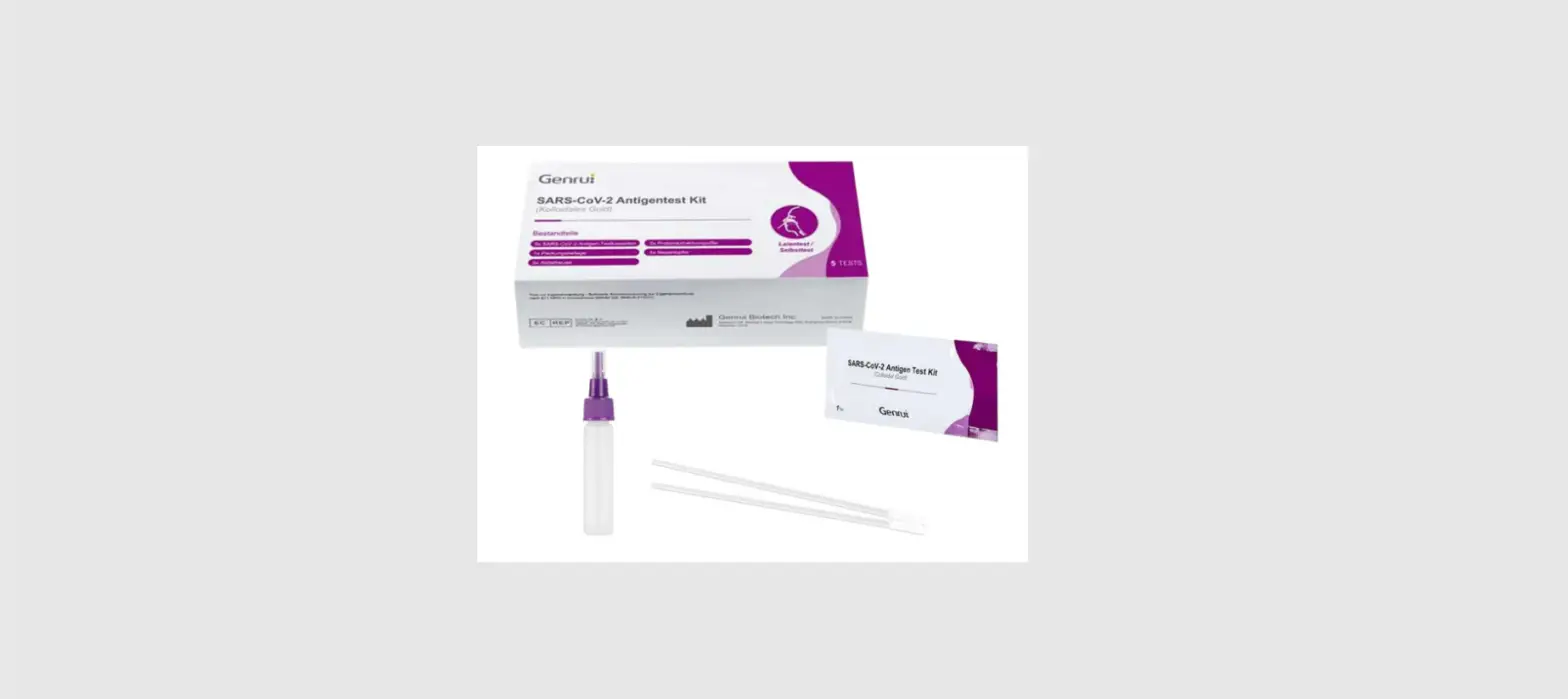
