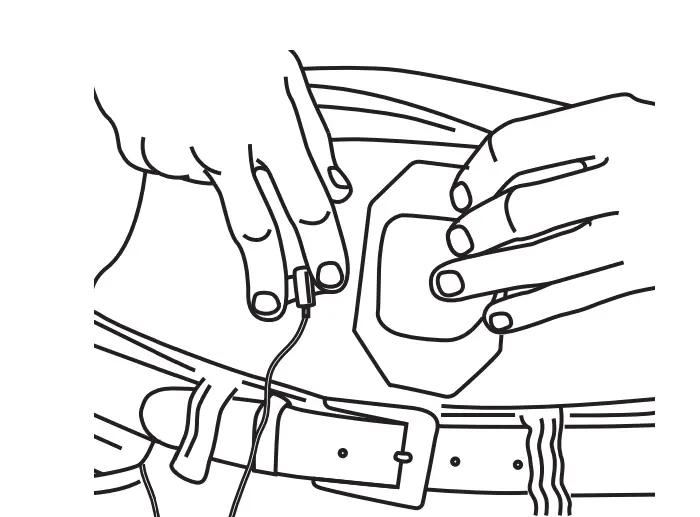
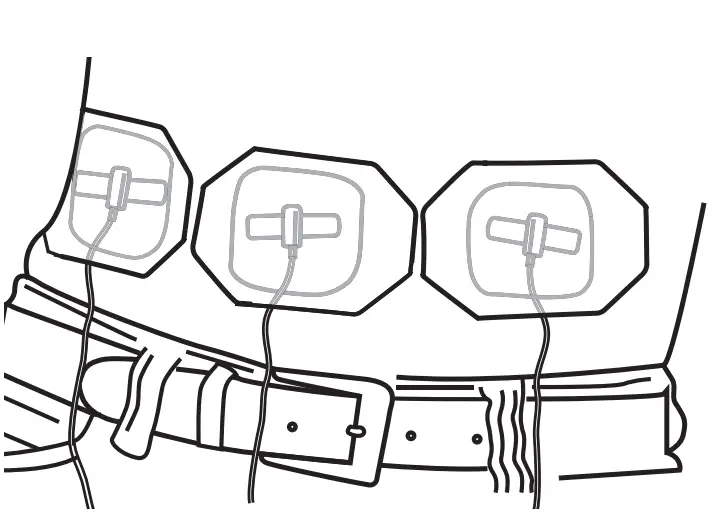
Check for Blood Return
Check for blood return if instructed by your healthcare provider by gently pulling back on the syringe plunger. Watch to make sure no red/pink appears in tubing near your sites. If blood return exists and if instructed by your healthcare provider, either clamp the flow to the needle site(s) or remove all needles, attach a new needle set, and start again from Step 4.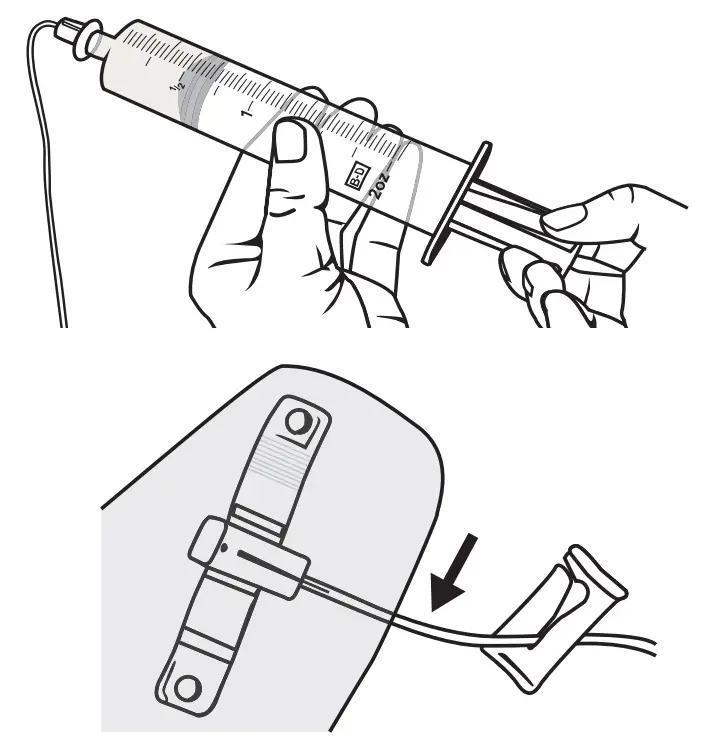
Starting & Ending Infusion
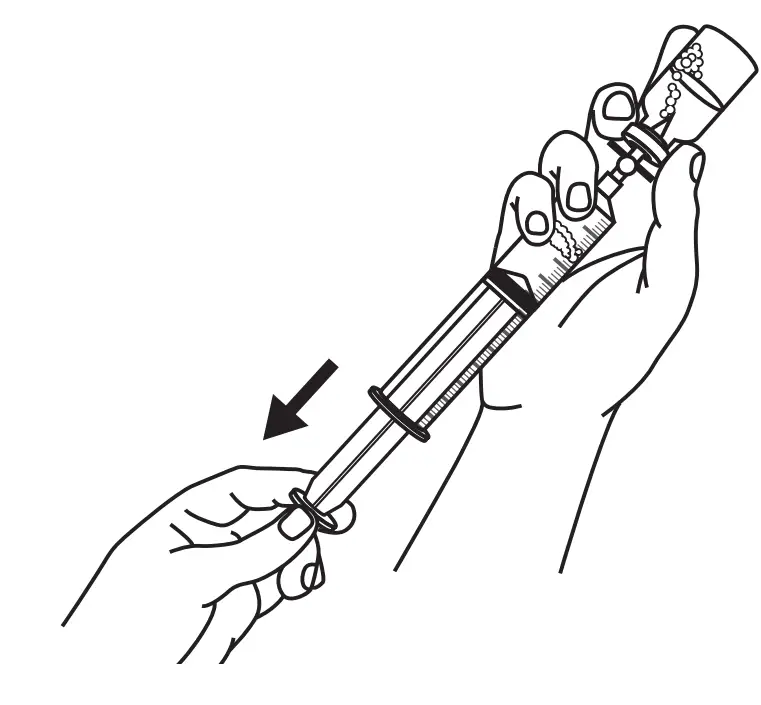
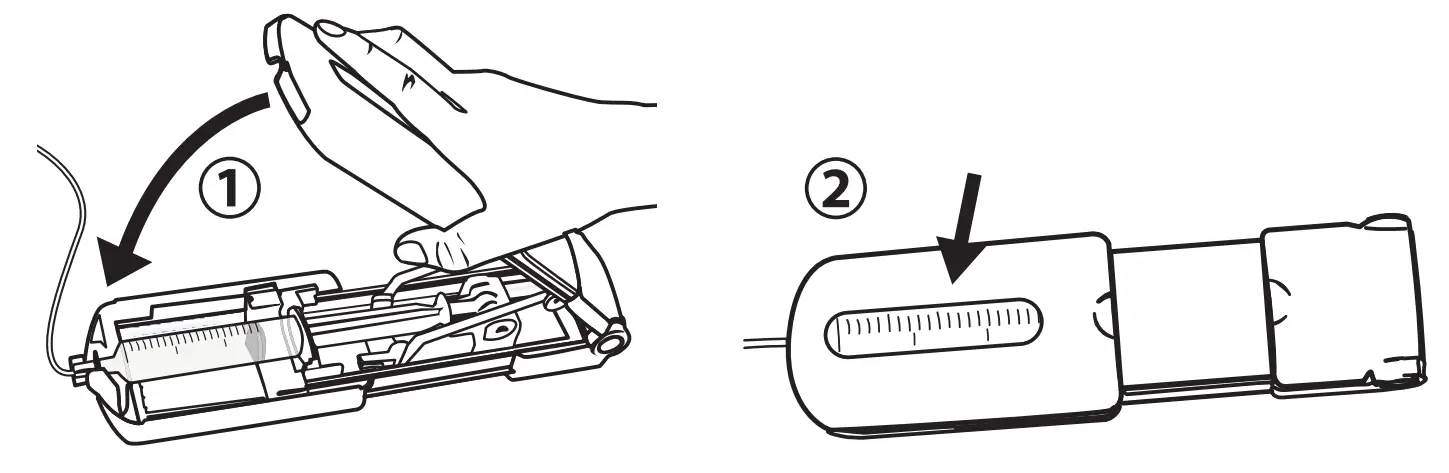
Open Syringe Driver
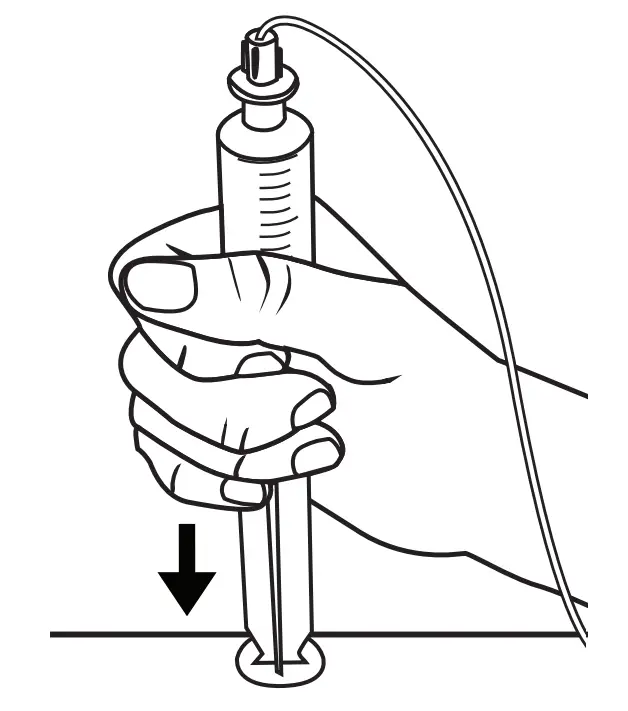
- Pull firmly to fully extend the syringe driver.

