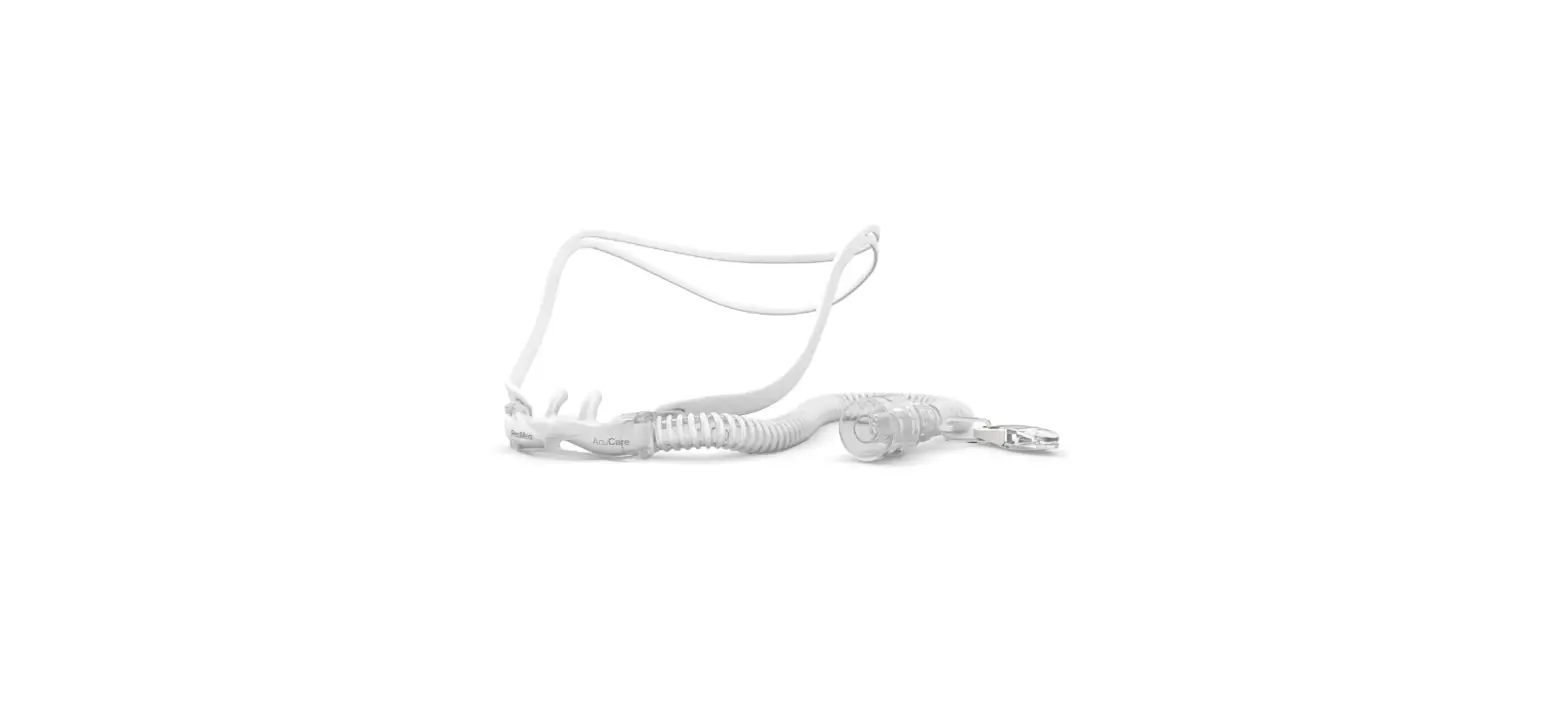
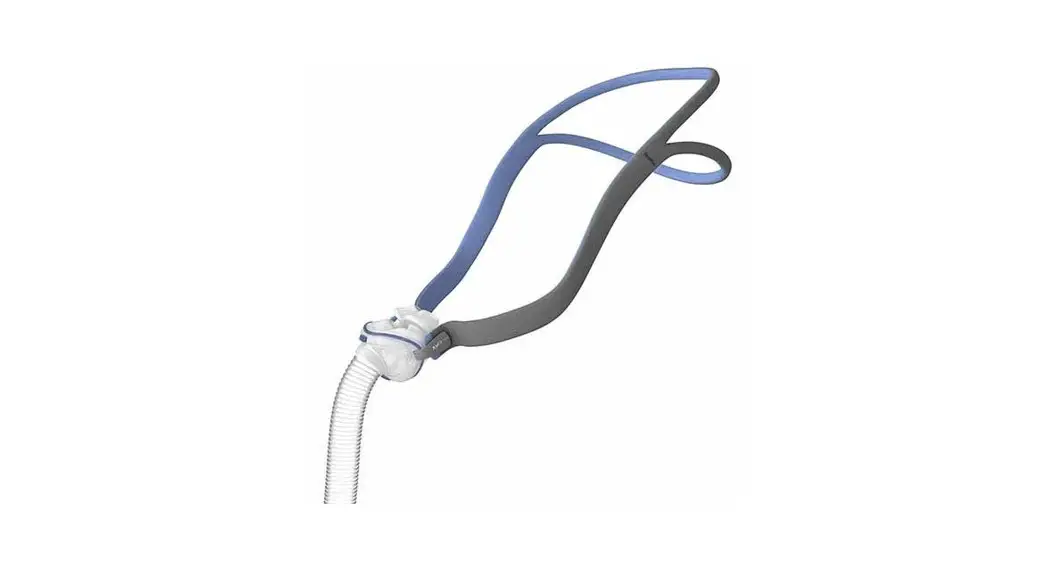
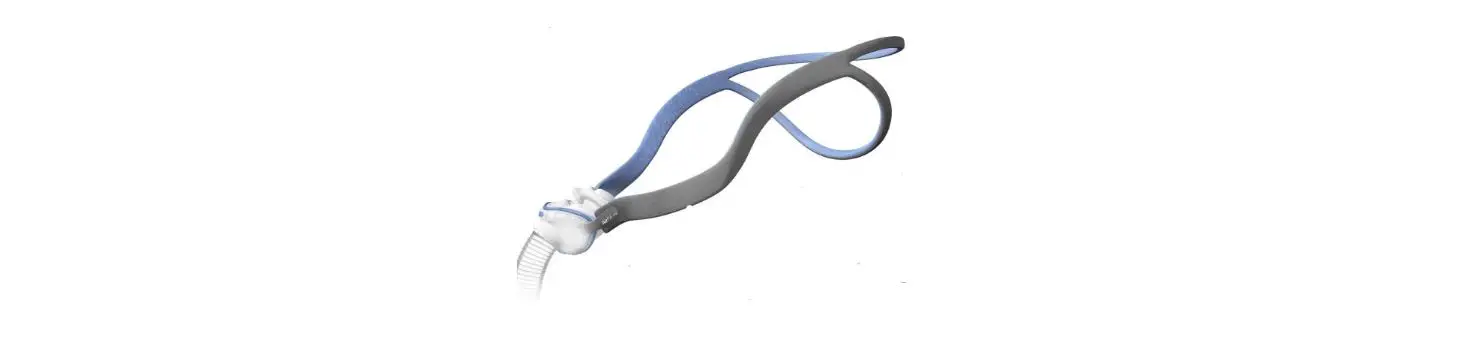
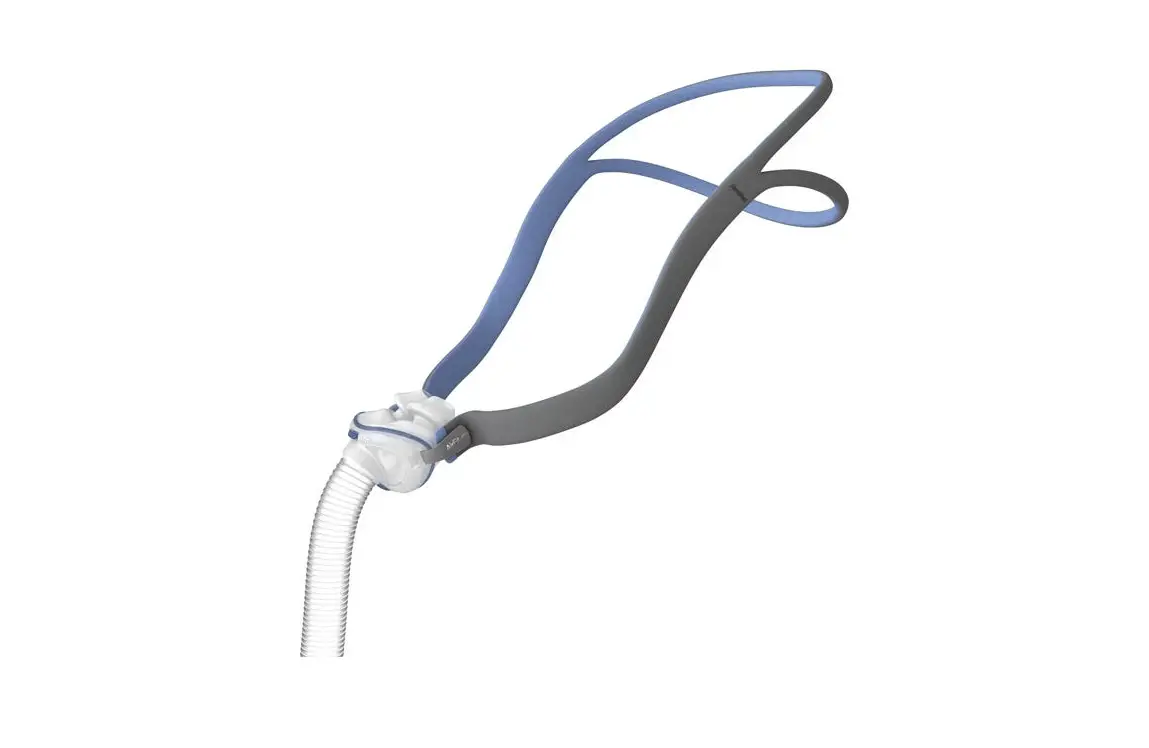
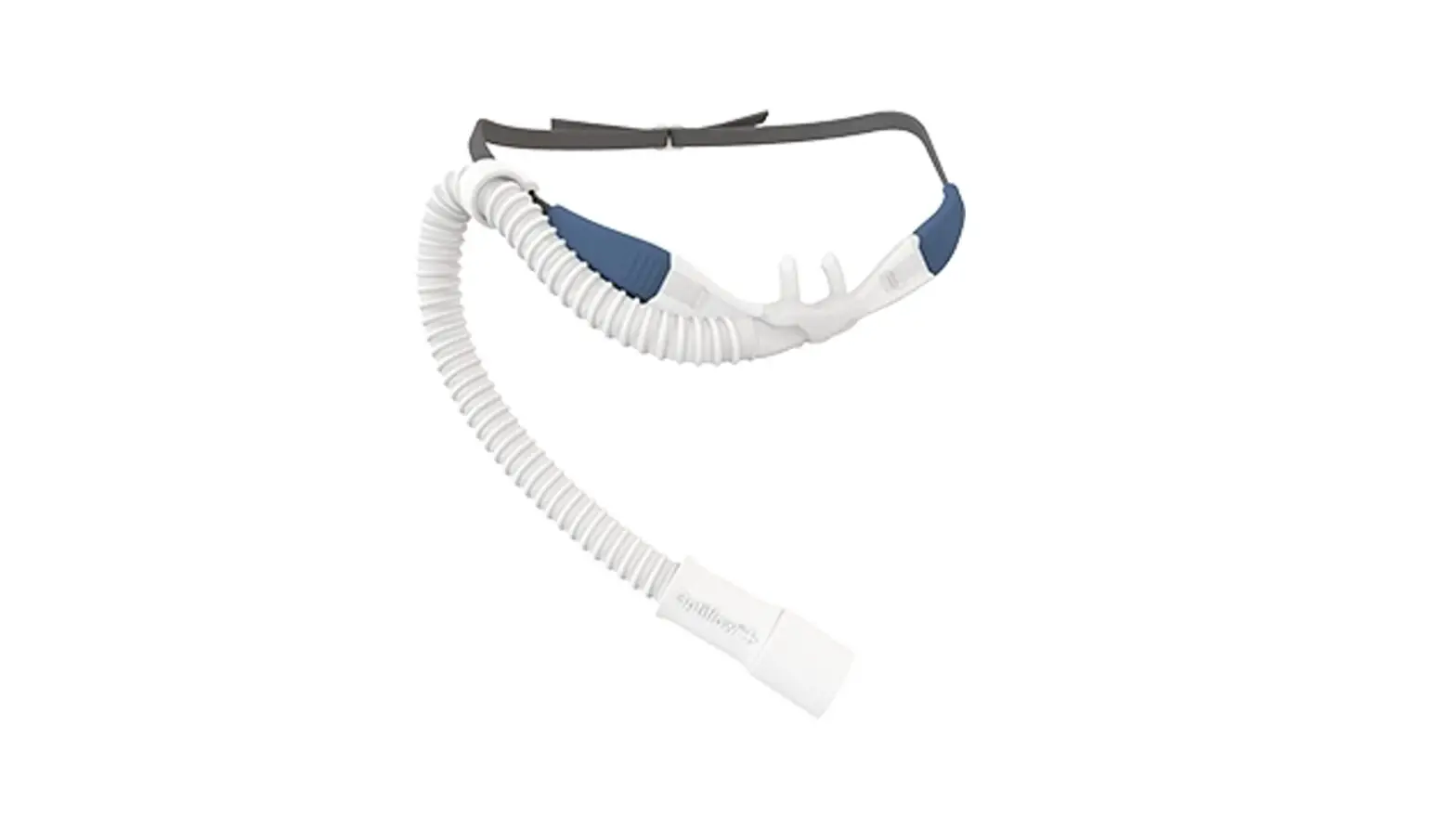
SHS-HW-001 Rapid Non-Pharmaceutical Relief from Nasal Congestion
Product Information
| Model Name | SHS-HW-001 |
|---|---|
| Brand Name | SONU |
| Manufacturer | Sound Health Systems Inc. |
| Address | 650B Fremont Avenue, #65, Los Altos, CA 94024 |
| Website | www.soundhealth.life |
Product Usage Instructions
Conditions for Use
The SONU device is intended for the temporary relief of nasal
congestion. It can be used by individuals aged 18 and older in home
healthcare or professional healthcare facility environments.
Contraindications
You should not use the SONU device if:
- You have a dental infection.
- You are receiving treatment for neurological conditions.
