Spravato Esketamine Nasal Spray Device

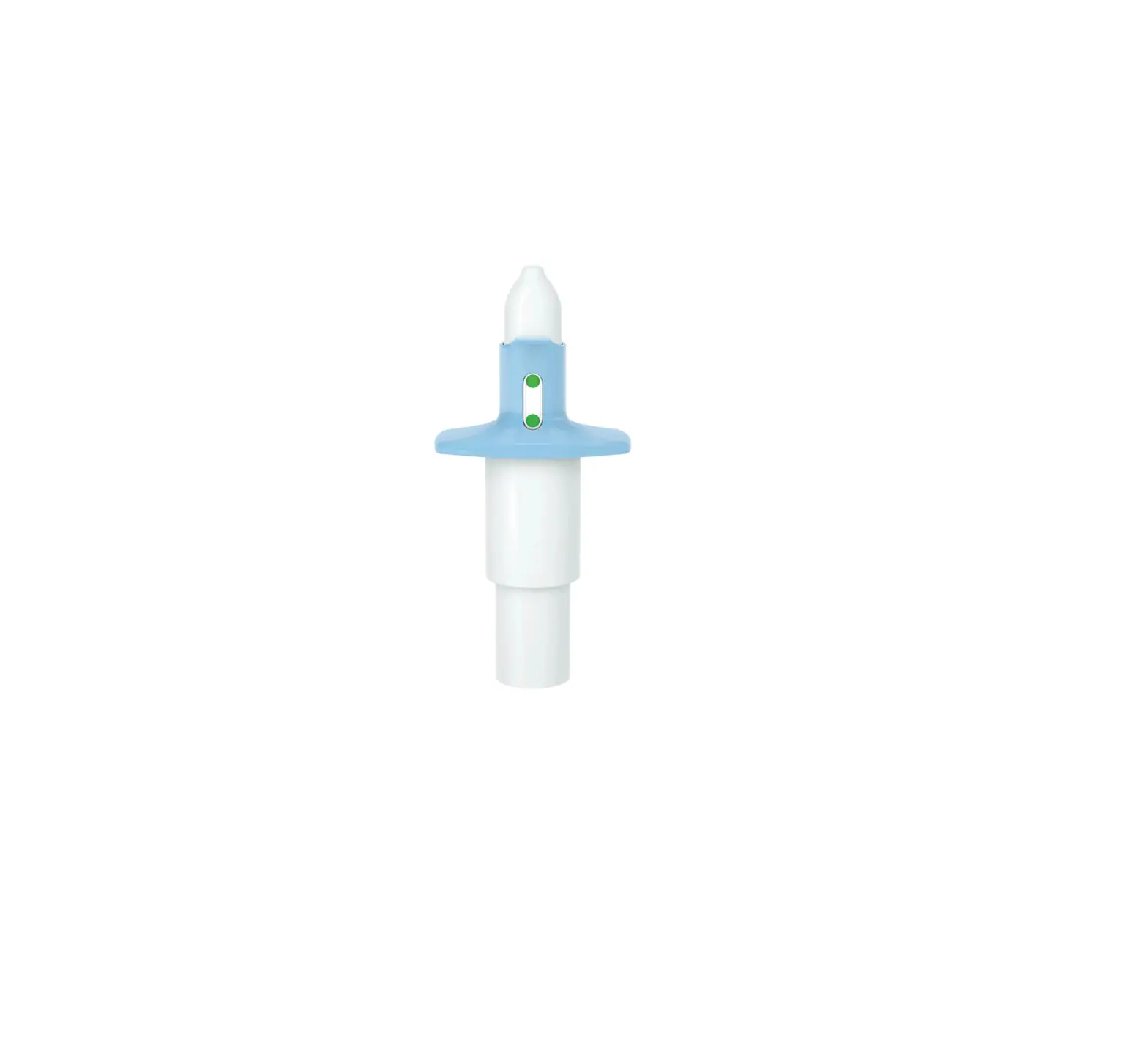
Nasal Spray Device
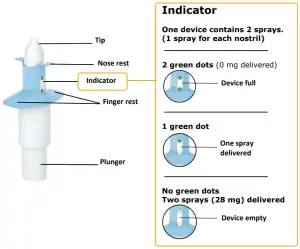
Each device delivers two sprays containing a total of 28 mg of esketamine.
Step 1 Get ready
Before first device only:
Instruct patient to blow nose before first device only.
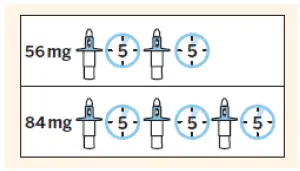
Confirm required number of devices.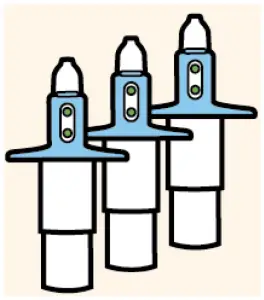
| 56 mg = 2 devices |
| 84 mg = 3 devices |
Step 2 Prepare device
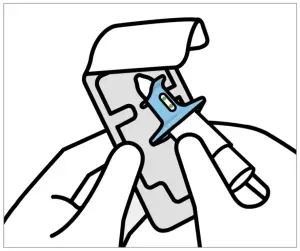
Healthcare professional:
- Check expiration date (`EXP’). If expired, get a new device.
- Peel blister and remove device.