- Then, open the syringe driver fully by lifting the top cover.

Load Syringe
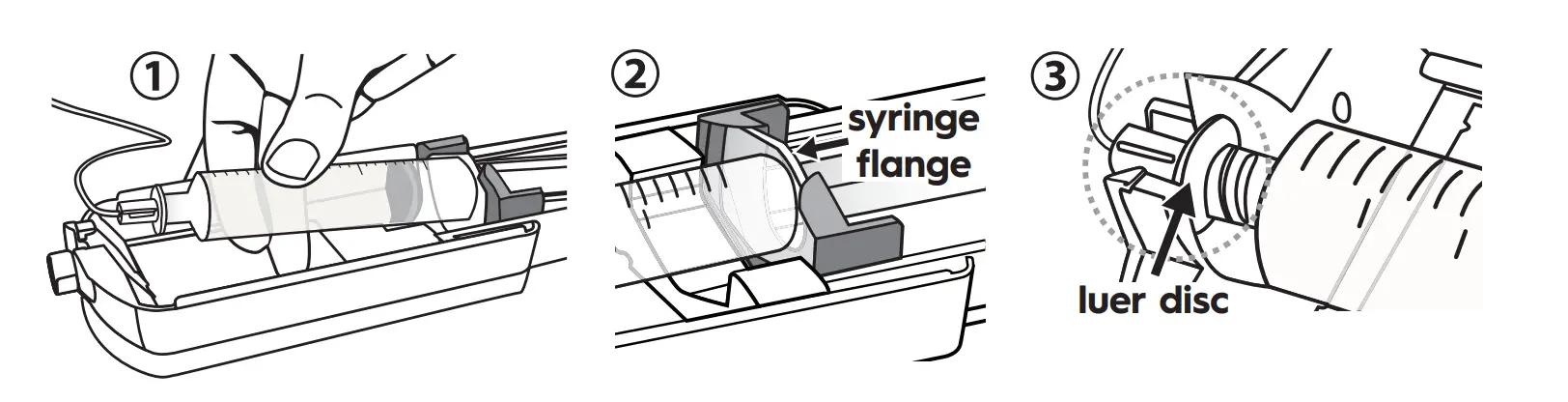
- With syringe gradations facing up, push the syringe flange against the orange syringe locater.
- Make sure the syringe flange is seated within the front part of the orange syringe locater.
- Verify that the Precision tubing (with the luer disc) is connected to syringe. Place the luer disc inside the syringe driver nose so that the syringe is firmly seated inside the syringe driver.
NOTE:
- You should not need to use significant force to load or remove the syringe. You can test proper fit by gently tugging on the syringe. It will stay in place if properly attached.
- When closing the syringe driver, make sure that the top lid is fully extended and aligns with the bottom portion.

Begin Infusion
- To start the infusion, close the top lid. The infusion will begin immediately. To reduce flow rate variability, try to keep the syringe driver level with your infusion sites.
- Periodically monitor the infusion by checking the progress window, until syringe is empty.
To pause infusion: Simply open the syringe driver. To continue, re-close the top lid.
If using multiple syringes: Once the first syringe is empty, open the FreedomEdge. Remove the syringe from the syringe driver and disconnect from tubing. With aseptic technique, connect the additional syringe to the luer disc end of the Precision tubing set. Load the prepared syringe into the syringe driver. Close the top lid to continue infusion. Repeat until total dosage is complete.
End of Infusion
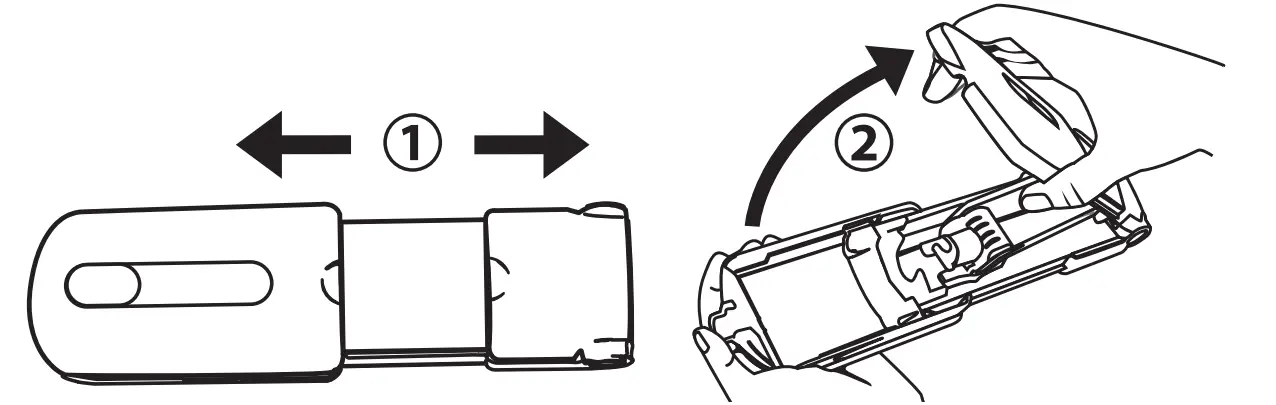
When the syringe is completely empty and total dosage is administered, open the top lid of the syringe driver. Remove the empty syringe and its tubing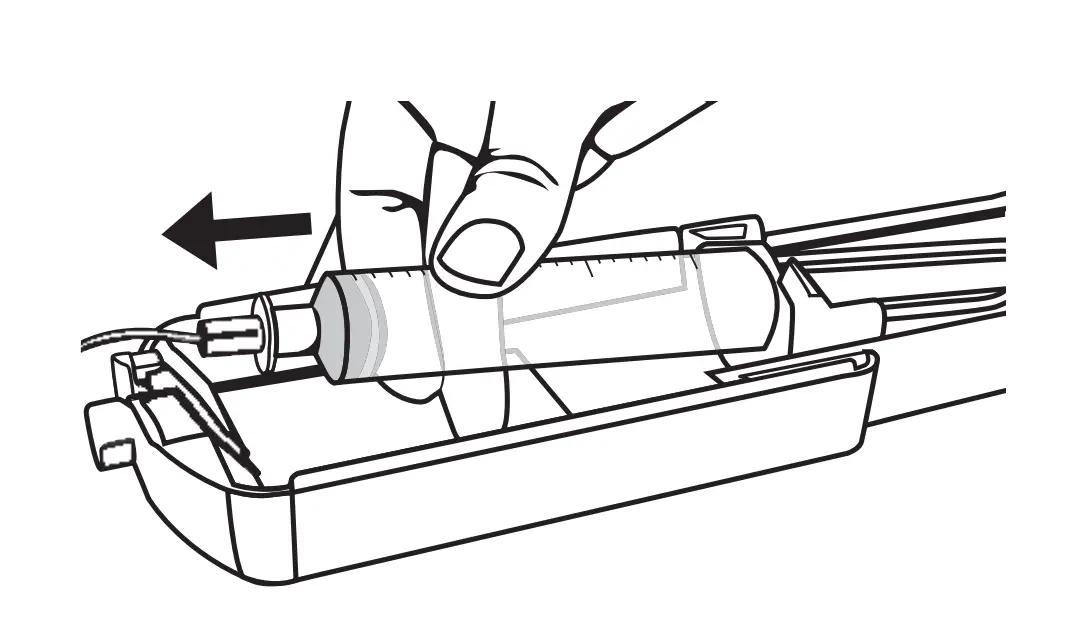
Remove Needle(s) & Cleanse Sites
Holding the needle in place, peel back the surrounding adhesive dressing. Remove the needle in a straight motion, opposite of the direction you inserted it. To use safety feature, close wings over the needle and snap shut.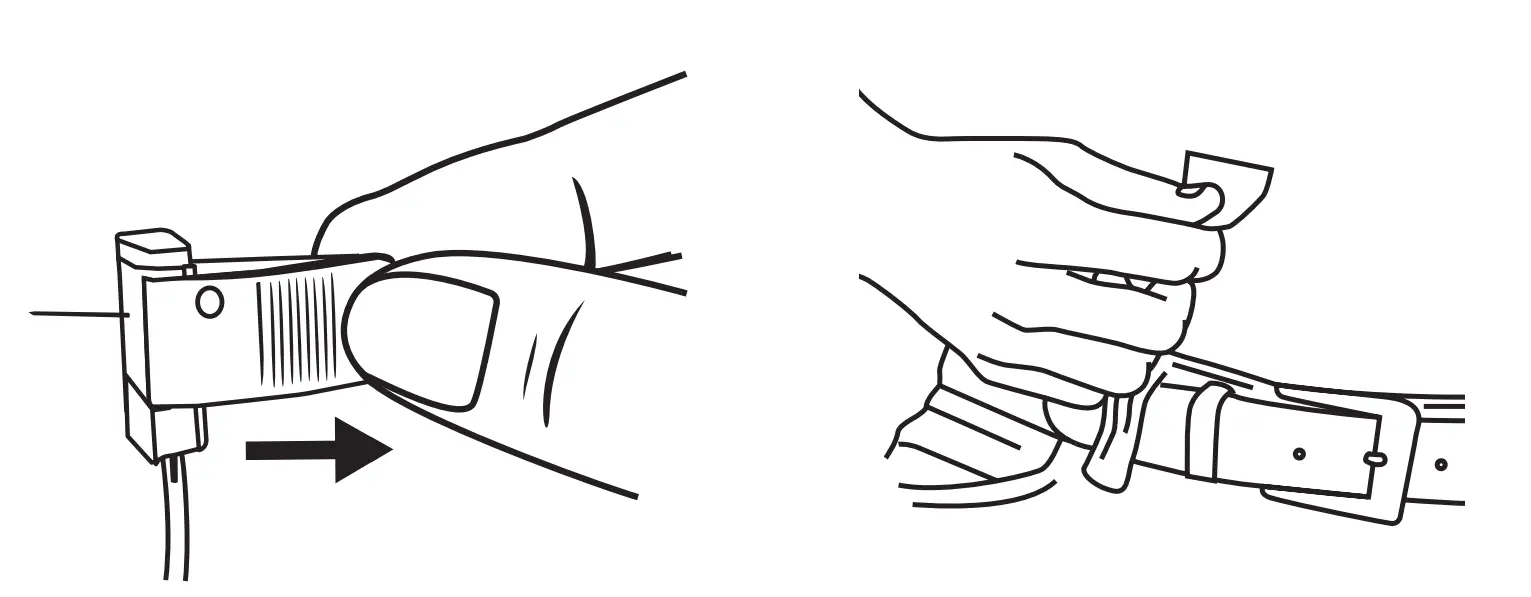
Discard Sharps & Clean
