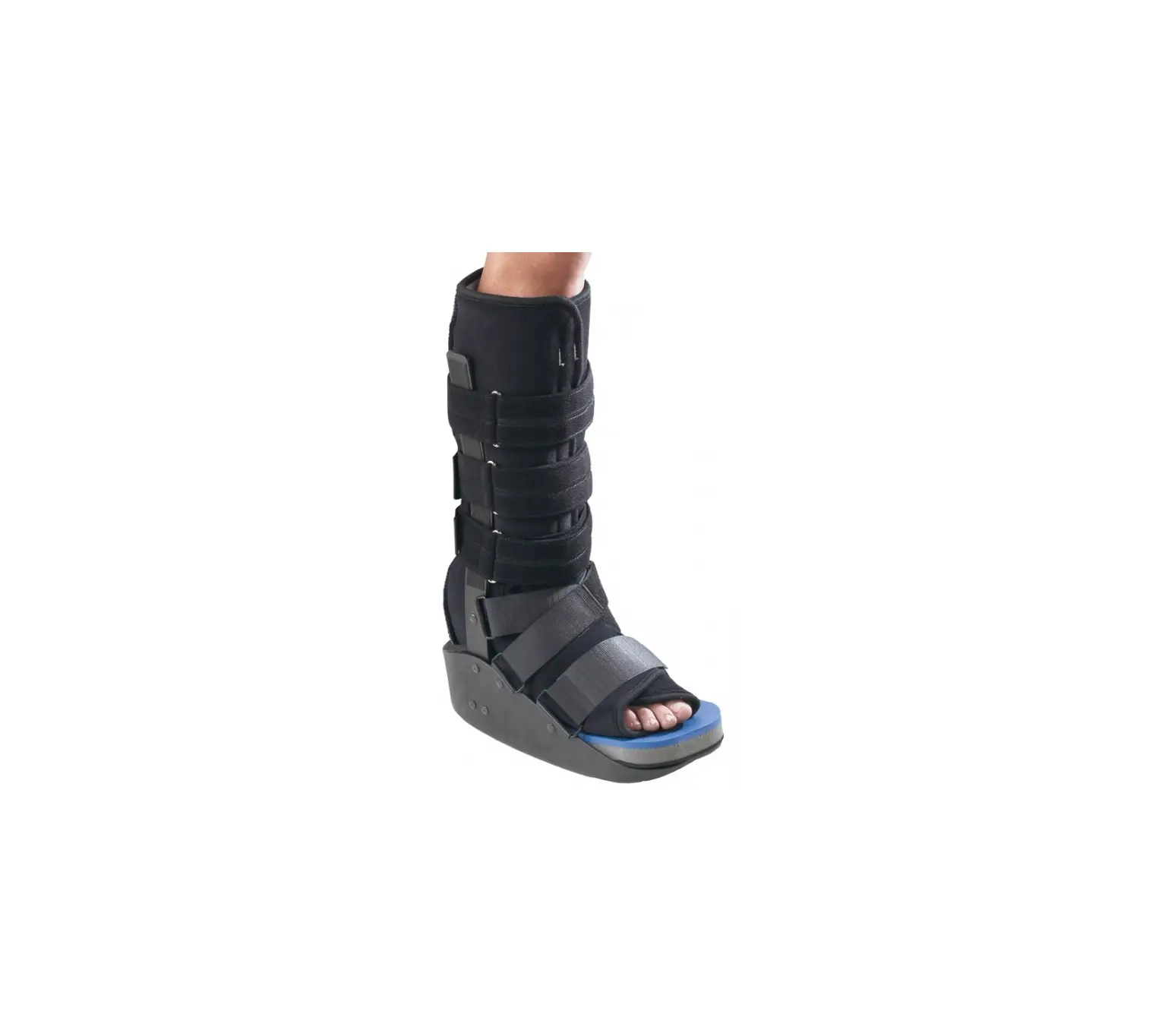
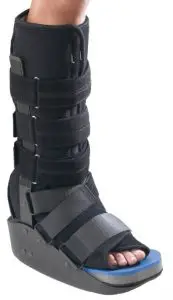
DJO ProCare Maxtrax Diabetic Walker
BEFORE USING THE DEVICE, PLEASE READ THE FOLLOWING INSTRUCTIONS COMPLETELY AND CAREFULLY. CORRECT APPLICATION IS VITAL TO THE PROPER FUNCTIONING OF THE DEVICE.
INTENDED USER PROFILE
The intended user should be a licensed medical professional, the patient or the patient’s caregiver. The user should be able to read, understand and be physically capable of performing the directions, warnings and cautions in the information for use.
INTENDED USE/INDICATIONS
The Procare MaxTrax Diabetic Walker is designed to provide support and immobilization to the foot. It may be suitable for the treatment of ulcerative or pre-ulcerative conditions of the plantar surface of the foot and moderate to severe Plantar Fasciitis. Providing immobilization or controlled movement of the limb or body segment.
CONTRAINDICATIONS
Pregnancy and Circulation, Pulmonary, Cardiovascular or Skeletal conditions which have risk to be made worse as a result of compression and/or pressure. If bony foot deformities exist, such as non-operative Charcot or other abnormalities, that cause the shape of the foot to deviate excessively from its natural shape. Foot pathology requiring support of the bony structures.
WARNINGS AND PRECAUTIONS
- This device is to be used under the supervision of a healthcare professional. The determination of when to apply the brace and the frequency and duration of use should be strictly at the discretion of the treating physician. Always consult with your physician before making changes to the brace.
- Not intended for patients in excess of 300 lbs.
- Never allow the bare foot to directly contact the innersole.
- Wear a sock to allow the foot to breathe.
- Patient, family member or medical personnel should inspect your feet before and after each use.