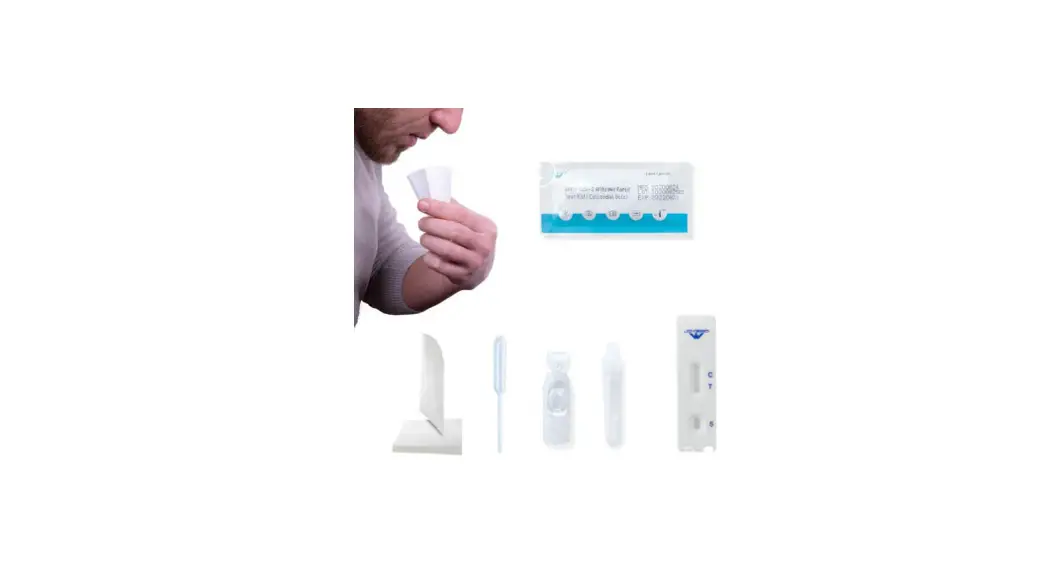
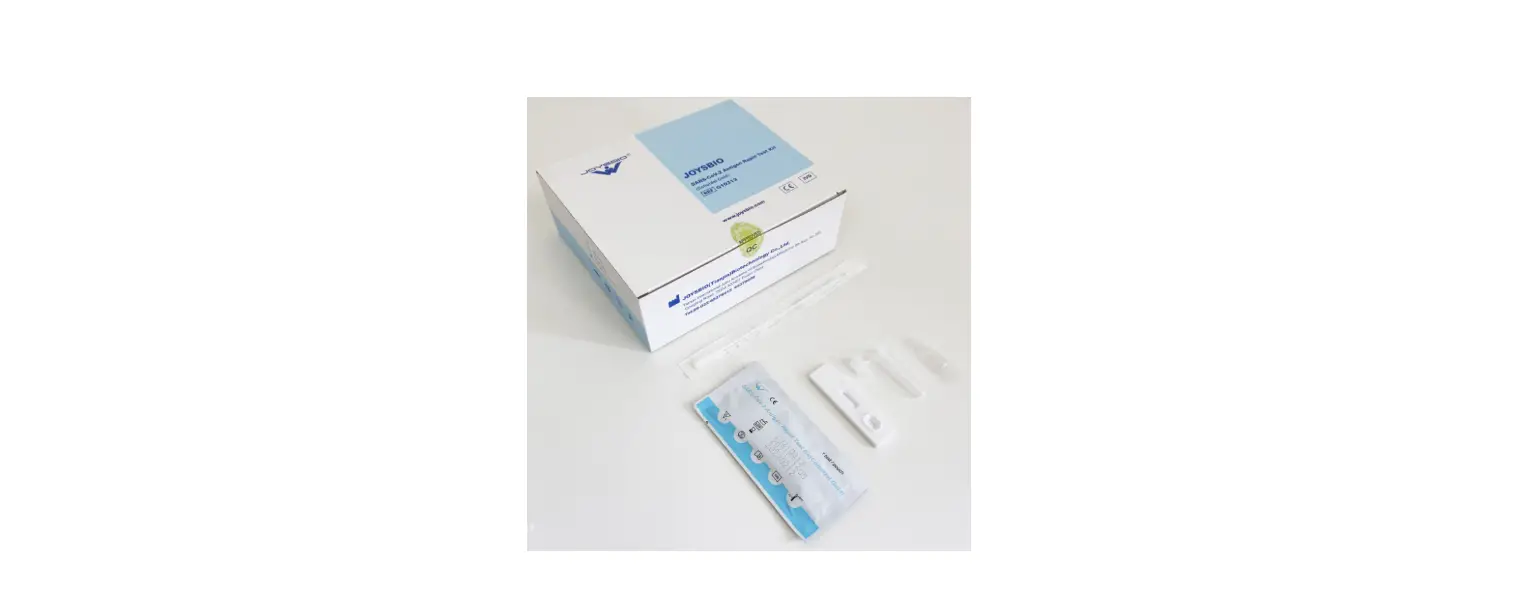