Discard all sharps and supplies as instructed by your healthcare provider. Remove visible soil as soon as possible after use of the device. Cleaning should be initiated as soon as possible after use of the device and delays between steps should be avoided. See page 11 for full cleaning instructions.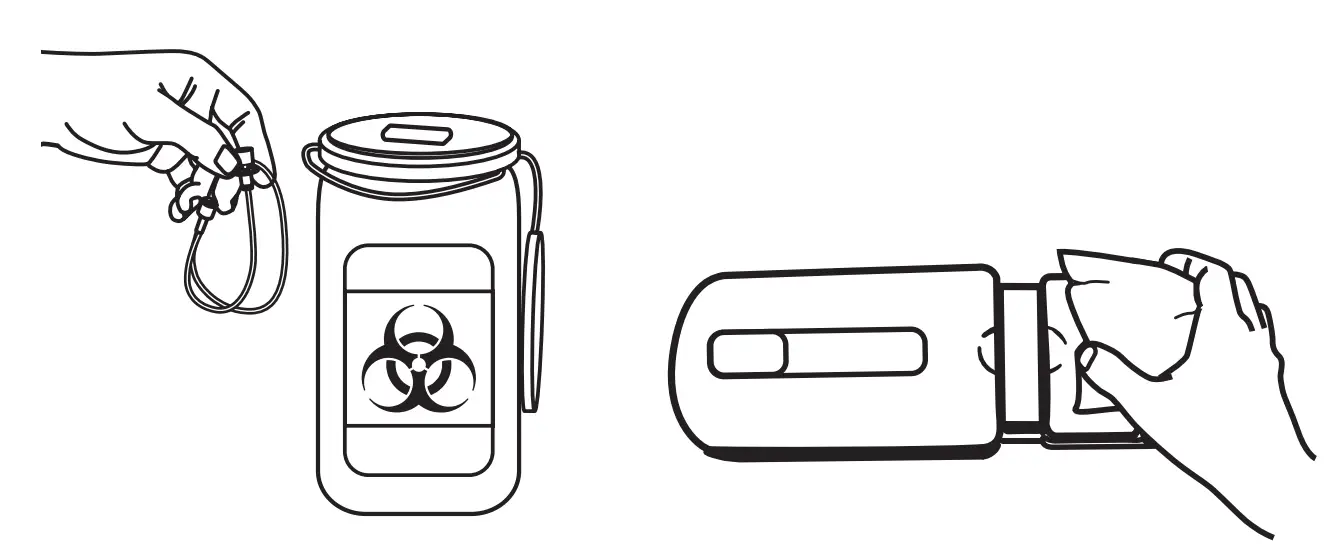
Step-by-Step Instructions for Intravenous Administration
Before intravenous self-administration, patients and/or caregivers should be properly trained by a qualified healthcare provider.
Infusion Preparation:
Gather Supplies & Sanitize
Clean your infusion work surface with antiseptic wipes or disinfecting solution. Wash your hands thoroughly. Lay out your supplies.
Verify Flow Rate Tubing
Verify that you are using the correct Precision Flow Rate tubing prescribed by your healthcare provider. Inspect the tubing set for damage. If damaged, replace and contact your healthcare provider.
Prepare Syringe(s)
Refer to the drug manufacturer’s instructions or ask your healthcare provider for detailed filling instructions. If using a prefilled syringe, go to Step 4. If using vials, fill the BD® 20 or 30 ml syringe(s) with the required dose.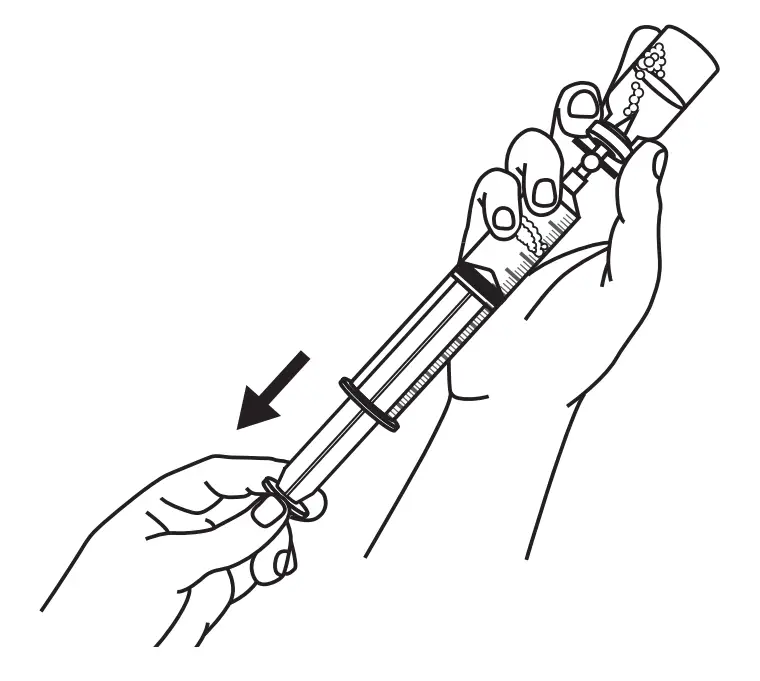
Attach Tubing
Remove cap from the luer disc end of the flow rate tubing set with aseptic technique and connect to the syringe.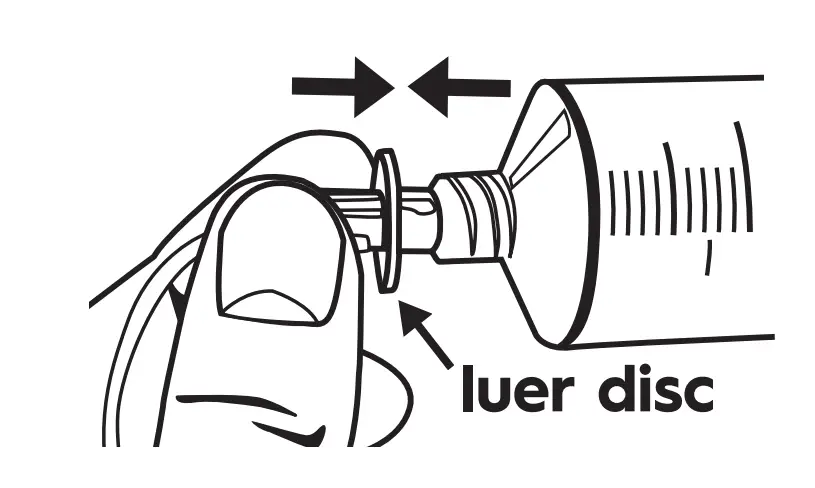
Prime (Fill) Tubing
Always follow your healthcare provider’s instructions. Loosen the cap on the Precision tubing set. Push the syringe plunger and follow the medicinal product as it flows through the tube. Release pressure from the plunger to stop the flow. When medication starts to drip, tighten the cap.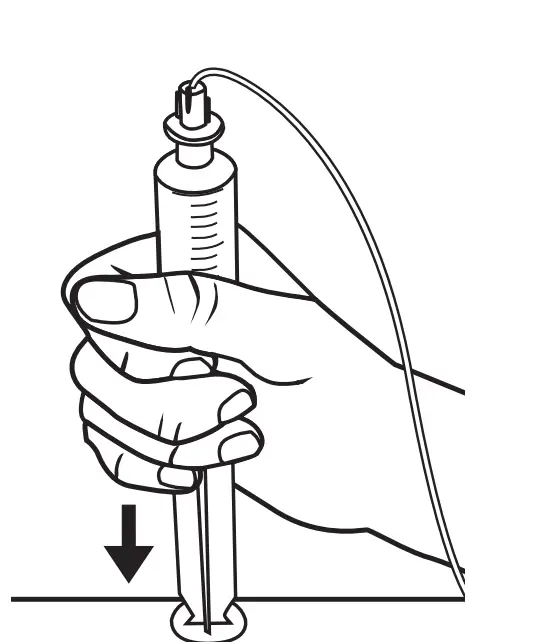
NOTE: To best see the medication, we suggest priming the tubing against a dark, solid-colored surface in a well-lit area.
Starting & Ending Infusion
Begin Infusion
Follow the instructions of the healthcare provider for cleansing and preparing the vascular access device.
- Cleanse with alcohol – after 15 seconds scrub allow to dry completely.
- Aspirate for blood return to ensure the vascular access device is open and unobstructed before each access.
Uncap the Precision tubing set and connect to the vascular access device or needle-free connector.
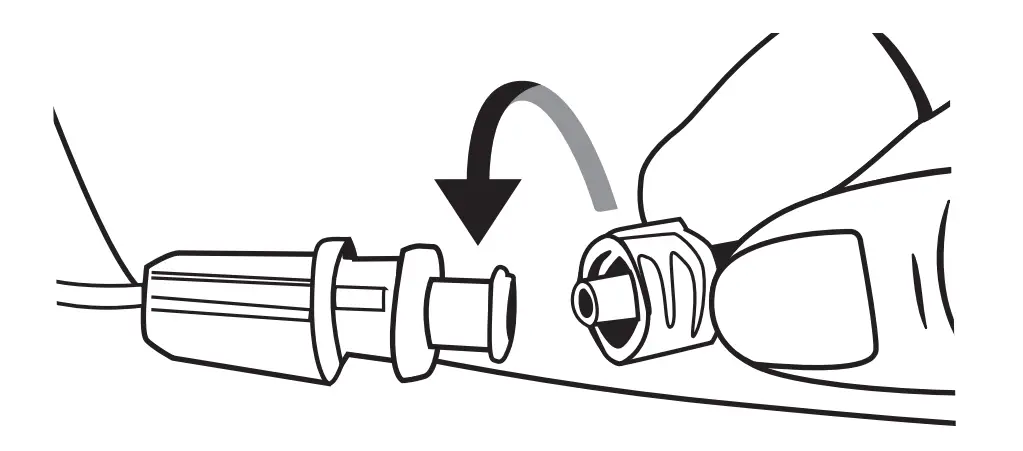
Open the syringe driver:
- Pull firmly to fully extend the syringe driver.