- If you develop any red inflamed areas, ulcers, sores or draining areas on your foot, contact your physician immediately.
- Wounds located at the beginning or the end of the gait path should be monitored closely to ensure that pressure relief is maintained.
- The insole should NOT be used in conjunction with any other walker other than the DJO MaxTrax Diabetic platform walker.
- Only remove the walker as instructed by your physician.
- Do not use over open wounds.
- Do not over tighten straps. This may result in reduction of blood flow or sensation.
- Do not modify the device or use the device other than intended.
- Do not use this device if it is damaged and/or the packaging has been opened.
- Use caution when walking on slippery or wet surfaces to avoid injury.
- Do not use this device on patient’s incapable of communicating physical discomfort.
- If pain, swelling, changes in sensation or other unusual reactions occur while using this product, consult your medical professional immediately
- If you develop an allergic reaction and/or experience itchy, red skin after coming into contact with any part of this device, please stop using it and contact your healthcare professional immediately.
NOTE: Contact manufacturer and competent authority in case of a serious incident arising due to usage of this device.
APPLICATION INFORMATION
PREPARATION
A) Be sure to treat and dress the plantar ulcer/s appropriately.
B) Wear a sock over the foot and dressing/s.
WALKER BOOT APPLICATION
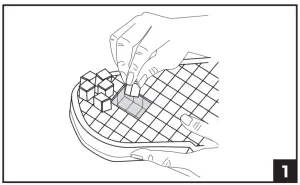
- Remove the blue foam pad from the walker boot. Place the blue side against the patient’s plantar surface, locate and remove the pre-cut foam cubes from the bottom of the blue foam pad around the wound site/s. Replace the blue foam pad back into its original location under the walker liner. (fig 1)