- You have electrostimulation devices implanted in your head,
including deep brain stimulation (DBS) devices or cochlear
implants. - You had any type of surgery to the face, head, nose, or sinuses
within the past 3 months. - You had intracranial or intracerebral bleeding in the past 6
months. - You have open wounds, rashes, swollen, red, infected or
inflamed areas, skin eruptions, or fragile skin on the forehead
(treatment location). - You have a history of cranial surgery.
- You are sensitive to sound.
Precautions
- Read the user manual and instructions for use carefully before
performing any treatment. - Use the device only on the forehead as directed. Do not use it
on the heart, chest, neck, or any other body location. - Do not share the device with other people. It is intended for
single-person use based on individual craniofacial anatomy. - Refer to Sound Health Systems’ website for detailed
information: https://soundhealth.life.
Warnings
The SONU device transmits acoustic vibrations from the bone
conduction transducers in the headband to the sinus cavities.
Ensure that you can pair the headband with the Sonu Chronic App on
your smartphone and hear sound when you play the audio track. If
you are unable to pair the headband or experience loss or
degradation of essential performance due to electromagnetic
disturbances, do not use the device.
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Sonu
by Sound Health Systems
Rapid Non-Pharmaceutical Relief from Nasal Congestion
USER MANUAL
Sound Health Systems
– CONFIDENTIAL –
1
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
© 2022 Sound Health Systems Inc. (“Sound Health”), all rights reserved. The manual or any of its parts should not be reproduced without the written permission of Sound Health Systems.
Sound Health Systems reserves the right to change or improve its products and accompanyingtechnical literature without specific notice.
Model name: SHS-HW-001 Brand name: SONU Manufacturer:
Sound Health Systems Inc. 650B Fremont Avenue, #65 Los Altos, CA 94024
Web: www.soundhealth.life
Sound Health Systems
– CONFIDENTIAL –
2
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Table of Contents
Sonu…………………………………………………………………………………………………………………………………………………… 1 About This Manual………………………………………………………………………………………………………………………………. 4 1. Product Overview …………………………………………………………………………………………………………………………. 4 2. Product Functions…………………………………………………………………………………………………………………………. 4 3. SAFETY…………………………………………………………………………………………………………………………………………. 5 3.1. Conditions for Use ………………………………………………………………………………………………………………………… 5 3.2. Contraindications………………………………………………………………………………………………………………………….. 5 3.3. Precautions ………………………………………………………………………………………………………………………………….. 5 3.4. Warnings ……………………………………………………………………………………………………………………………………… 5 4. Package Contents………………………………………………………………………………………………………………………….. 6 5. Symbols ……………………………………………………………………………………………………………………………………….. 7 6. Setup and Use ………………………………………………………………………………………………………………………………. 8 6.1. Downloading and Installing the Application ……………………………………………………………………………………. 8 6.2. Band Placement and Adjustment …………………………………………………………………………………………………. 16 7. Treatment duration …………………………………………………………………………………………………………………….. 16 8. Treatment Completed …………………………………………………………………………………………………………………. 17 9. Cleaning and Maintenance…………………………………………………………………………………………………………… 17 10. Storing the device for next use …………………………………………………………………………………………………….. 17 11. Technical Specifications ……………………………………………………………………………………………………………….. 18 12. FCC Statements …………………………………………………………………………………………………………………………… 19
Sound Health Systems
– CONFIDENTIAL –
3
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
About This Manual
This manual provides the information necessary for you to effectively use Sonu. · Please follow all manufacturer recommendations · If you have any questions, please email customer support at [email protected]
1. Product Overview · Sonu is a non-invasive, over-the-counter (OTC) device designed to provide temporary relief of nasal congestion through the delivery of sound, or acoustic vibrational energy, to the sinus cavities and nasal passages
2. Product Functions · The Sonu headband is battery-powered. The battery is rechargeable and integrated with the bone conduction transducer assembly. · The headband only includes a USB type C cable for charging. · During charging, the headband does not function as a medical device (Bluetooth is disabled and band cannot be used as a medical device when it is charging) · The headband can be charged via a DC power source e.g. a notebook computer’s USB port · The headband includes two bone conduction transducers providing acoustic vibrational energy to the nasal passages. · The headband is activated by a power button. · The headband should be wrapped around the head and adjusted so that the transducers are placed on the forehead above the eyebrows and maintain contact between the device and the skin. · The Sonu Chronic Application (App) is installed on a smartphone to capture your craniofacial landmarks, and to deliver and monitor the personalized treatment.
Sound Health Systems
– CONFIDENTIAL –
4
Sound Health Systems
3. SAFETY
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
3.1. Conditions for Use
Sonu is intended to be used for the temporary relief of nasal congestion. Sonu treatment can be used in a home healthcare or professional healthcare facility environments by individuals 18 and older.
3.2. Contraindications You should not use this device if:
I. You have a dental infection. II. You are receiving treatment for neurological conditions. III. You have electrostimulation devices implanted in your head, including a deep brain
stimulation (DBS) devices or cochlear implants IV. You had any type of surgery to the face, head, nose or sinuses within the past 3
months V. You had an intracranial or and intracerebral bleeding in the past 6 months. VI. You have open wounds, rashes, over swollen, red, infected or inflammed areas,
skin eruptions or fragile skin on the forehead (treatment location). VII. You have a history of cranial surgery. VIII. You are sensitive to sound.
