Instruction Manual
Instruction Manual
EU-24052-HPMSB Pressure Injectable Two-Lumen PICC

Pressure Injectable Two-Lumen PICC
Contents:
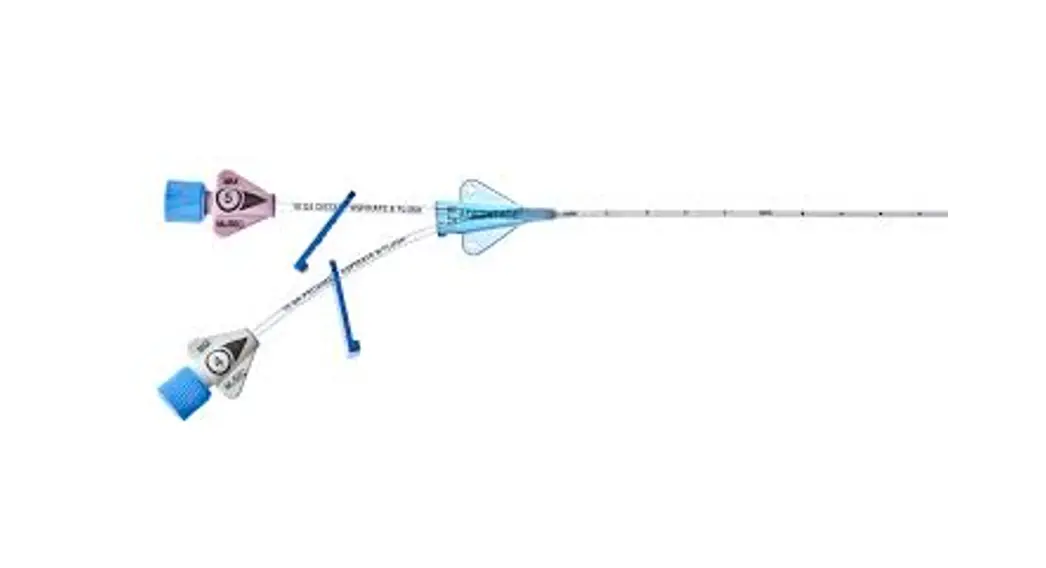
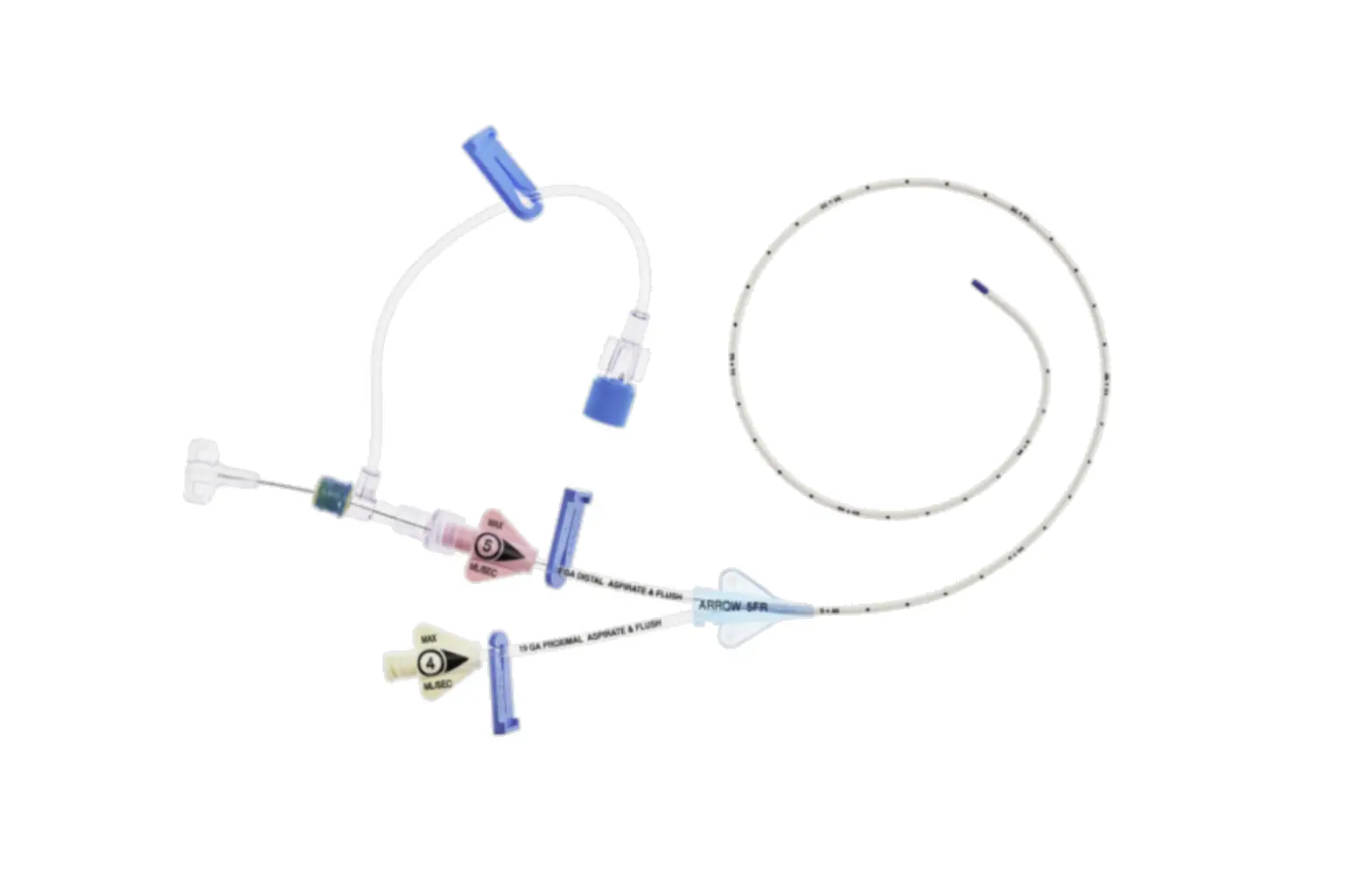
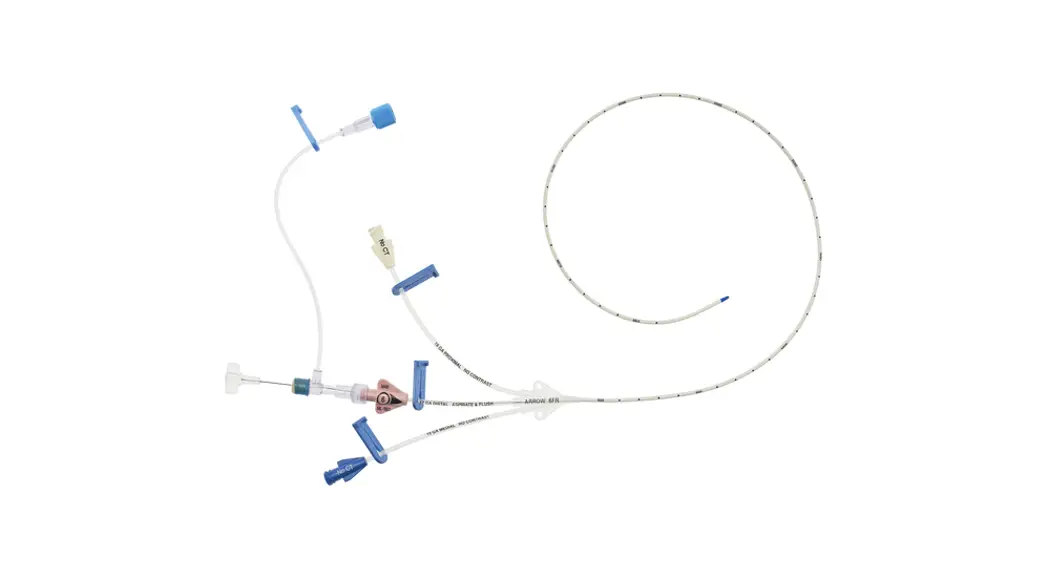
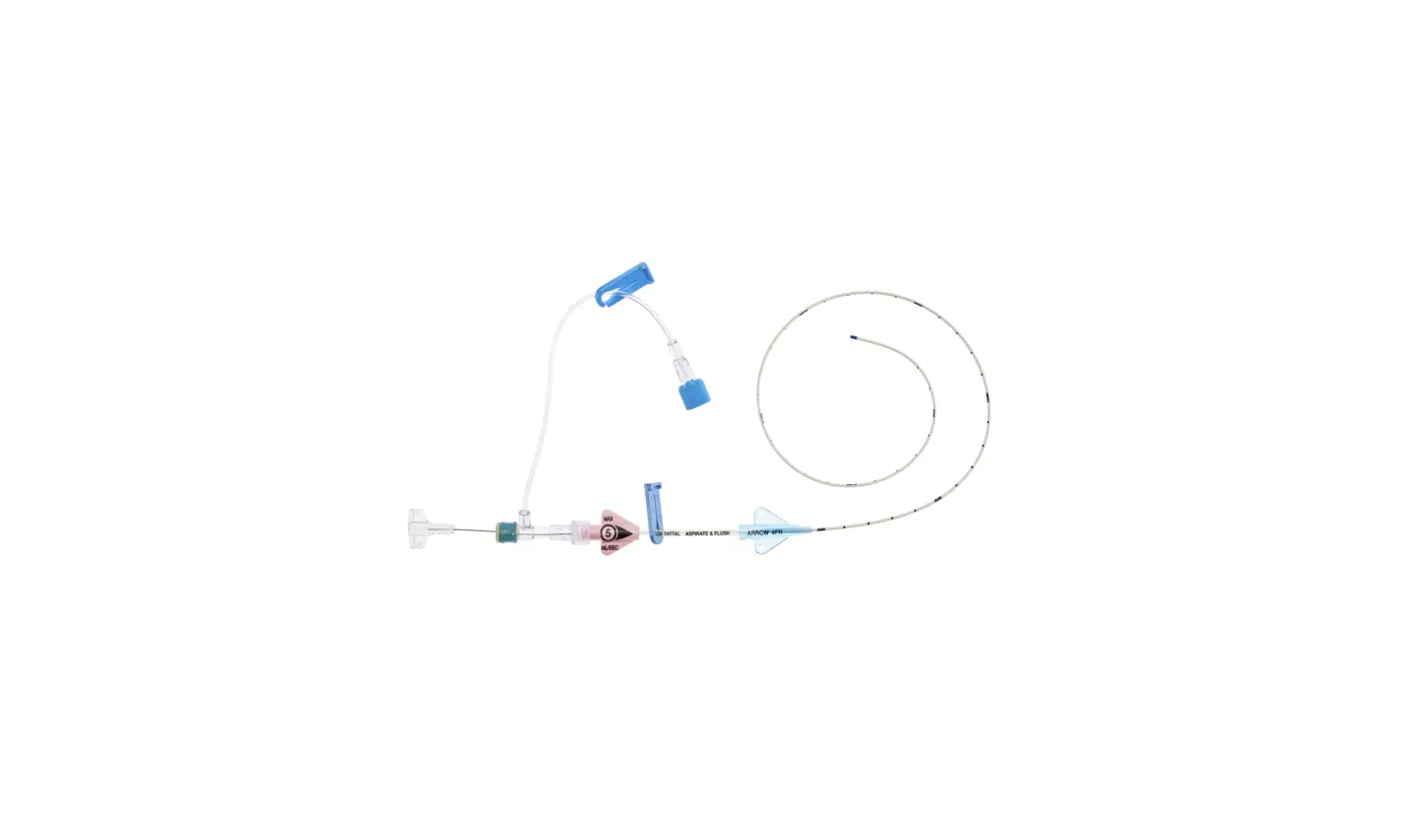
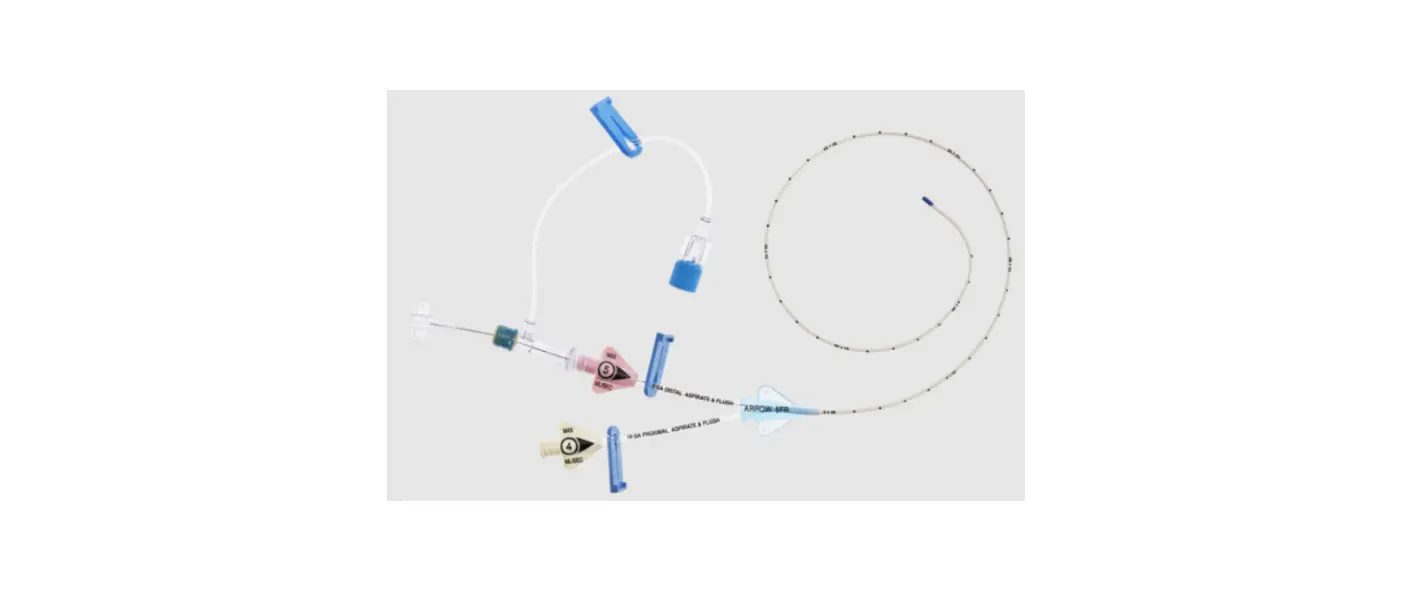
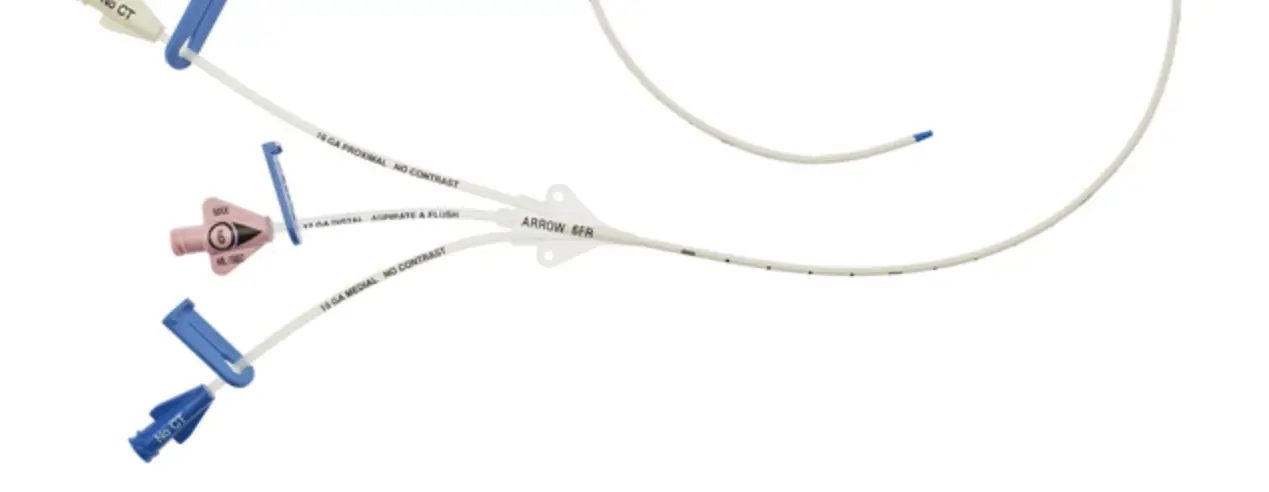
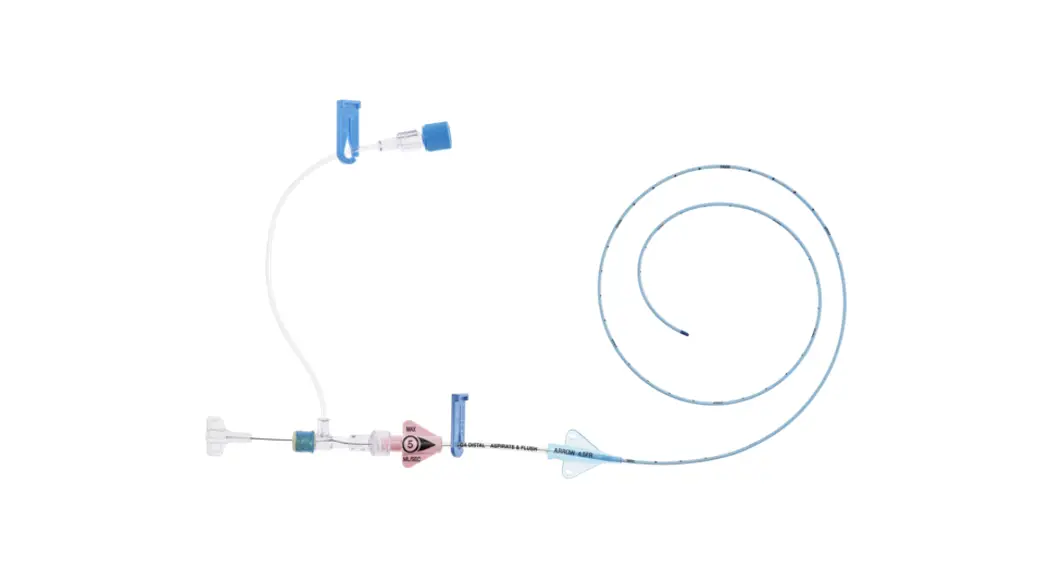
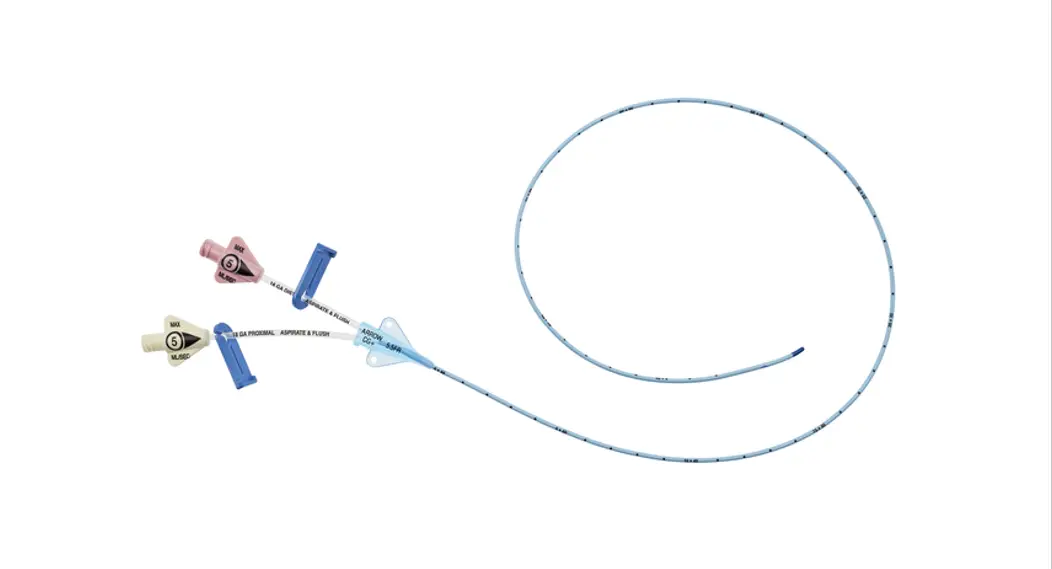
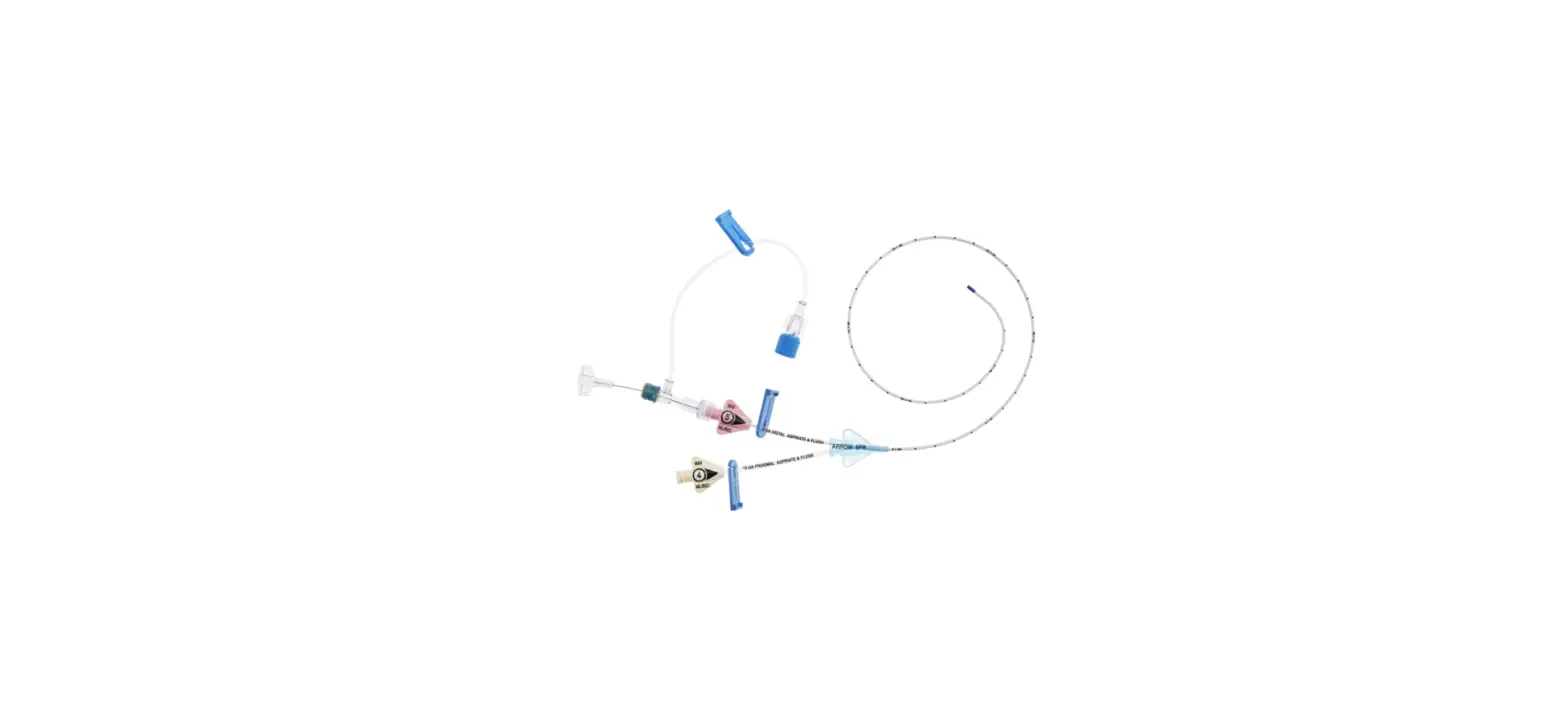