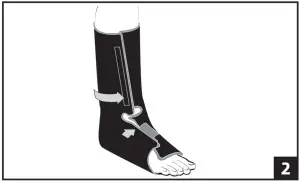
- Remove the walker liner from the walker boot. Place the foot into the liner and ensuring that the heel fits snugly into the posterior portion of the liner. Secure the forefoot flap first followed by the tibial closure. Ensure that the liner fits snug top to bottom and that there are no sharp edges or creases against the skin. a) Two optional upright pads are included with each walker to provide additional padding in area of metal uprights. Once liner is applied to lower leg, place one pad on either side of the liner with hook portion of the pad against the liner. Make sure the pads are aligned with midline of leg. Top of pads should be aligned with top of liner. (fig 2)

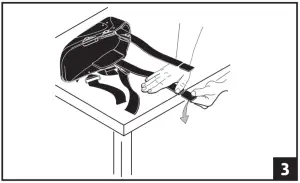
- Both aluminum uprights on the walker can be shortened by 2″ to accommodate various patients heights. a) To breakdown Uprights: Secure uprights on edge of table or hard surface. Firmly push down on breakdown portion of bar. Soft Velcro® strips are included with each brace. Liner may be folded over uprights if necessary for added comfort and fit. (fig 3)

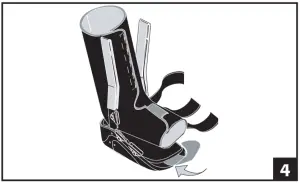
- A Toe Wrap is included with each Walker. Attach the Toe Wrap by lifting the end of the innersole and laying the Toe Wrap on the hook and loop strip. Replace the inner sole to it’s original position. (fig 4)

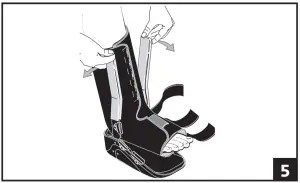
- Spread the uprights using both hands and step into boot, aligning uprights with midline of the ankle. Remove plastic sheaths on uprights and secure liner. The uprights may be bent to accommodate any leg contour. (fig 5)

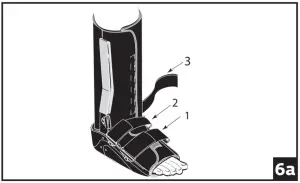
- The two straps at the ankle joint may be adapted to accommodate individual patient needs. Prior to securing bootstraps, determine desired ankle strapping method.
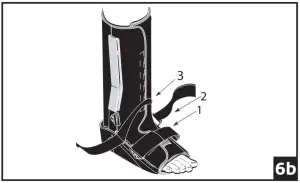
Ankle Cross Strap Conversion: (fig 6a) Strap 3 is removable and may be secured by feeding through d-ring on opposing upright. (fig 6b) If Cross Strapping is desired, feed Strap 3 through opposing forefoot d-ring. Cross Strap 2 over Strap 3, feed through d-ring on opposing upright and secure. Secure all other bootstraps, starting at the toes and working up the leg.

CLEANING INSTRUCTIONS
To clean liners, blue foam pad and walker boot hand wash in cold water using mild soap, or wipe with a damp cloth and air dry.
Note: If not rinsed thoroughly, residual soap may case irritation and deterioration of materials.
WARRANTY: DJO, LLC will repair or replace all or part of the unit and its accessories for material or workmanship defects for a period of six months from the date of sale. To the extent the terms of this warranty are inconsistent with local regulations, the provisions of such local regulations will apply.
INTENDED FOR SINGLE PATIENT USE.![]() NOT MADE WITH NATURAL RUBBER LATEX.
NOT MADE WITH NATURAL RUBBER LATEX.
RX ONLY.
| NOTICE: WHILE EVERY EFFORT HAS BEEN MADE IN STATE-OF-THE-ART TECHNIQUES TO OBTAIN THE MAXIMUM COMPATIBILITY OF FUNCTION, STRENGTH, DURABILITY AND COMFORT, THERE IS NO GUARANTEE THAT INJURY WILL BE PREVENTED THROUGH THE USE OF THIS PRODUCT. |


MDSS GmbH
Schiffgraben 41
30175 Hannover, Germany

DJO, LLC
5919 Sea Otter Place
Suite 200
Carlsbad, CA 92010
U.S.A.