3.3. Precautions I. Do not attempt to perform any treatment before carefully reading the user manual and
instructions for use. II. Do not use the device on the heart, chest, neck, or any other body location other than the
forehead as directed. III. Do not share the device with other people. The device is intended to be used by a single
person to receive personalized therapy based on their individual craniofacial anatomy. IV. Refer to Sound Health Systems’ website: https://soundhealth.life for detailed information.
3.4. Warnings
1. Sonu transmits acoustic vibrations from the bone conduction transducers in the headband to the sinus cavities. After you pair the headband with your smartphone and play the audio
Sound Health Systems
– CONFIDENTIAL –
5
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
track, you will hear sound during treatment (essential performance). Do not use Sonu if you are unable to pair the headband with the Sonu Chronic App on your smartphone or do not hear sound when you play the audio track (i.e., essential performance is lost or degraded) due to electromagnetic disturbances.
2. Do not use Sonu if it shuts down or the performance has degraded in any way. 3. Do not use Sonu near active high-frequency (HF) surgical equipment or in the radio-
frequency (RF) shielded room of a magnetic resonance imaging (MRI) scanner or near RF emitting equipment where electromagnetic disturbances are high, because it could result in improper operation. 4. Avoid using Sonu adjacent to or stacked with other equipment because it could result in improper operation. If such use is necessary, please verify that Sonu and the other equipment are operating normally.
5. Use of accessories, transducers and cables other than those specified or provided by the manufacturer of Sonu could result in increased electromagnetic emissions or decreased electromagnetic immunity of Sonu and result in improper operation.
6. Portable RF communications equipment (including peripherals such as antenna cables and external antennas) should be used no closer than 30 cm (12 inches) to any part of Sonu, including cables specified by the manufacturer. Otherwise, degradation in performance of Sonu could result.
4. Package Contents
· 1 Sonu Headband · 1 Micro-USB charging cable · 1 Storage Box · 1 Quick-start Guide
Sound Health Systems
– CONFIDENTIAL –
6
Sound Health Systems 5. Symbols
Symbol
Qty
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Description Read and fully understand user manual before using this device
Quantity of devices in the package
Manufacturer
Model identifier
Lot Number
Use by date – indicates the date after which the device is not to be used Keep dry
Non-Sterile
MAC ID
MAC address of the device
Do not use if package is damaged
Sound Health Systems
– CONFIDENTIAL –
7
Sound Health Systems
6. Setup and Use
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Before using the device for the first time, install the Sonu Chronic App, and connect the headband to the App. Make sure that Bluetooth connection on your Smartphone is enabled.
6.1. Downloading and Installing the Application
Step 1: Verify that your smartphone is compatible with the Sonu Chronic App. You will need an iPhone 10 (x) or better that has a FaceID front facing camera. Step 2: Download and install the `TestFlight’ App from the Apple App Store. Step 3: Install the Sonu `Chronic’ App via the TestFlight App.
Sound Health Systems
– CONFIDENTIAL –
8
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
How to operate SONU BAND?
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
This device is only available in iOS system and iPhone needs to be 10 or above.
Sound Health Systems
– CONFIDENTIAL –
9
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Step 4: Press and hold the power button until the prototype flashes green. This indicates band is in pairing mode and ready to connect to the iPhone via Bluetooth.
button
Step 5: Open the mobile phone Bluetooth connection and ensure Bluetooth is turned on. LINK SONU_1.8.13 Step 6: Open the APP Chronic Step 7: Connect Sonu band and follow the instructions in the app screens
Sound Health Systems
– CONFIDENTIAL –
10
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
Step 8: Start a session
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Sound Health Systems
– CONFIDENTIAL –
11
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
Step 9: Start face scan.
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Sound Health Systems
– CONFIDENTIAL –
12
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
Step 10: Start a session.
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Sound Health Systems
– CONFIDENTIAL –
13
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Step 11: Sonu band fit test. Follow the instructions on APP.
Sound Health Systems
– CONFIDENTIAL –
14
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
STEP 12: Start to Use
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Step 13: Begin treatment. You should hear audio from the band when you press `Begin’
Sound Health Systems
– CONFIDENTIAL –
15
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
You may continue to use the Band away from the phone as long as the band is within normal Bluetooth accessory wireless range (10-15 feet). Should the App lose connection to the Band, the treatment will be paused. You can restart paused treatments when the Band and the smartphone are in range.