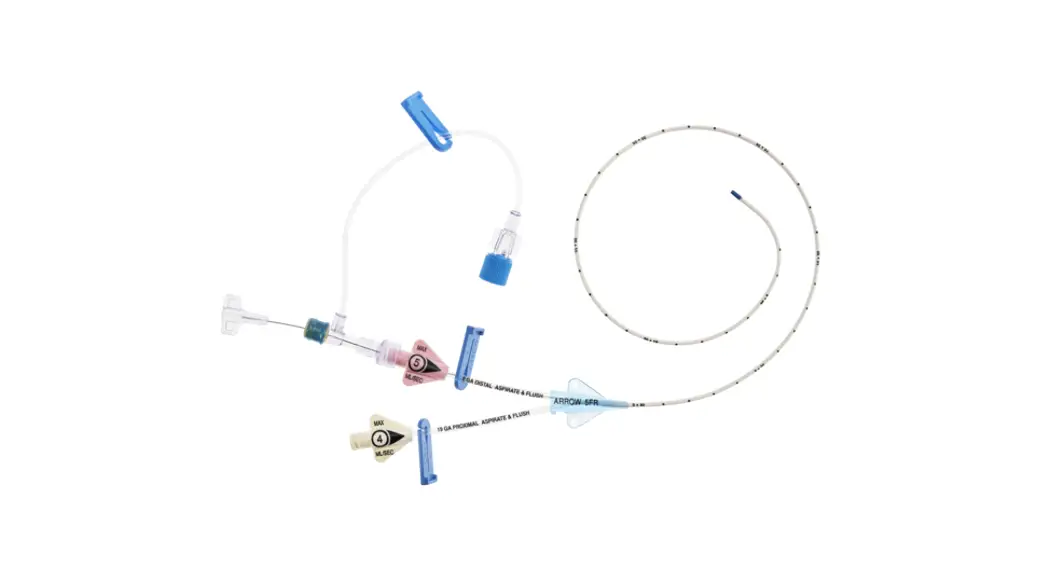
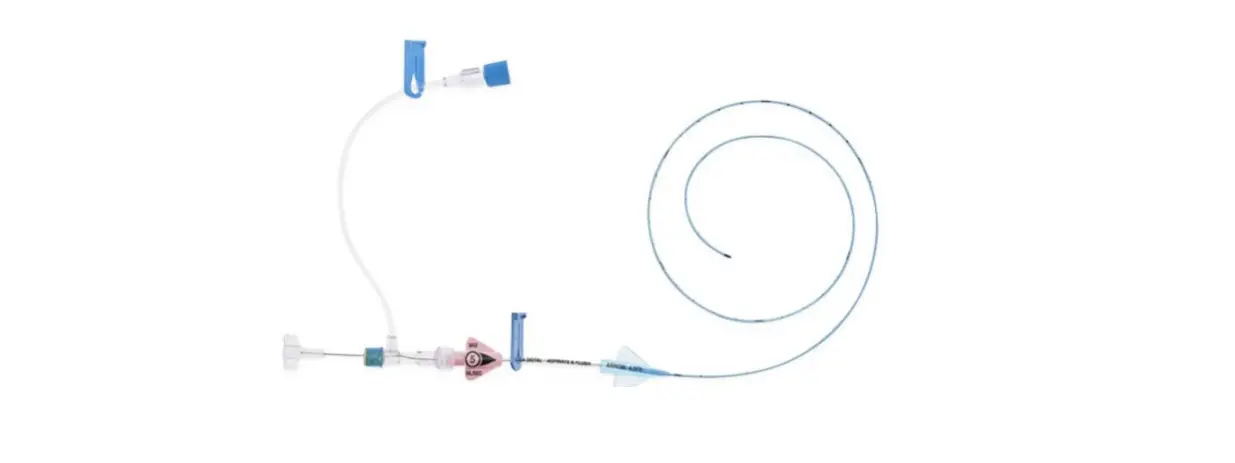
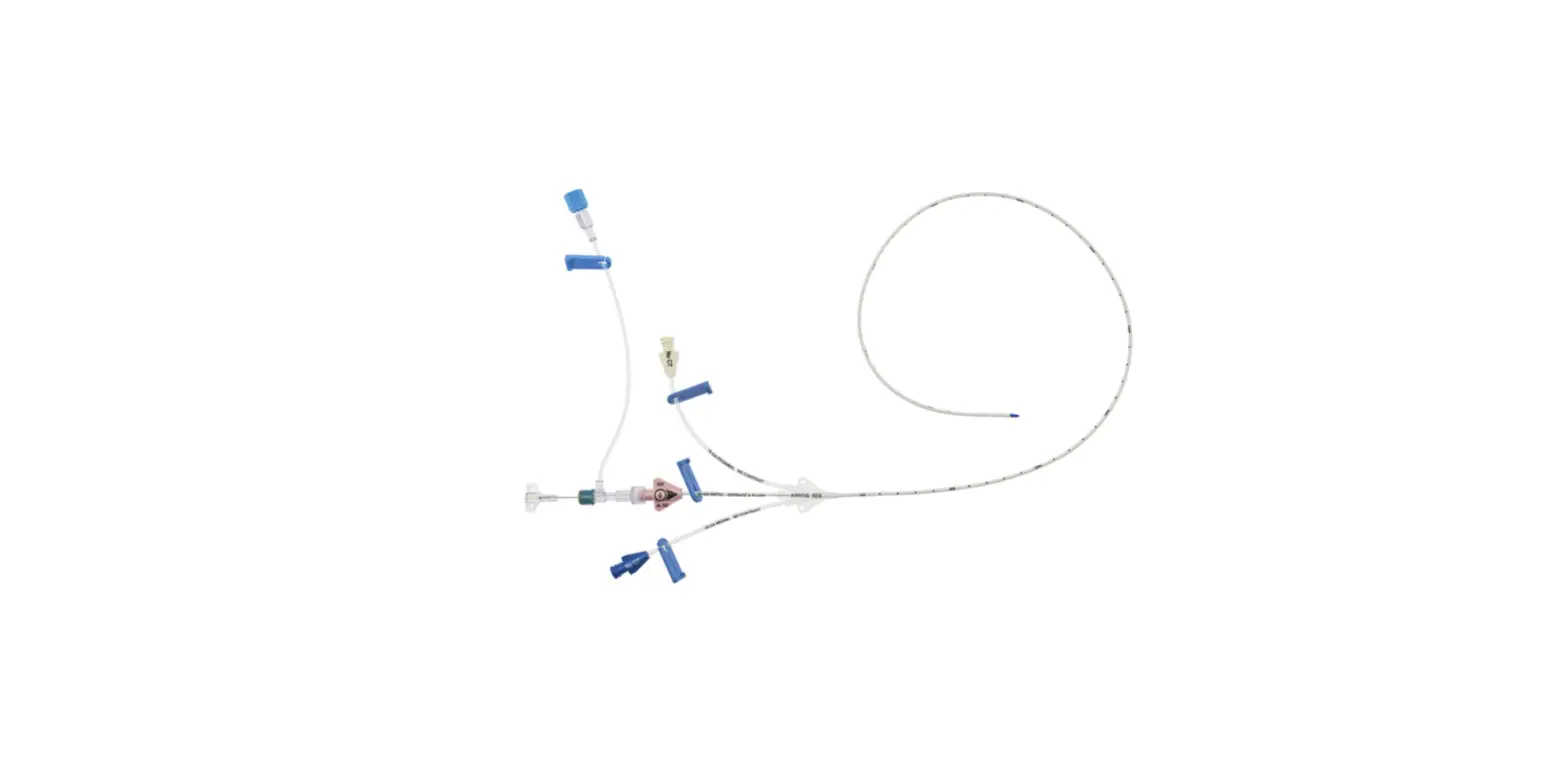
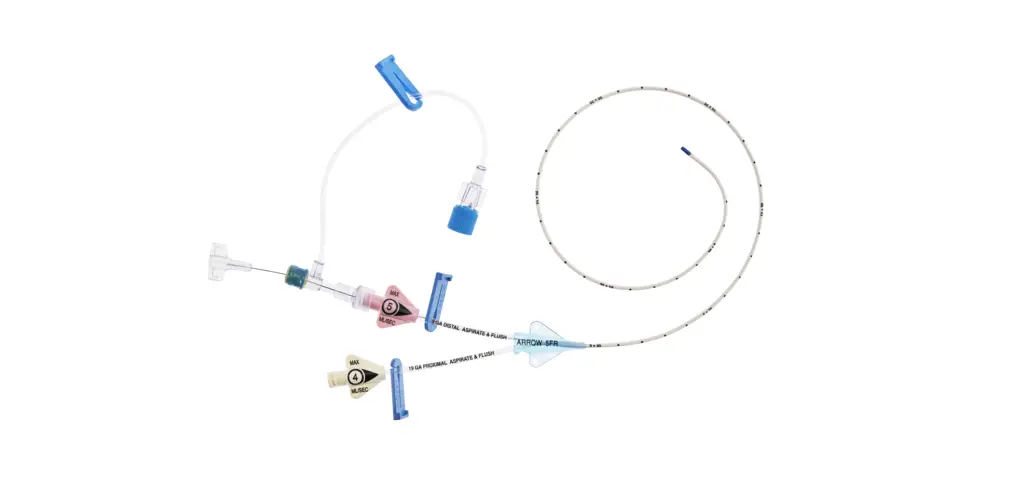
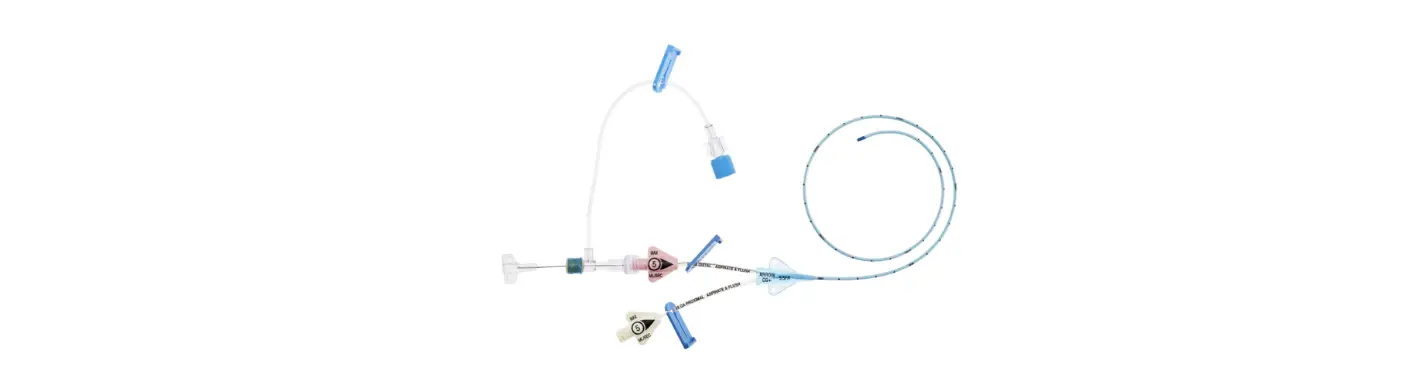
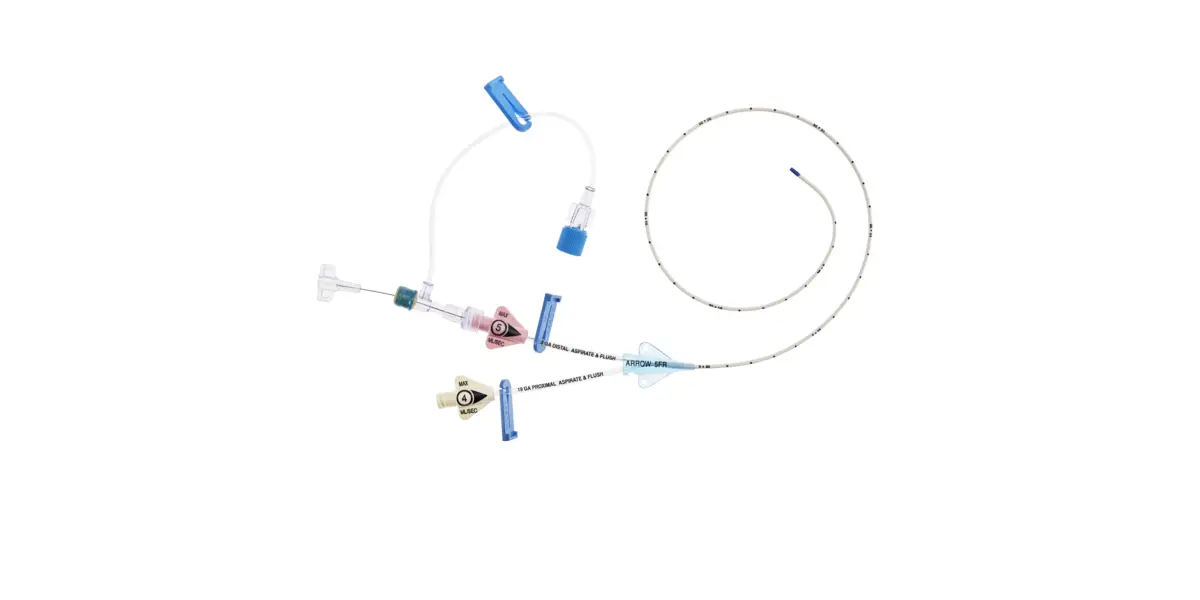
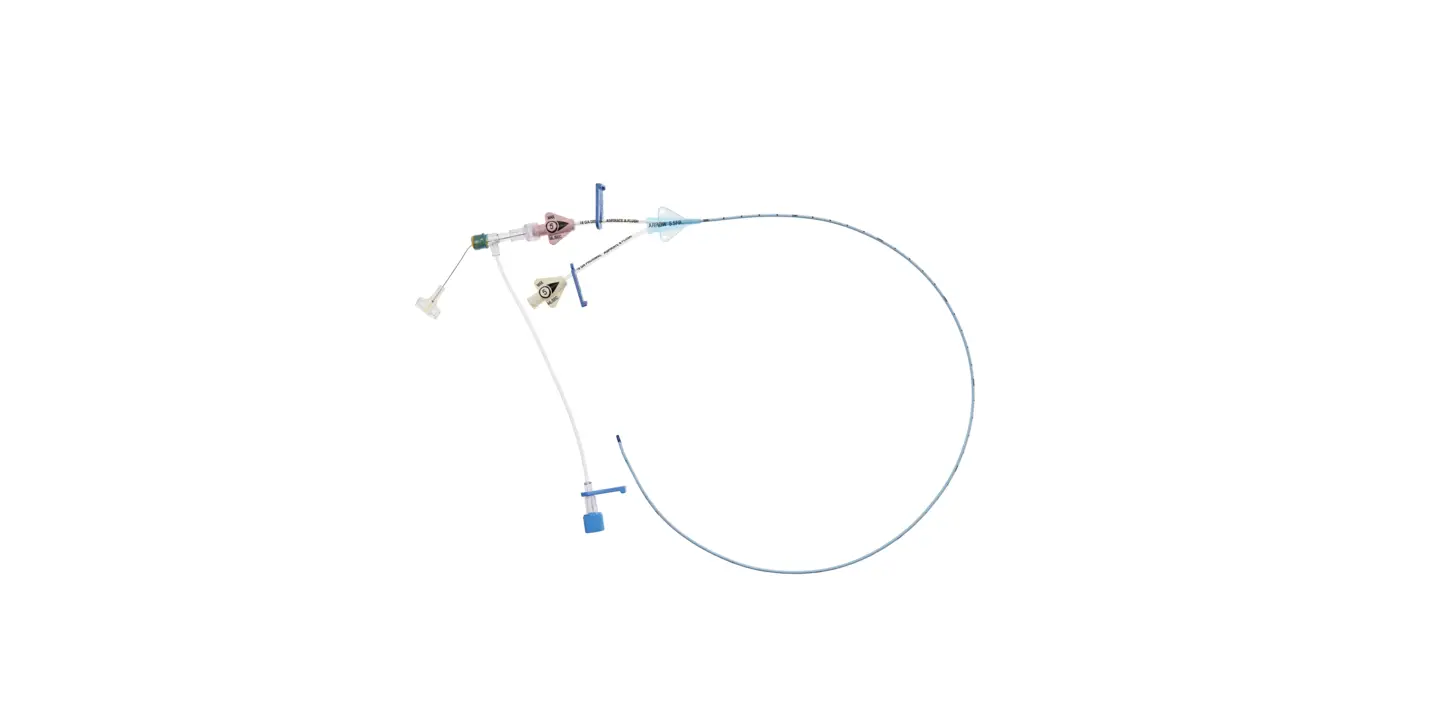
1:TwoLumenCatheter:5Fr.(1.81mmOD)x40cm,PressureInjectable,T-PortConnector,BlueFlexTip®,ContaminationGuardandPlacementWire
1:GlideThru™Peel-AwaySheath:5Fr.x2-3/4″(7cm)Radiopaqueover5Fr.Dilator