- Then, open the syringe driver fully by lifting the top lid.
Load syringe and begin:
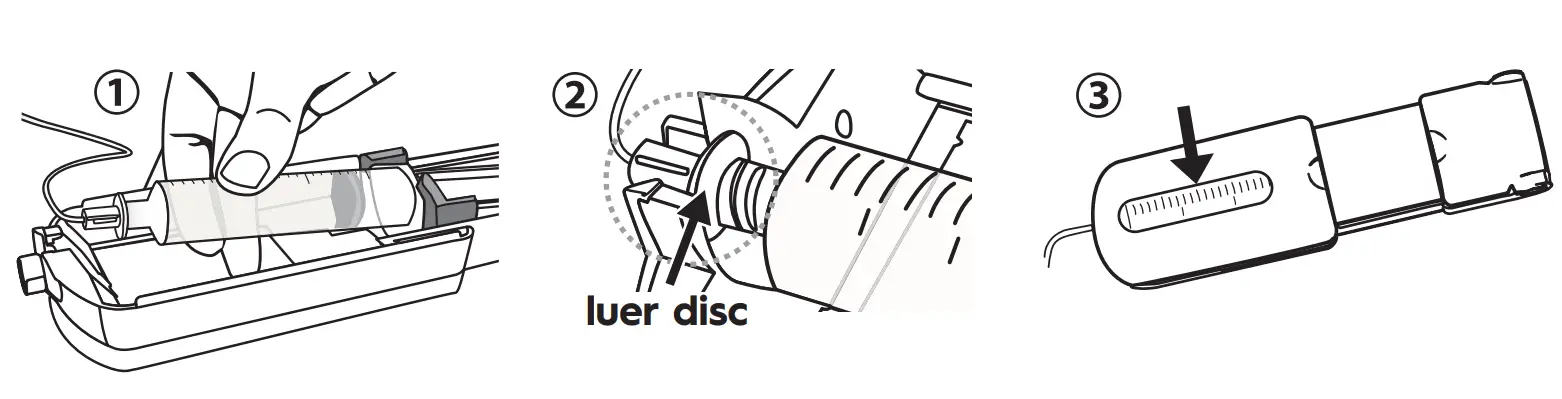
- With syringe gradations facing up, push the syringe flange against the orange syringe locater.
- Make sure the syringe flange is seated within the front part of the orange syringe locater. Place the luer disc inside the syringe driver nose so the syringe is firmly seated inside the syringe driver.
- Close the top lid to start the infusion. Periodically monitor by checking the progress window, until syringe is empty. To pause infusion: Simply open the syringe driver. To continue, re-close the top lid.
NOTE:
- You should not need to use significant force to load or remove the syringe. You can test proper fit by gently tugging on the syringe. It will stay in place if properly attached.
- When closing the syringe driver, make sure that the top lid is fully extended and aligns with the bottom portion.
If using multiple syringes: Once the first syringe is empty, open the FreedomEdge. If instructed, close the clamp on the vascular access device. Remove the syringe from the syringe driver and disconnect from tubing. With aseptic technique, connect the additional syringe to the luer disc end of the Precision tubing set and load back into the syringe driver. If closed, open the clamp on the vascular access device. Close the top lid to continue infusion. Repeat until total dosage is complete.
End of Infusion
When the syringe is completely empty and the total dosage is infused, open the Freedom Edge and remove the empty syringe. If instructed, close the clamp on the vascular access device. Disconnect Precision tubing from the vascular access device or needlefree connector.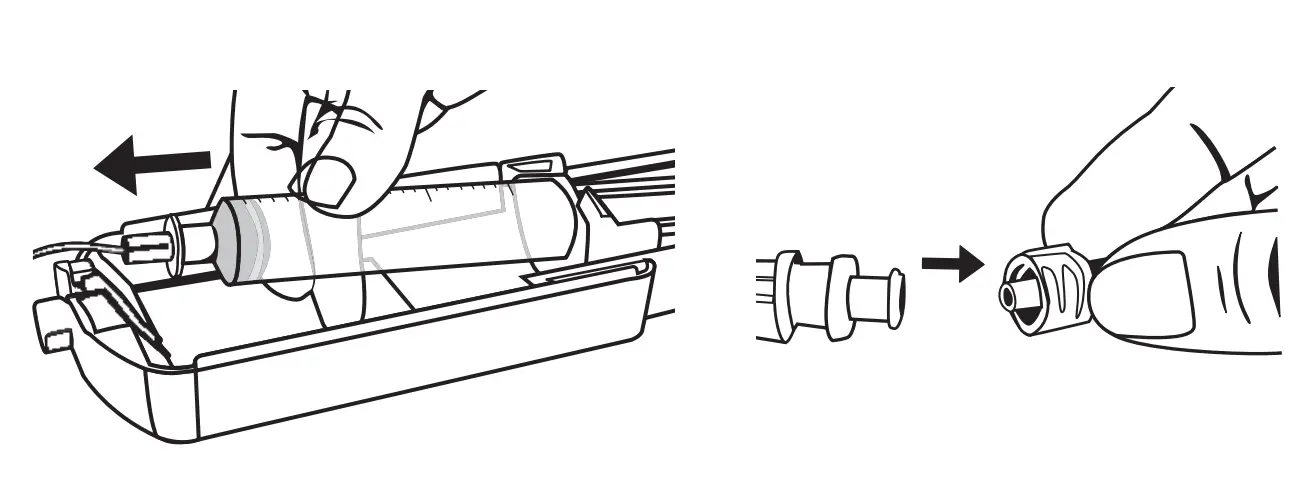
Flush
Always follow the healthcare provider’s instructions on flushing the vascular access device. Refer to the SASH technique below.*
 Saline Flush: Ensure the vascular access device is open and unobstructed.
Saline Flush: Ensure the vascular access device is open and unobstructed. Administer: Administer the medication.
Administer: Administer the medication. Saline Flush: Clear the residual medication from the vascular access device and ensure the vascular access device is open and unobstructed.
Saline Flush: Clear the residual medication from the vascular access device and ensure the vascular access device is open and unobstructed. Heparin (If required for patency): Minimize the potential of a blood clot forming inside the vascular access device.
Heparin (If required for patency): Minimize the potential of a blood clot forming inside the vascular access device.
Discard Supplies & Clean

Discard all supplies as instructed by your healthcare provider. Remove visible soil as soon as possible after use of the device. Cleaning should be initiated as soon as possible after use of the device and delays between steps should be avoided. See page 11 for full cleaning instructions
Troubleshooting
If the suggestions in this section do not solve your problem, or if problems persist, discontinue use and consult your healthcare provider. Any serious incident should be reported to your healthcare provider and KORU Medical Systems at +1 845-469-2042.
Syringe will not load or remove from syringe driver:
NOTE: You should not need to use significant force to load or remove a syringe.
- Ensure the syringe driver is fully open, and that nothing is blocking the syringe locator.
- Confirm you are not overfilling the syringe (filling a 20 ml syringe with more than 20 ml of solution or a 30 ml syringe with more than 30 ml of solution), or using a syringe larger than 30 ml.
- If you still have difficulty, use one hand to slide the orange syringe locator all the way back, then place the syringe.
Syringe will not stay inside in the syringe driver:
- Make sure you are using the proprietary Precision Flow Rate Tubing™ sets and that the luer disc end of the tubing has been connected to a BD® 20 or 30 ml syringe or a Hizentra® 20 ml prefilled syringe.
- Make sure the luer disc is seated properly in the nose of the syringe driver.
- Make sure the flange shape of the syringe is correctly seated into the shape of the orange syringe locator.
No flow:
- Open and close the lid to ensure the syringe pusher slides freely and does not bind.
- Make sure all the slide clamps are unclamped. If vascular access device is being used, make sure its clamps, if any, are open.
- Use aseptic technique as recommended by the healthcare provider; disconnect the tubing set from the needle set, vascular access device or needle-free connector, and check for medication drip. If the medication does not drip:
- Subcutaneous administration: replace the tubing as it may be damaged.
- Intravenous administration: check that the catheter is open and unobstructed.
Slow flow:
- If the slide clamp has been used, the tubing may be damaged.
- Verify that you are using the proper syringe. 30 ml syringes will flow approx. 73% of the rate of a 20 ml syringe.
- Ensure the syringe driver is level with the infusion sites. If the syringe driver is positioned lower than the sites, the flow rate may be slower than expected.
- Subcutaneous administration:
- Administration may be slow based on how well the medication is absorbed through the tissue. Some infusions may be faster than others. The first infusions may take longer than expected because the body may need to adapt.
- Avoid placing needles on top of scar tissue or muscle.
- It is possible you may need more sites, longer needles or a faster flow rate tubing set. Talk to your healthcare provider.
Stopping the flow quickly:
- The syringe driver is designed to maintain pressure during and after the infusion to prevent blood/drug backflow.
- To stop the flow, fully open the top lid to relieve pressure from the syringe plunger.
- The slide clamp can be used in the case of an emergency.
Medication (5 ml or less) left in the syringe:
- Verify that you are using a proper recommended syringe: BD® 20 or 30 ml, or Hizentra® 20 ml prefilled syringe.
- If the syringe does not completely empty, contact the healthcare provider.
Subcutaneous swelling, pain or redness at the site:
- It is recommended to insert subcutaneous needles dry as the medication may irritate the skin.
- Assure that the needles are long enough to reach the subcutaneous layer. If the selected needle is too short, leaking at the site may occur.