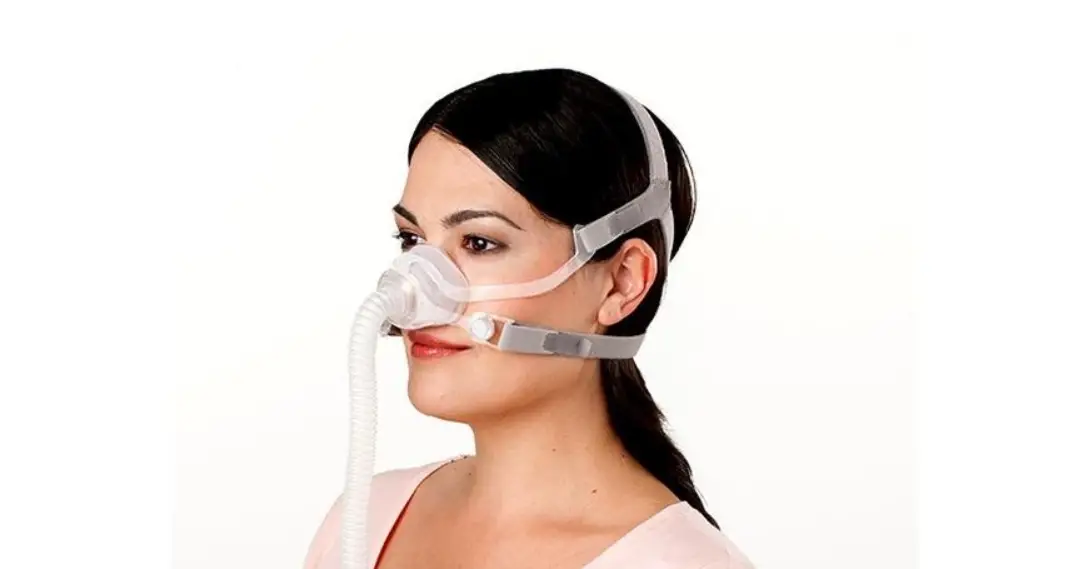
6.2. Band Placement and Adjustment
Positioning the Headband on the forehead with a snug fit is important for treatment efficacy. You should adjust the Band to ensure that it does not slip side to side when you turn your head. Make sure that the bone conduction transducers in the center of the Band are in good contact with the skin on your forehead.
Also note that the silver markers on the band should be aligned with the middle of your forehead right above your brow line as shown in the Figure below:
7. Treatment duration Each treatment dose is 15 minutes. A maximum of two 15-minute treatment sessions (30 minutes total) per day is recommended.
CAUTION: Do not share the Sonu Headband or App. Sonu is intended for a single user and the treatment is specific to you.
Sound Health Systems
– CONFIDENTIAL –
16
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
8. Treatment Completed
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Step 1: When the treatment is complete, remove the headband. The headband will turn off automatically within 15 minutes of the end of a treatment session or inactivity.
Step 2: Close the App on the phone
9. Cleaning and Maintenance · Clean the device with a dry cloth. · Please contact customer support if the package and/or device labeling are damaged. · The lifetime of the band is approximately 1 year and varies depending on skin conditions, individual use, storage conditions, and climate. · You can update the App using the standard update procedures on Apple’s iOS operating system.
10. Storing the device for next use Once the treatment has been completed, please clean and store the device until the next treatment. Store the device in its original package in an indoor environment, away from direct sunlight and according to storage environment conditions specified in this user manual.
Sound Health Systems
– CONFIDENTIAL –
17
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
11. Technical Specifications
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Parameter
Power Source Weight Dimensions
Maximum current consumption Rated input power Impedance Maximum Ouptut Power (Corrected for Position) Transducer Frequency Range
Frequency Range
Modulation Type Bluetooth version Antenna Type Antenna Gain Number of Hopping Frequency Measured Output Peak Power 20 dB Bandwidth Carried Frequency Separation Time of Occupancy (Dwell Time) Conducted Spurious Emission Restricted Frequency Bands
Conducted Emission
Radiated Emission
Specification Transducer 3.7 V rechargeable battery supply 1.20oz / 0.034kg Width: 28mm Height: 36mm Depth: 105mm 500 mA 1 W 8 Ohms ±10% 88dB SPL
100Hz to 15KHz Bluetooth
The frequency range used is 2402MHz 2480MHz (79 channels, at intervals of
1MHz); the frequency block is 2400MHz to 2483.5MHz. Bluetooth: FHSS (GFSK) 5.1 PCB Antenna 5.46 dBi max
>15 < 0.6 dBm; <1.20 mW 0.9567MHz 0.64MHz (2/3rd of the 20dB bandwidth of the hopping channel) <0.4 sec
At least 20dB below that in the 100kHz bandwidth within the band that contains
the highest level of the desired power
Radio frequency voltage that is conducted back onto the AC power line on any
frequency within the band 150kHz to 30MHz shall not exceed the limits
Comply with the radiated emission limits specified in FCC section 15.209(a).
Sound Health Systems
– CONFIDENTIAL –
18
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
12. FCC Statements
15.19 This device complies with part 15 of the FCC Rules. Operation is subject to the following two conditions: (1) This device may not cause harmful interference, and (2) this device must accept any interference received, including interference that may cause undesired operation.