1:Spring-WireGuide,Nitinol,Marked:.018″(0.46mm)dia.x17-3/4″(45cm)(StraightSoftTiponOneEnd-StraightStiffTiponOther)withArrowAdvancer
1:InjectionNeedle:Eclipse™122Ga.x1-5/8″(4cm)
1:InjectionNeedle:Eclipse™125Ga.x1″(2.54cm)RW
1:IntroducerNeedle:Echogenic21Ga.x2-3/4″(7cm)TW
1:FilterNeedle:18Ga.TWx1-1/2″(3.81cm)withBluntFillTip2:Syringe:10mLLuer-Lock
1:Syringe:3mLLuer-Lock
1:SecondSite™AdjustableHub:Fastener
1:SecondSite™AdjustableHub:CatheterClamp
1:SharpsAway®DisposalCup
1:SharpsAway®IILockingDisposalCup
1:CatheterTrimmer
1:Towel:24″x36″(61cmx91cm)
1:Drape:60″x76″(152cmx193cm)1:Drape:36″x41″(91cmx104cm)with3″x5″(8cmx13cm)fenestration
1:Towel
1:SafetyScalpel:#111:PatientIDCard1:SterileProcedureSign
1:PatientInformationBooklet1:ChartSticker
1:PICCLineInsertionChecklist
1:PaperTapeMeasure
1:Tourniquet5:GauzePad:2″x2″(5cmx5cm)
1:Dressing:Tegaderm®2I.V.4″x6″(10cmx15.5cm)
1:Dressing:GRIP-LOK®3CatheterFixationDevice10:GauzePad:4″x4″(10cmx10cm)
1:SurgicalApparel:Cap1:SurgicalApparel: ImperviousGown