- Assure that the needles are not too long, as they may hit muscle.
- Try a slower flow rate tubing set as the rate may be too fast.
- Rotate infusion sites if recommended by your healthcare provider. Periodically returning to sites that worked well in the past may provide best results.
Care, Maintenance and Reprocessing
The FreedomEdge® does not require any preventative maintenance. Since it works as a system, the tubing determines the flow rate, not the syringe driver; therefore the syringe driver needs no calibration. If you choose the correct tubing set, the proper flow rate will be achieved.
Between uses, the FreedomEdge syringe driver needs to be first thoroughly cleaned, and then disinfected.
After cleaning and disinfection, inspect the device for unacceptable deterioration such as corrosion, discoloration, pitting, and cracked seals and properly dispose any devices that fail the inspection.
Cleaning Procedure:
- The FreedomEdge may be cleaned with a soft cloth dampened with a weak mixture of mild detergent and warm water (minimum ratio of 1 part detergent to 50 parts water by volume).
- Using the prepared detergent solution and a clean non-linting wipe or soft cloth, wipe all the external surfaces of the syringe driver, including the driver nose and syringe tray up to the syringe shield for at least one (1) minute. During the one (1) minute wipe, pay special attention to the ridges, crevices, raised lettering during wiping. Replace soiled cloths or wipes as needed, changing wipes when necessary to ensure that all surfaces are cleaned. Caution: Clean only those areas that are exposed and external. No attempt should be made to clean any part of the syringe driver that is not easily accessible.
- Using a clean non-linting wipe or soft cloth wetted with room temperature tap water (wet but not dripping), wipe all the external surfaces of the syringe driver, including the driver nose and syringe tray up to the syringe shield. Pay special attention to the ridges, crevices, raised lettering during wiping. Continue wiping until all residue is removed to ensure the syringe driver is thoroughly clean. Replace or re-wet cloth or wipes as needed, changing wipes when necessary to ensure that all surfaces are rinsed.
- Dry the device using a clean non-linting wipe or soft cloth.
- Inspect the device for any visible soil after the cleaning steps (but before the disinfection steps) to ensure that the device is thoroughly cleaned between uses prior to disinfection. If the device has remaining visible soil following cleaning, repeat the cleaning steps (1 through 4).
Disinfection Procedure:
- Wipe the outside surfaces of the FreedomEdge® syringe driver with 70% Isopropyl Alcohol (IPA) and a non-linting cloth or wipe, or pre-saturated IPA wipe.
- Use pre-saturated IPA wipes, or non-linting wipes saturated with 70% Isopropyl Alcohol (IPA) (wetted but not dripping) to thoroughly wipe all exterior surfaces of the device. Ensure all external surfaces of the syringe driver, including the driver nose, syringe tray and top lid are wiped. Pay special attention to the ridges and crevices during wiping. Allow all surfaces to remain visibly wet for a minimum of five (5) minutes.
Caution: Clean only those areas that are exposed and external. No attempt should be made to clean any part of the syringe driver that is not easily accessible. - During the five (5) minute contact time, use additional wipes to ensure all contacted surfaces remain wet for the full contact duration time.
- Thoroughly dry the device using non-linting wipe(s) or allow to air dry.
- Visually inspect devices for signs of damage or wear.
Storage:
The Freedom Edge syringe driver and its components (Precision tubing sets and HIgH-Flo needle sets) recommended to be stored in a cool, dry place at room temperature (approximately 68 -77°F or 20 -25°C).
Testing Flow Accuracy (if required by your local protocol):
- Remove all air from a new BD® 20 ml syringe with sterile water. Do NOT use a 30 ml syringe for this test.
- Completely fill with 20 ml of sterile water.
- Attach a sterile F120 Precision Flow Rate Tubing set to the syringe.
- Remove all air from the tubing set.
- Load the syringe into the syringe driver.
- Using a stop watch or similar time tracking device, start the timer when the top lid of the syringe driver is fully closed (flow will begin).
- Monitor and stop the timer when 10 ml of water has left the syringe.
- The elapsed time should fall between 3:50-5:11 minutes.
NOTE: If the test results fall outside the range indicated in Step 8, factory refurbishment and testing are available. Please contact KORU Medical Systems at +1 845-469-2042.
Technical Specifications
Testing was performed in a controlled test lab environment and as a result infusions should be administered within the same environmental conditions of 68 -77°F (20 – 25°C) and atmospheric pressure of 1.01 bar (±0.09).
Syringe Driver:
Weight: 12 oz (0.34 kg)
Length:
Closed: 9” (229 mm), Extended: 11.75“ (299 mm)
Width: 3.25” (83 mm)
Height: 1.5” (38 mm)
Syringe: Reservoir volume: 20/30 ml
(BD® brand 20/30 ml syringes or
Hizentra® prefilled 20 ml syringes)
Target Operating Temperature:
68 -77°F (20 – 25°C)
Height Sensitivity:
| Vertical Height (inches) | % Variation From Target Flow Rate |
| ±3 inches from infusion site | Equivalent to Level |
| ±6 inches from infusion site | up to ±1.2% from target flow rate |
| ±12 inches from infusion site | up to ±2.4% from target flow rate |
| ±24 inches from infusion site | up to ±4.8% from target flow rate |
System Max Operating Pressure:
| Tubing/Needle Combo | Pressure at the Beginning of Needle Set (psi) | Measured Pressure at End of Needle Set (psi) |
| F60 + 24G | 0.3 psi | 0 psi |
| F2400 + 24G | 7.7 psi | 0 psi |
Data represents pressure changes through the Freedom System (Freedom syringe driver, Precision Flow Rate Tubing™, and HIgH-Flo Subcutaneous Safety Needle Sets™) with the slowest flow rate parameter (F60) and the fastest flow rate parameter (F2400). The net effect: the pressure at the needle is significantly reduced from the initial head pressure.
Ancillary Supply Product Information
Precision Flow Rate Tubing™ Sets:
| Description | Item # | Residual Vol. | p/Box |
| Very Low Flow | F0.5 | 0.09 ml | 50 |
| Very Low Flow | F1 | 0.08 ml | 50 |
| Very Low Flow | F2 | 0.10 ml | 50 |
| Very Low Flow | F3 | 0.09 ml | 50 |
| Very Low Flow | F3.8 | 0.09 ml | 50 |
| Very Low Flow | F5 | 0.08 ml | 50 |
| Very Low Flow | F8 | 0.08 ml | 50 |
| Very Low Flow | F10 | 0.14 ml | 50 |
| Very Low Flow | F15 | 0.11 ml | 50 |
| Low Flow | F30 | 0.13 ml | 50 |
| Low Flow | F45 | 0.11 ml | 50 |
| Description | Item # | Residual Vol. | p/Box |
| Low Flow | F60 | 0.14 ml | 50 |
| Low Flow | F120 | 0.16 ml | 50 |
| Low Flow | F180 | 0.13 ml | 50 |
| High Flow | F275 | 0.11 ml | 50 |
| High Flow | F420 | 0.10 ml | 50 |
| High Flow | F500 | 0.09 ml | 50 |
| High Flow | F600 | 0.09 ml | 50 |
| High Flow | F900 | 0.08 ml | 50 |
| High Flow | F1200 | 0.13 ml | 50 |
| High Flow | F2400 | 0.15 ml | 50 |
Flow Rate Starter Kits:
| Item Number | Description | Contents per Box |
| H20KT | High Flow Starter Kit | (2) F275, (5) F600, (5) F900, (4) F1200, (4) F2400 |
| L20KT | Low Flow Starter Kit | (2) F30, (5) F45, (5) F60, (4) F120, (4) F180 |
KORU Related Accessories:
| Item # | Description | Residual Vol. |
| LRVY | Low Residual Volume Y-Connector | 0.14 ml |
| FEXT | 24” Extension Set | 0.4 ml |
| Low Flow | F60 | 0.14 ml | 50 |
| Low Flow | F120 | 0.16 ml | 50 |
| Low Flow | F180 | 0.13 ml | 50 |
| High Flow | F275 | 0.11 ml | 50 |
| High Flow | F420 | 0.10 ml | 50 |
| High Flow | F500 | 0.09 ml | 50 |
| High Flow | F600 | 0.09 ml | 50 |
| High Flow | F900 | 0.08 ml | 50 |
| High Flow | F1200 | 0.13 ml | 50 |
| High Flow | F2400 | 0.15 ml | 50 |
26G HIgH-Flo Subcutaneous Safety Needle Sets™
| Single-Needle Sets | |||
| Length | Item # | Residual Vol. | p/ Box |
| 4 mm | RMS12604 | 0.1 ml | 20 |
| 6 mm | RMS12606 | 0.1 ml | 20 |
| 9 mm | RMS12609 | 0.1 ml | 20 |
| 12 mm | RMS12612 | 0.1 ml | 20 |
| 14 mm | RMS12614 | 0.1 ml | 20 |
| Two-Needle Sets | |||
| Length | Item # | Residual Vol. | p/ Box |
| 4 mm | RMS22604 | 0.2 ml | 10 |
| 6 mm | RMS22606 | 0.2 ml | 10 |
| 9 mm | RMS22609 | 0.2 ml | 10 |
| 12 mm | RMS22612 | 0.2 ml | 10 |
| 14 mm | RMS22614 | 0.2 ml | 10 |
| Three-Needle Sets | |||
| Length | Item # | Residual Vol. | p/ Box |
| 4 mm | RMS32604 | 0.3 ml | 10 |
| 6 mm | RMS32606 | 0.3 ml | 10 |
| 9 mm | RMS32609 | 0.3 ml | 10 |
| 12 mm | RMS32612 | 0.3 ml | 10 |
| 14 mm | RMS32614 | 0.3 ml | 10 |
| Four-Needle Sets | |||
| Length | Item # | Residual Vol. | p/ Box |
| 4 mm | RMS42604 | 0.4 ml | 10 |
| 6 mm | RMS42606 | 0.4 ml | 10 |
| 9 mm | RMS42609 | 0.4 ml | 10 |
| 12 mm | RMS42612 | 0.4 ml | 10 |
| 14 mm | RMS42614 | 0.4 ml | 10 |
| Five-Needle Sets | |||
| Length | Item # | Residual Vol. | p/ Box |
| 4 mm | RMS52604 | 0.5 ml | 10 |
| 6 mm | RMS52606 | 0.5 ml | 10 |
| 9 mm | RMS52609 | 0.5 ml | 10 |
| 12 mm | RMS52612 | 0.5 ml | 10 |
| 14 mm | RMS52614 | 0.5 ml | 10 |
| Six-Needle Sets | |||
| Length | Item # | Residual Vol. | p/ Box |
| 4 mm | RMS62604 | 0.6 ml | 10 |
| 6 mm | RMS62606 | 0.6 ml | 10 |
| 9 mm | RMS62609 | 0.6 ml | 10 |
| 12 mm | RMS62612 | 0.6 ml | 10 |
| 14 mm | RMS62614 | 0.6 ml | 10 |
24G HIgH-Flo Subcutaneous Safety Needle Sets™:
| Single-Needle Sets | |||
| Length | Item # | Residual Vol. | p/ Box |
| 6 mm | RMS12406 | 0.4 ml | 20 |
| 9 mm | RMS12409 | 0.4 ml | 20 |
| 12 mm | RMS12412 | 0.4 ml | 20 |
| 14 mm | RMS12414 | 0.4 ml | 20 |
| Two-Needle Sets | |||
| Length | Item # | Residual Vol. | p/ Box |
| 6 mm | RMS22406 | 0.7 ml | 10 |
| 9 mm | RMS22409 | 0.7 ml | 10 |
| 12 mm | RMS22412 | 0.7 ml | 10 |
| 14 mm | RMS22414 | 0.7 ml | 10 |
| Three-Needle Sets | |||
| Length | Item # | Residual Vol. | p/ Box |
| 6 mm | RMS32406 | 1.1 ml | 10 |
| 9 mm | RMS32409 | 1.1 ml | 10 |
| 12 mm | RMS32412 | 1.1 ml | 10 |
| 14 mm | RMS32414 | 1.1 ml | 10 |
| Four-Needle Sets | |||
| Length | Item # | Residual Vol. | p/ Box |
| 6 mm | RMS42406 | 1.4 ml | 10 |
| 9 mm | RMS42409 | 1.4 ml | 10 |
| 12 mm | RMS42412 | 1.4 ml | 10 |
Selected Flow Rate Tables
The following section is to guide healthcare providers in selecting the Precision Flow Rate Tubing™ and HIgH-Flo Subcutaneous Safety Needle Sets™* to achieve the desired flow rate based on the selected medication and number of infusion sites.