15.105(b) This equipment has been tested and found to comply with the limits for a Class B digital device, pursuant to part 15 of the FCC Rules. These limits are designed to provide reasonable protection against harmful interference in a residential installation. This equipment generates, uses and can radiate radio frequency energy and, if not installed and used in accordance with the instructions, may cause harmful interference to radio communications. However, there is no guarantee that interference will not occur in a particular installation. If this equipment does cause harmful interference to radio or television reception, which can be determined by turning the equipment off and on, the user is encouraged to try to correct the interference by one or more of the following measures:
– Reorient or relocate the receiving antenna.
– Increase the separation between the equipment and receiver.
– Connect the equipment into an outlet on a circuit different from that to which the receiver is connected.
– Consult the dealer or an experienced radio/TV technician for help.
15.21 The user is cautioned that changes or modifications not expressly approved by the party responsible for compliance could void the user’s authority to operate the equipment.
FCC RF Radiation Exposure Statement:
This device complies with FCC radiation exposure limits set forth for general population. This transmitter must not be co-located or operating in conjunction with any other antenna or transmitter. Users should avoid using other body-worn accessories with metallic components in them to maintain compliance with FCC RF exposure guidelines.
FCC ID: 2A8X7SHS-HW-001X
Sound Health Systems
– CONFIDENTIAL –
19
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Guidance and Manufacturer’s declaration- Electromagnetic emissions
Sonu is intended for use in the electromagnetic environment (for home healthcare) specified below. The customer or
the user of Sonu should assure that it is used in such an environment.
Emission test
Compliance
Electromagnetic environment-guidance (for home healthcare environment)
RF emissions CISPR 11
Group 1
Sonu uses RF energy only for its internal function. Therefore,
its RF emissions are very low compared to the FCC Group 1
limits and are not likely to cause any interference with nearby
electronic equipment.
RF emissions CISPR 11
Class B
Sonu is suitable for use in all establishments, including
domestic establishments and those directly connected to the
public low-voltage power supply network that supplies buildings
used for domestic purposes. RF emissions are low compared
to FCC Class B limits.
Guidance and Manufacturer’s declaration- Electromagnetic immunity
Sonu is intended for use in the electromagnetic environment (for home healthcare) specified below. The customer or
the user of Sonu should assure that it is used in such an environment.
Immunity test
IEC 60601
Compliance level
Electromagnetic environment-
Test level
guidance (for home healthcare
environment)
Electrostatic discharge Contact: ±8 kV
Contact: ±8 kV
Floors should be wood, concrete
(ESD)
Air: ±2 kV,±4 kV,±8 kV,±15 Air: ±2 kV,±4 kV,±8
or ceramic tile. If floors are
IEC 61000-4-2
kV
kV,±15 kV
covered with synthetic material,
the relative humidity should be at
least 30%
Power frequency (50, 30 A/m
30 A/m
Sonu power frequency magnetic
60 Hz) magnetic field 50 Hz or 60 Hz
50 Hz
fields should be at levels
IEC 61000-4-8
characteristic of a typical location
in a typical home healthcare
environment.
Sound Health Systems
– CONFIDENTIAL –
20
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Guidance and Manufacturer’s declaration- Electromagnetic immunity
Sonu is intended for use in the electromagnetic environment (for home healthcare) specified below. The customer or
the user of Sonu should assure that it is used in such and environment.
Immunity test
IEC 60601 test level Compliance level
Electromagnetic environment-guidance
(for home healthcare environment)
Portable and mobile RF communications
equipment should be used no closer to
any part of Sonu including cables, than the
recommended separation distance calculated
from the equation applicable to the frequency
of the transmitter.
Radiated RF IEC 61000-4-3
10 V/m 80 MHz 2,7 GHz 80 % AM at 1 kHz
10 V/m 80 MHz 2,7 GHz 80 % AM at 1 kHz
Recommended separation distance: d = 1,2 d = 1,2 80MHz to 800 MHz d = 2,3 800MHz to 2,7 GHz Where P is the maximum output power rating othfethtreatnrasmnsitmteitrtemrainnuwfaactttsur(eWr )aancdcdoridsinthgeto recommended separation distance in metres (m).
Interference may occur in the vicinity of equipment marked with the following symbol:
NOTE1 At 80 MHz and 800 MHz, the higher frequency range applies. NOTE2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and
reflection from structures, objects and people.
Sound Health Systems
– CONFIDENTIAL –
21
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Recommended separation distance between portable and mobile RF communications equipment and Sonu
Sonu is intended for use in an electromagnetic environment (for home healthcare) in which radiated RF disturbances are controlled. The customer or the user of Sonu can help prevent electromagnetic interference by maintaining a minimum distance between portable and mobile RF communications equipment (transmitters) and Sonu as recommended below, according to the maximum output power of the communications equipment.