Healthcare professional: - Do not prime device. This will result in a loss of medication.
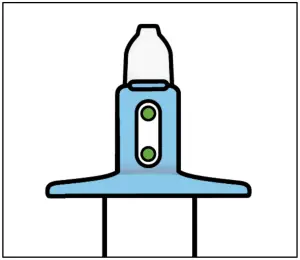
- Check that indicator shows 2 green dots. If not, dispose of device and get a new one
- Hand device to patient. cp-81317v2
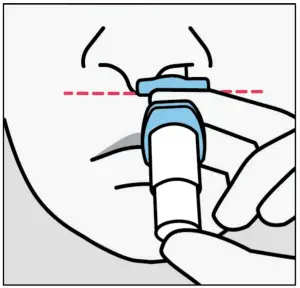
Step 3 Prepare patient
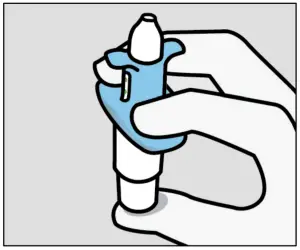
Instruct the patient to:
- Hold device as shown with the thumb gently supporting the plunger.
- Do not press the plunger.

Instruct the patient to: - Recline head at about
45 degrees during administration to keep medication inside the nose.
Step 4 Patient sprays once into each nostril
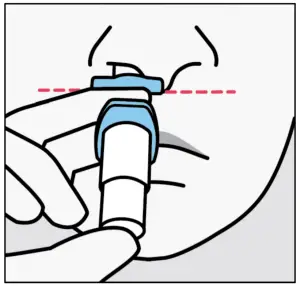
Instruct the patient to:
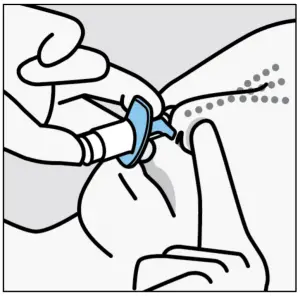
- Insert tip straight into the first nostril.
- Nose rest should touch the skin between the nostrils.

Instruct the patient to: - Close opposite nostril.
- Breathe in through nose whilepushing plunger all the way up until it stops.

Instruct the patient to: - Sniff gently after spraying to keep medication inside nose.

Instruct the patient to: - Switch hands to insert tip into the second nostril.
- Repeat Step 4 to deliver second spray.
Step 5 Confirm delivery and rest
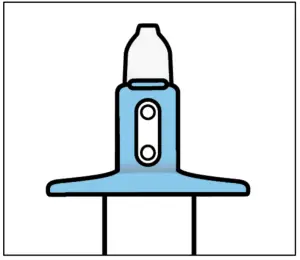
Healthcare professional:
- Take device from patient.
- Check that indicator shows no green dots. If you see a green dot, have patient spray again into the second nostril.
- Check indicator again to confirm device is empty.

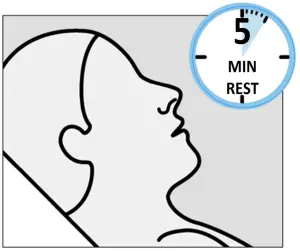
Instruct the patient to: - Rest in a comfortable position (preferably, semi -reclined) for 5 minutes
after each device. - If liquid drips out, dab nose with a tissue.
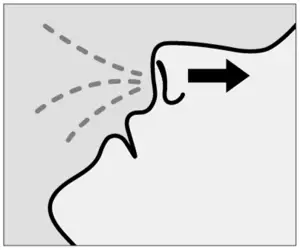
![]() Do not blow nose.
Do not blow nose.
Next device
Healthcare professional:
- Repeat Steps 2-5 for the next device.
IMPORTANT: Ensure that patient waits 5 minutes after each device to allow medication to absorb.
Disposal
Dispose of used device(s) per facility procedure for a Schedule III drug product and per applicable federal, state, and local regulations.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured for:
Janssen Pharmaceuticals, Inc. Titusville, NJ 08560
Revised: November 2019
© 2019 Janssen Pharmaceutical Companies