*HIgH-Flo Subcutaneous Safety Needle sets are only to be used for subcutaneous administration.
*All flow rate tables are based on bench top testing which was performed with 0 psi of back pressure
How to Use Flow Rate Tables for Subcutaneous Administration:
- Select prescribed medication and refer to its prescribing information for recommended infusion flow rate and infusion time.
- Select the subcutaneous needle type – 26G or 24G Verify the correct flow rate table.
- Evaluate and select flow rate tubing and number of needles based on the infusion phase and flow
EMPAVELI® (pegcetacoplan) Flow Rate Combinations:
The following table indicates the average, minimum, and maximum predicted infusion times with HIgH-Flo Subcutaneous Safety Needle Sets™ (Standard 26G) when used in combination with KORU Precision Flow Rate Tubing™ and Freedom Edge® Syringe Infusion System with a 20 ml BD syringe.
To determine the appropriate infusion time, please refer to the drug package insert. Only the specific combinations of Precision Flow Rate Tubing™ and Highs-Flo
Subcutaneous Safety Needle Sets™ identified in the table below are indicated for use with Empanel. Please note that Highs-Flo Super26 needle sets should not be confused with HIgH-Flo 26G. Do NOT use HIgH-Flo Super26 needle sets with Empanel.
HIgH-Flo 26G with Precision Tubing
Average (Min-Max) Infusion Time for 20 ml Syringe (Minutes)
| F500 | F600 | F900 | F1200 | F2400 | |
| 1 needle | 47 (28-65) | 44 (26-62) | 38 (22-54) | 37 (21-52) | 30 (16-43) |
| 2 needles | 35 (22-48) | 33 (20-45) | 27 (16-37) | 25 (14-36) | 18 (10-26) |
Hizentra®, Cuvier® and Gamma Gard® Flow Rate Combinations:
The following tables indicate selected predicted flow rates with HIgH-Flo Subcutaneous Safety Needle Sets™ (26G and 24G) when used in combination with KORU Precision Flow Rate Tubing™ and Freedom Edge® Syringe Infusion System for the subcutaneous use of Hizentra, Cuvitru, and Gamma Gard Liquid.
To determine the appropriate flow rate tubing and subcutaneous needle configure- tion, please refer to drug package insert for the maximum indicated flow rate and volume per infusion site for initial and subsequent infusions.
| Hizentra – Freedom Edge® with 20 ml syringe | |||||||
| Drug volume (ml) | Flow Rate Tubing | HIgH-Flo Needle Set* | Total Flow Rate (ml/hr) | Flow rate/site (ml/hr) | Vol/site (ml) | Time | NOTES: |
| 10 | F120 | RMS12609 | 8.28 | .2 | 10 | 1:12 | Suggested start Peds |
| 10 | F180 | RMS12609 | 10.5 | 10.5 | 10 | 0:57 | Suggested start Peds |
| 20 | F275 | RMS22609 | 17.1 | 8.51 | 01 | :10S | uggested start Peds |
| 20 | F600 | RMS22609 | 29.6 | 14.8 | 10 | 0:40 | Suggested start Peds |
| 40 | F600 | RMS32609 | 33.9 | 11.3 | 13.3 | 1:10 | Suggested start Adult |
| 40 | F900 | RMS32609 | 44.3 | 14.8 | 13.3 | 0:54 | Suggested start Adult |
| 60 | F900 | RMS42609 | 49 | 12.3 | 15 | 1:13 | Suggested start Adult |
| 50 | F2400 | RMS32609 | 72.2 | 24.1 | 16.670 | :41 | 6th Infusion of biologic and beyond |
| 100 | F2400 | RMS42609 | 85.5 | 21.4 | 25 | 1:10 | 6th Infusion of biologic and beyond |
| Hizentra – with Freedom Edge® with 30 ml syringe | |||||||
| 20 | F600 | RMS22609 | 22.5 | 11.2 | 10 | 0:53 | Suggested start Peds |
| 30 | F900 | RMS22609 | 28.3 | 14.2 | 15 | 1:03 | Suggested start Adult |
| 30 | F2400 | RMS22609 | 41.9 | 20.9 | 15 | 0:42 | 6th Infusion of biologic and beyond |
| Cuvitru – with Freedom Edge® with 20 ml syringe | |||||||
| Drug volume (ml) | Flow Rate Tubing | HIgH-Flo Needle Set* | Total Flow Rate (ml/hr) | Flow rate/site (ml/hr) | Vol/site (ml) | Time | NOTES: |
| 10 | F275 | RMS12609 | 12.1 | 12.1 | 10 | 0:49 | 1st Two Infusions patients under 40kg |
| 20 | F275 | RMS12609 | 12.1 | 12.1 | 20 | 1:39 | 1st Two Infusions patients under 40kg |
| 20 | F600 | RMS22609 | 25.7 | 12.8 | 10 | 0:47 | 1st Two Infusions patients under 40kg |
| 50 | F600 | RMS22609 | 25.7 | 12.8 | 25 | 1:57 | 1st Two Infusions patients over 40kg |
| 60 | F1200 | RMS22609 | 37.1 | 18.6 | 30 | 1:37 | Subsequent Infusions |
| 60 | F2400 | RMS22409 | 110.5 | 55.4 | 30 | 0:32 | Subsequent Infusions |
| 60 | F1200 | RMS12409 | 55.3 | 55.3 | 60 | 1:05 | Subsequent Infusions |
| 100 | F2400 | RMS42409 | 132.8 | 33.2 | 25 | 0:45 | Subsequent Infusions |
| Cuvitru – with Freedom Edge® with 30 ml syringe | |||||||
| 20 | F500 | RMS22609 | 12.9 | 12.9 | 20 | 1:32 | 1st Two Infusions patients under 40kg |
| 30 | F900 | RMS22609 | 24.6 | 12.3 | 15 | 1:13 | 1st Two Infusions patients under 40kg |
| 30 | F2400 | RMS12609 | 21.2 | 21.2 | 30 | 1:24 | Maintenance Infusions |
| 30 | F1200 | RMS12409 | 42.1 | 42.1 | 30 | 0:42 | Maintenance Infusions |
| Gammagard Liquid – with Freedom Edge® with 20 ml syringe | |||||||
| Drug volume (ml) | Flow Rate Tubing | HIgH-Flo Needle Set* | Total Flow Rate (ml/hr) | Flow rate/site (ml/hr) | Vol/site (ml) | Time | NOTES: |
| 20 | F45 | RMS12609 | 14.2 | 14.2 | 20 | 1:24 | Patients under 40kg (Initial) |
| 60 | F120 | RMS22609 | 39.8 | 19.9 | 30 | 1:30 | Patients over 40kg (Initial) |
| 100 | F420 | RMS42609 | 119.1 | 29.8 | 25 | 0:50 | Patients over 40kg (maintenance infusions) |
| Gammagard Liquid – with Freedom Edge® with 20 ml syringe | |||||||
| 20 | F120 | RMS22609 | 30 | 15 | 10 | 0:40 | Patients under 40kg(Initial) |
| 30 | F180 | RMS22609 | 39.8 | 19.9 | 15 | 0:45 | Patients over 40kg (Initial) |
| 30 | F120 | RMS12609 | 27 | 27 | 30 | 1:06 | Patients over 40kg (Maintenance) |
Hizentra® 20 ml Prefilled Syringe for Primary Immunodeficiency (PI) Flow Rate Combinations:
The following tables indicate the average (min-max) predicted flow rates per site with HIgH-Flo Subcutaneous Safety Needle Sets™ (26G and 24G) when used in combi- nation with KORU Precision Flow Rate Tubing™ and FreedomEdge® Syringe Infusion System with a 20 ml syringe for the subcutaneous use of Hizentra for the treatment of Primary Immunodeficiency (PI).