Rated maximum output power of transmitter W
Separation distance according to frequency of transmitter m
150 kHz to 80 MHz
80 MHz to 800 MHz
800 MHz to 2,7 GHz
d =1,2
d =1,2
d =2,3
0,01
0,12
0,12
0,23
0,1
0,38
0,38
0,73
1
1,2
1,2
2,3
10
3,8
3,8
7,3
100
12
12
23
For transmitters rated at a maximum output power not listed above, the recommended separation distance d in meters
(m) can be estimated using the equation applicable to the frequency of the transmitter, where p is the maximum output
power rating of the transmitter in watts (W) according to the transmitter manufacturer.
NOTE1 At 80 MHz and 800 MHz, the separation distance for the higher frequency range applies.
NOTE2 These guidelines may not apply in all situations. Electromagnetic propagation is affected by absorption and reflection from structures, objects and people.
Sound Health Systems
– CONFIDENTIAL –
22
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Guidance and Manufacturer’s declaration- Electromagnetic immunity Test specifications for ENCLOSURE PORT IMMUNITY to RF wireless communications equipment Sonu is intended for use in the electromagnetic environment (for home healthcare) specified below. The customer or the user of Sonu should assure that it is used in such an environment.
Test frequency
(MHz)
Band a) (MHz)
Service a)
Modulation b)
Maximum power (W)
Distance (m)
IMMUNITY TEST LEVEL (V/m)
Compliance LEVEL (V/m) (for home
healthcare)
Pulse
385
380 390 TETRA 400 modulation b)
1,8
0,3
27
27
18 Hz
FM c)
450
430 470
GMRS 460, FRS 460
±5 kHz deviation
2
0,3
28
28
1 kHz sine
710 745 780
704 787
LTE Band 13, 17
Pulse modulation b)
217 Hz
0,2
0,3
9
9
810
GSM 800/900,
TETRA 800,
Pulse
870
800 960 iDEN 820, modulation b)
2
0,3
28
28
CDMA 850,
18 Hz
930
LTE Band 5
1 720
GSM 1800;
1 845
1 700 1 990
CDMA 1900; GSM 1900; DECT;
LTE Band 1, 3,
Pulse modulation b)
217 Hz
2
0,3
28
28
1 970
4, 25; UMTS
Bluetooth,
2 450
2 400 2 570
WLAN, 802.11 b/g/n, RFID 2450,
Pulse modulation b)
217 Hz
2
0,3
28
28
LTE Band 7
5 240 5 500 5 785
5 100 5 800
WLAN 802.11 a/n
Pulse modulation b)
217 Hz
0,2
0,3
9
9
NOTE If necessary to achieve the IMMUNITY TEST LEVEL, the distance between the transmitting antenna and the ME EQUIPMENT or ME SYSTEM may be reduced to 1 m. The 1 m test distance is permitted by IEC 61000-4-3.
a)
For some services, only the uplink frequencies are included.
b)
The carrier shall be modulated using a 50 % duty cycle square wave signal.
c)
As an alternative to FM modulation, 50 % pulse modulation at 18 Hz may be used because while it does not
represent actual modulation, it would be worst case.
Sound Health Systems
– CONFIDENTIAL –
23
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
Emission Test: IEC 61000-4-39
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
Manufacturer’s declaration-electromagnetic immunity Test specifications for ENCLOSURE PORT IMMUNITY to proximity magnetic fields Sonu is intended for use in the electromagnetic environment (for home healthcare) specified below.
The customer or the user of Sonu should ensure that it is used in such an environment.
Frequencies Test Level [A/m]
Point / Window
Modulation
Dwell time [s]
Compliance LEVEL [A/m] (for home healthcare)
30 kHz (a)
8
All points on photo below
CW
3
8
Pulse modulation (b)
134,2 kHz
65 All points on photo below
3
2,1 kHz
65 (c)
Pulse modulation (b)
13,56 MHz
7,5 All points on photo below
3
50 kHz
7,5 (c)
Note: (a) This test is applicable only to ME EQUIPMENT and ME SYSTEMS intended for use in the HOME HEALTHCARE
ENVIRONMENT. (b) The carrier shall be modulated using a 50 % duty cycle square wave signal. (c) r.m.s., before modulation is applied.
Sound Health Systems
– CONFIDENTIAL –
24
Sound Health Systems
SONU USER MANUAL – COMMERCIAL
CD-013.E Effective Date: 18 Nov 2022 DCO: 027
REVISION HISTORY
Revision Description of Change
A
Initial Release
B
Updated warnings and EMC compatibility information from
EMC testing
C
Updated instructions for use to be understandable to lay users.
Included language on two 10-minute treatments per day.
D
Updated company name and product name to Sound Health
Systems and Sonu respectively
Updated instructions to turn on headband and pairing with the
App; and treatment time
Updated App screen images
Updated with EMC tests to be conducted on Sonu
E
Updated with App Screen Images and EMC Testing results
Effective Date July 27, 2021 Dec 3, 2021 May 6, 2022 Aug 29, 2022
Nov 18, 2022
Originator KT VRao KT VRao KT VRao KT VRao
Vivek Mohan
Sound Health Systems
– CONFIDENTIAL –
25