1:Dressing:Steri-Strip®21/2″x4″(1.2cmx10cm)1:SurgicalApparel:Mask
1:SurgicalApparel: MaskwithShield
1:TransducerCover:4-12/16″x95″(12cmx244cm)2:Valve:MicroCLAVE®4NeutralDisplacementConnector
1:HemoHopper®FluidReceptacle1AtrademarkofBecton,DickinsonandCompany.
2Aregisteredtrademarkof3MCompany.
3AregisteredtrademarkofTIDI.
4AregisteredtrademarkofICUMedical,Inc.
AllcomponentsareCE2797unlessotherwisenoted.
Warning: Read all package insert warnings, precautions, and instructions prior to use. Failure to do so may result in severe patient injury or death. www.teleflex.com/IFU
Not made with natural rubber latex.
Fluid path components are non-pyrogenic.
Contraindications: The Pressure Injectable PICC is contraindicated wherever there is the presence of device-related infections or the presence of thrombosis in the intended insertion vessel or catheter pathway. A clinical assessment of the patient must be completed to ensure no contraindications exist.
| Priming Volume* (mL) | Gravity Flow Rate† (mL/hr) | Pump Flow Rate†† (mL/hr) | MAX Pressure Injection Flow Rate** (mL/sec) | |
| Distal ( 18 G a .) Proximal ( 18 G a.) | 0.36 0.38 | 416 475 | 3500 4090 | 4 4 |
* Priming volumes are approximate and are done without accessories.
† Flow rate values are approximate and are determined using deionized water at 100 cm head height.
††Pump flow rates are determined at a maximum pump pressure of 10 PSIG and represent approximate flow capabilities.
** Pressure injection flow rates are determined at the injector pressure setting of 300 psi maximum using media of 11.8 centipoise viscosity, with 152 cm pressure tubing.
![]()
![]() EU Authorized
EU Authorized
Representative and
Importer:![]() Teleflex Medical
Teleflex Medical
IDA Business and![]() 2797Technology Park
2797Technology Park
Dublin Road, Athlone
Co. Westmeath, Ireland![]() Arrow International LLC
Arrow International LLC
A subsidiary of Teleflex Incorporated
3015 Carrington Mill Blvd.
Morrisville, NC 27560 USA
www.teleflex.com
LBL056501 R 00 (2021-06)