To determine the appropriate flow rate tubing and subcutaneous needle configura- tion, please refer to drug package insert for the maximum indicated flow rate and volume per infusion site for initial and subsequent infusions.
HIgH-Flo 26G with Precision Tubing -Average (Min-Max) Flow Rate Per Site (ml/hr/site
 | F120 | F180 | F275 | F420 | F500 | F600 | F900 | F1200 | F2400 | ||||||||||||||
| 1 needle | 9.9 (7.0 – 12.9) | 12.5 (9.7- 15.4) | 17.2 |  |  |  |  |  |  | ||||||||||||||
| (13.1 – 21.2) | |||||||||||||||||||||||
| 2 needles | 5.4 (3.8 – 7.1) | 7.0 (5.4 – 8.7) | 10.1 (7.6 – 12.6) | 14.2 | 16.1 | 18.3 |  |  |  | ||||||||||||||
| (10.1 – 18.2) | (12.6 – 19.6) | (14.0 – 22.5) | |||||||||||||||||||||
| 3 needles | 3.7 (2.6 – 4.9) | 4.9 (3.7- 6.0) | 7.2 (5.4 – 8.9) | 10.3 (7.3 – 13.3) | 11.9 (9.3 – 14.5) | 13.6 | 19.3 |  |  | ||||||||||||||
| (10.3 – 16.9) | (14.8 – 23.7) | ||||||||||||||||||||||
| 4 needles | 2.9 (2.0 – 3.7) | 3.7 (2.9 – 4.6) | 5.5 (4.2 – 6.9) | 8.1 (5.7- 10.5) | 9.4 (7.3 – 11.5) | 10.9 (8.2 – 13.5) | 15.8 | 18.2 |  | ||||||||||||||
| (12.1 – 19.5) | (13.3- 23.2) | ||||||||||||||||||||||
| 5 needles | 2.3 (1.6 – 3.0) | 3.0 (2.3 – 3.8) | 4.5 (3.4 – 5.7) | 6.7 (4.7- 8.7) | 7.8 (6.0 – 9.5) | 9.0 (6.8 – 11.2) | 13.3 | 15.6 |  | ||||||||||||||
| (10.2 – 16.5) | (11.2 – 19.9) | ||||||||||||||||||||||
| 6 needles | 1.9 (1.3 – 2.5) | 2.5 (1.9 – 3.2) | 3.8 (2.9 – 4.8) | 5.7 (3.9 – 7.4) | 6.6 (5.1 – 8.1) | 7.7 (5.8 – 9.6) | 11.6 (8.8 – 14.4) | 13.6 |  | ||||||||||||||
| (9.8 – 17.4) | |||||||||||||||||||||||
| 7 needles | 1.7 (1.1 – 2.2) | 2.2 (1.7- 2.7) | 3.3 (2.5 – 4.1) | 4.9 (3.4 – 6.4) | 5.8 (4.5 – 7.1) | 6.7 (5.1 – 8.4) | 10.2 (7.7- 12.7) | 12.1 |  | ||||||||||||||
| (8.6 – 15.5) | |||||||||||||||||||||||
| 8 needles | 1.5 (1.0 – 1.9) | 1.9 (1.5 – 2.4) | 2.9 (2.2 – 3.7) | 4.4 (3.0 – 5.7) | 5.1 (4.0 – 6.3) | 6.0 (4.5 – 7.5) | 9.1 (6.9 – 11.4) | 10.8 (7.7- 13.9) | 18.7 | ||||||||||||||
 Exceeds drug manufacturer’s maximum indicated flow rate.
Exceeds drug manufacturer’s maximum indicated flow rate. Subsequent infusions after 6th infusion only.
Subsequent infusions after 6th infusion only.
HIgH-Flo 24G with Precision Tubing-Average (Min-Max) Flow Rate Per Site (ml/hr/site)
| F120 | F180 | F275 | F420 | F500 | F600 | F900 | F1200 | F2400 | |
| 1 needle | 11.5 (8.0 – 15.1) | 15.1 (11.5 – 18.7) |  |  |  |  |  |  |  |
| 2 needles | 5.9 (4.0 – 7.7) | 7.8 (5.9 – 9.6) | 11.7 (8.7- 14.7) | 17.6 (12.2 – 23.0) |  |  |  |  |  |
| 3 needles | 4.0 (2.7- 5.2) | 5.2 (4.0 – 6.5) | 7.9 (5.9 – 10.0) | 12.0 (8.3 – 15.7) | 14.2 (11.0 – 17.4) | 16.7 (12.5 – 21.0) |  |  |  |
| 4 needles | 3.0 (2.0 – 3.9) | 3.9 (3.0 – 4.9) | 6.0 (4.5 – 7.5) | 9.1 (6.3 – 11.9) | 10.8 (8.3 – 13.2) | 12.8 (9.5 – 16.0) |  |  |  |
| 5 needles | 2.4 (1.6 – 3.1) | 3.2 (2.4 – 3.9) | 4.8 (3.6 – 6.1) | 7.3 (5.1 – 9.6) | 8.7 (6.7- 10.7) | 10.3 (7.7- 12.9) | 16.3 (12.2 – 20.4) |  |  |
| 6 needles | 2.0 (1.4 – 2.6) | 2.6 (2.0 – 3.3) | 4.0 (3.0 – 5.1) | 6.1 (4.2 – 8.1) | 7.3 (5.6 – 8.9) | 8.6 (6.4 – 10.8) | 13.8 (10.3 – 17.2) | 16.7 (11.7- 21.8) |  |
| 7 needles | 1.7 (1.2 – 2.2) | 2.3 (1.7- 2.8) | 3.5 (2.6 – 4.3) | 5.3 (3.6 – 6.9) | 6.3 (4.8 – 7.7) | 7.4 (5.5 – 9.3) | 11.9 (8.9 – 14.9) | 14.5 (10.1 – 18.9) |  |
| 8 needles | 1.5 (1.0 – 2.0) | 2.0 (1.5 – 2.5) | 3.0 (2.3 – 3.8) | 4.6 (3.2 – 6.1) | 5.5 (4.2 – 6.7) | 6.5 (4.9 – 8.2) | 10.5 (7.8 – 13.1) | 12.7 (8.9 – 16.6) |  |
 Exceeds drug manufacturer’s maximum indicated flow rate.
Exceeds drug manufacturer’s maximum indicated flow rate. Subsequent infusions after 6th infusion only
Subsequent infusions after 6th infusion only
Hizentra® 20 ml Prefilled Syringe for CIDP Flow Rate Combinations:
The following tables indicate the average (min-max) predicted flow rates per site with HIgH-Flo Subcutaneous Safety Needle Sets™ (26G and 24G) when used in combination with KORU Precision Flow Rate Tubing™ and FreedomEdge® Syringe Infusion System with a 20 ml syringe for the subcutaneous use of Hizentra for the treatment of chronic inflam- matory demyelinating polyneuropathy (CIDP).
To determine the appropriate flow rate tubing and subcutaneous needle configura- tion, please refer to drug package insert for the maximum indicated flow rate and volume per infusion site for initial and subsequent infusions.
HIgH-Flo 26G with Precision Tubing –Average (Min-Max) Flow Rate Per Site (ml/hr/site)
| F120 | F180 | F275 | F420 | F500 | F600 | F900 | F1200 | F2400 | |||||||||||||||
| 1 needle | 9.9 (7.0 – 12.9) | 12.5 (9.7- 15.4) | 17.2 | 22.7 | 25.2 | 27.7 | 34.6 | 37.3 |  | ||||||||||||||
| (13.1 – 21.2) | (16.7- 28.7) | (19.9 – 30.5) | (21.6 – 33.9) | (27.3 – 42) | (28.8 – 45.9) | ||||||||||||||||||
| 2 needles | 5.4 (3.8 – 7.1) | 7.0 (5.4 – 8.7) | 10.1 (7.6 – 12.6) | 14.2 (10.1 – 18.2) | 16.1 (12.6 – 19.6) | 18.3 | 24.8 | 27.6 | 37.8 | ||||||||||||||
| (14.0 – 22.5) | (19.2 – 30.3) | (20.7- 34.6) | (28.7- 46.9) | ||||||||||||||||||||
| 3 needles | 3.7 (2.6 – 4.9) | 4.9 (3.7- 6.0) | 7.2 (5.4 – 8.9) | 10.3 (7.3 – 13.3) | 11.9 (9.3 – 14.5) | 13.6 (10.3 – 16.9) | 19.3 | 22.0 | 32.3 | ||||||||||||||
| (14.8 – 23.7) | (16.2 – 27.7) | (24 – 40.6) | |||||||||||||||||||||
| 4 needles | 2.9 (2.0 – 3.7) | 3.7 (2.9 – 4.6) | 5.5 (4.2 – 6.9) | 8.1 (5.7- 10.5) | 9.4 (7.3 – 11.5) | 10.9 (8.2 – 13.5) | 15.8 (12.1 – 19.5) | 18.2 | 28.2 | ||||||||||||||
| (13.3 – 23.2) | (20.6- 35.8) | ||||||||||||||||||||||
| 5 needles | 2.3 (1.6 – 3) | 3.0 (2.3 – 3.8) | 4.5 (3.4 – 5.7) | 6.7 (4.7- 8.7) | 7.8 (6.0 – 9.5) | 9.0 (6.8 – 11.2) | 13.3 (10.2 – 16.5) | 15.6 (11.2 – 19.9) | 25.0 | ||||||||||||||
| (18.1 – 32) | |||||||||||||||||||||||
| 6 needles | 1.9 (1.3 – 2.5) | 2.5 (1.9 – 3.2) | 3.8 (2.9 – 4.8) | 5.7 (3.9 – 7.4) | 6.6 (5.1 – 8.1) | 7.7 (5.8 – 9.6) | 11.6 (8.8 – 14.4) | 13.6 (9.8 – 17.4) | 22.5 | ||||||||||||||
| (16.1 – 28.9) | |||||||||||||||||||||||
| 7 needles | 1.7 (1.1 – 2.2) | 2.2 (1.7- 2.7) | 3.3 (2.5 – 4.1) | 4.9 (3.4 – 6.4) | 5.8 (4.5 – 7.1) | 6.7 (5.1 – 8.4) | 10.2 (7.7- 12.7) | 12.1 (8.6 – 15.5) | 20.4 | ||||||||||||||
| (14.5 – 26.4) | |||||||||||||||||||||||
| 8 needles | 1.5 (1 – 1.9) | 1.9 (1.5 – 2.4) | 2.9 (2.2 – 3.7) | 4.4 (3.0 – 5.7) | 5.1 (4.0 – 6.3) | 6.0 (4.5 – 7.5) | 9.1 (6.9 – 11.4) | 10.8 (7.7- 13.9) | 18.7 | ||||||||||||||
| (13.2 – 24.2) | |||||||||||||||||||||||
 Exceeds drug manufacturer’s maximum indicated flow rate.
Exceeds drug manufacturer’s maximum indicated flow rate. Subsequent infusions after 6th infusion only
Subsequent infusions after 6th infusion only
HIgH-Flo 24G with Precision Tubing –Average (Min-Max) Flow Rate Per Site (ml/hr/site
| F120 | F180 | F275 | F420 | F500 | F600 | F900 | F1200 | F2400 | |||||||||||||||
| 1 needle | 11.5 (8.0 – 15.1) | 15.1 (11.5 – 18.7) | 22.5 | 33.1 | 38.5 |  |  |  |  | ||||||||||||||
| (16.8 – 28.2) | (23.2 – 43) | (29.9 – 47.1) | |||||||||||||||||||||
| 2 needles | 5.9 (4.0 – 7.7) | 7.8 (5.9 – 9.6) | 11.7 (8.7- 14.7) | 17.6 | 20.7 | 24.4 | 37.5 |  |  | ||||||||||||||
| (12.2 – 23.0) | (16.0 – 25.4) | (18.2 – 30.5) | (28.3- 46.7) | ||||||||||||||||||||
| 3 needles | 4.0 (2.7- 5.2) | 5.2 (4.0 – 6.5) | 7.9 (5.9 – 10.0) | 12.0 (8.3 – 15.7) | 14.2 (11.0 – 17.4) | 16.7 | 26.2 | 31.5 |  | ||||||||||||||
| (12.5 – 21.0) | (19.7- 32.7) | (22.2 – 40.9) | |||||||||||||||||||||
| 4 needles | 3.0 (2.0 – 3.9) | 3.9 (3.0 – 4.9) | 6 (4.5 – 7.5) | 9.1 (6.3 – 11.9) | 10.8 (8.3 – 13.2) | 12.8 (9.5 – 16.0) | 20.1 | 24.3 |  | ||||||||||||||
| (15.1 – 25.2) | (17.0 – 31.7) | ||||||||||||||||||||||
| 5 needles | 2.4 (1.6 – 3.1) | 3.2 (2.4 – 3.9) | 4.8 (3.6 – 6.1) | 7.3 (5.1 – 9.6) | 8.7 (6.7- 10.7) | 10.3 (7.7- 12.9) | 16.3 | 19.8 |  | ||||||||||||||
| (12.2 – 20.4) | (13.8 – 25.8) | ||||||||||||||||||||||
| 6 needles | 2.0 (1.4 – 2.6) | 2.6 (2 – 3.3) | 4.0 (3.0 – 5.1) | 6.1 (4.2 – 8.1) | 7.3 (5.6 – 8.9) | 8.6 (6.4 – 10.8) | 13.8 (10.3 – 17.2) | 16.7 | 32.7 | ||||||||||||||
| (11.7- 21.8) | (22.0 – 43.4) | ||||||||||||||||||||||
| 7 needles | 1.7 (1.2 – 2.2) | 2.3 (1.7- 2.8) | 3.5 (2.6 – 4.3) | 5.3 (3.6 – 6.9) | 6.3 (4.8 – 7.7) | 7.4 (5.5 – 9.3) | 11.9 (8.9 – 14.9) | 14.5 (10.1 – 18.9) | 28.6 | ||||||||||||||
| (19.2 – 38.0) | |||||||||||||||||||||||
| 8 needles | 1.5 (1.0 – 2.0) | 2.0 (1.5 – 2.5) | 3.0 (2.3 – 3.8) | 4.6 (3.2 – 6.1) | 5.5 (4.2 – 6.7) | 6.5 (4.9 – 8.2) | 10.5 (7.8 – 13.1) | 12.7 (8.9 – 16.6) | 25.3 | ||||||||||||||
| (17.0 – 33.7) | |||||||||||||||||||||||
 Exceeds drug manufacturer’s maximum indicated flow rate.
Exceeds drug manufacturer’s maximum indicated flow rate. Subsequent infusions after 6th infusion only
Subsequent infusions after 6th infusion only
Warranty Information
This warranty and the rights and obligations hereunder, shall be construed under
and governed by the laws of the State of New York, USA.
Limited Warranty: KORU Medical Systems (“Manufacturer”) warrants the syringe driver to be free from defects in materials and workmanship under normal use. Warranty is limited to Original Purchaser and covers the FreedomEdge® for a period of two years from the purchase date. This warranty is not valid for any damage caused by the use of non-KORU products. The “Original Purchaser” is the person purchasing the syringe driver from the Manufacturer or Manufacturer’s Representative. Warranty does not extend to subsequent purchasers. Subject to the conditions of and upon compliance with the procedures set forth in this limited warranty, the Manufacturer will repair or replace, at its option, any syringe driver, or part thereof, which has been actually received by the Manufacturer or Manufacturer’s Representative within the two-year warranty period, and which examination discloses, to the Manufacturer’s satisfaction, that the product is defective. Replacement product and parts are warranted only for the remaining portion of the original two-year warranty period.
KORU tests the FreedomEdge using KORU accessories to ensure that the FreedomEdge operates in accordance with published specification standards. If non-KORU accessories are used in conjunction with the FreedomEdge, KORU does not represent that the FreedomEdge will operate in accordance with published specification standards. The FreedomEdge warranty does not cover third-party products or accessories.
The following conditions, procedures, and limitations apply to the Manufacturer’s obligations under this warranty
- Parties Covered by this Warranty: This warranty extends only to the Original Purchaser of the infusion syringe driver. This warranty does not extend to subsequent purchasers.
- Warranty Performance Procedure: Notice of the defect must be made in writing to Customer Support Department, KORU Medical Systems, 24 Carpenter Road, Chester, NY 10918, USA. Notice to KORU Medical Systems must include the model and serial number, date of purchase, and description of the defect in sufficient detail to facilitate repairs. Authorization must be obtained by the Original Purchaser from the Manufacturer or Manufacturer’s Representative prior to returning the product to the Manufacturer. The defective syringe driver must be properly packaged and returned to the Manufacturer, postageprepaid. Any loss or damage during shipment is at the risk of the Original Purchaser.
- Conditions of Warranty: This warranty does not apply to any product, or part thereof, which has been repaired or altered outside of the Manufacturer’s facility in a way so as, in Manufacturer’s judgment, to affect its stability or reliability, or which has been subjected to misuse, negligence or accident.
- Limitations and Exclusions: Repair or replacement of a syringe driver or component part is the EXCLUSIVE remedy offered by the Manufacturer. The following exclusions and limitations shall apply: • No agent, representative, or employee of the Manufacturer has authority to bind the Manufacturer to any representation or warranty, expressed or implied, or to change this limited warranty in any way.
- THIS LIMITED WARRANTY IS IN LIEU OF ALL OTHER WARRANTIES, EXPRESS OR IMPLIED, INCLUDING, BUT NOT LIMITED TO, WARRANTIES OF MERCHANTABILITY AND FITNESS FOR A PARTICULAR PURPOSE. THERE ARE NO WARRANTIES THAT EXTEND BEYOND THE DESCRIPTION ON THE FACE HEREOF.
- Manufacturer’s liability under this Limited Warranty Agreement shall not extend to special, indirect, or consequential damages.
- The syringe driver can only be used under the supervision of medical personnel whose skill and judgment determine the suitability of the syringe driver for a particular medical treatment.
- All recommendations, information, and descriptive literature supplied by the Manufacturer or its agents are believed to be accurate and reliable, but do not constitute warranties.
Definition of Symbols
 | Caution |
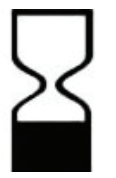
| Use by YYYY-MM-DD or YYYY-MM |
 | Consult Instructions For Use |
| Manufacturer |
| Authorized Representative in the European Community |  | Do Not Reuse |
| Batch Code |
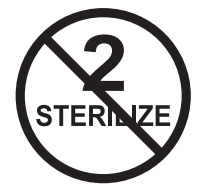
| Do Not Resterilize |
| Quantity |  | Not Made with Natural Rubber Latex |
| Catalog Number |  | Do Not Use if Package is Damaged |
| Serial Number |
| MR Unsafe |
| Sterilized Using Irradiation |
| Prescription Only |
| Medical Device |
| European Conformity |
CUSTOMER SUPPORT
+1 845•469•2042 | korumedical.com | @korumedical
 Manufacturer
Manufacturer
KORU Medical Systems
24 Carpenter Road
Chester, NY 10918 USA
+1 (845) 469 2042

European Representative
ICON (LR) Limited
South County Business Park,
Leopardstown, Dublin 18,
D18 X5R3, Ireland
+353 1 291 2000
The FreedomEdge® Syringe Infusion System, Precision Flow Rate Tubing™, HIgH-Flo Subcutaneous Safety Needle Sets™, and DynEQ® are trademarks of KORU Medical Systems and are compliant with Medical Device Directive 93/42/EEC. All other trademarks are the property of their respective owners. KORU Medical Systems is ISO 13485 certified. ©2022 KORU Medical Systems; All Rights Reserved.