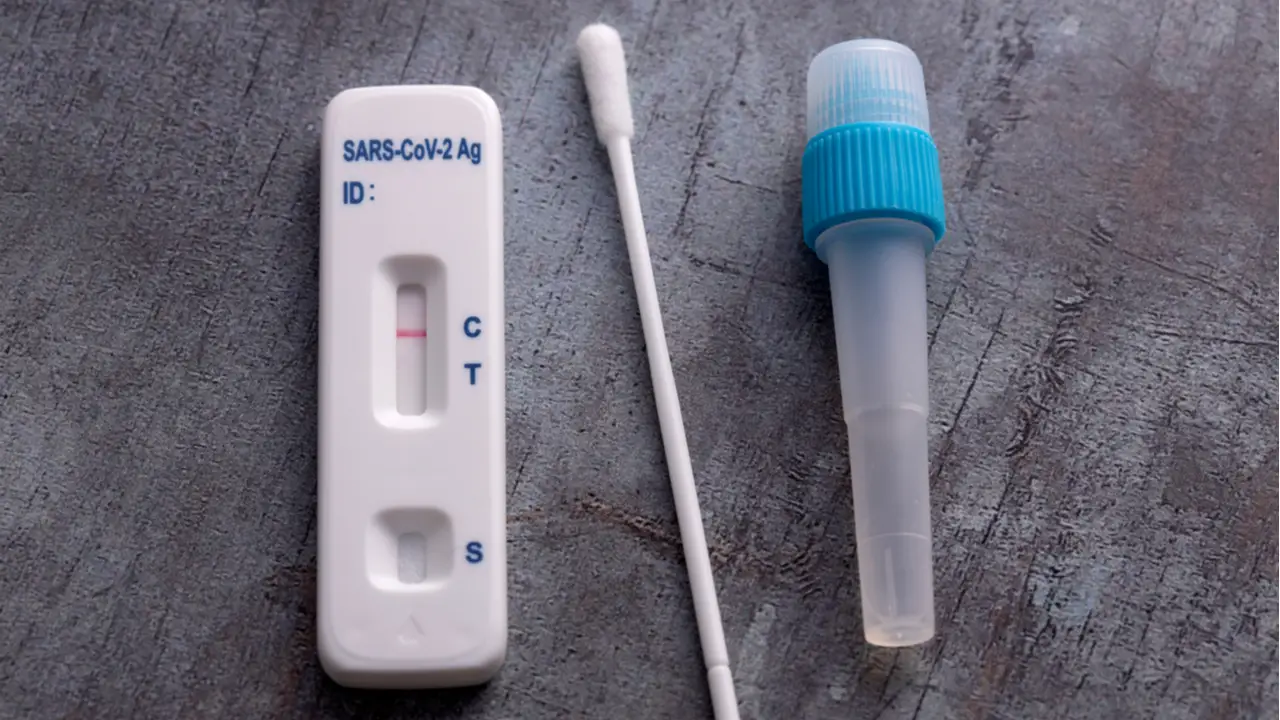
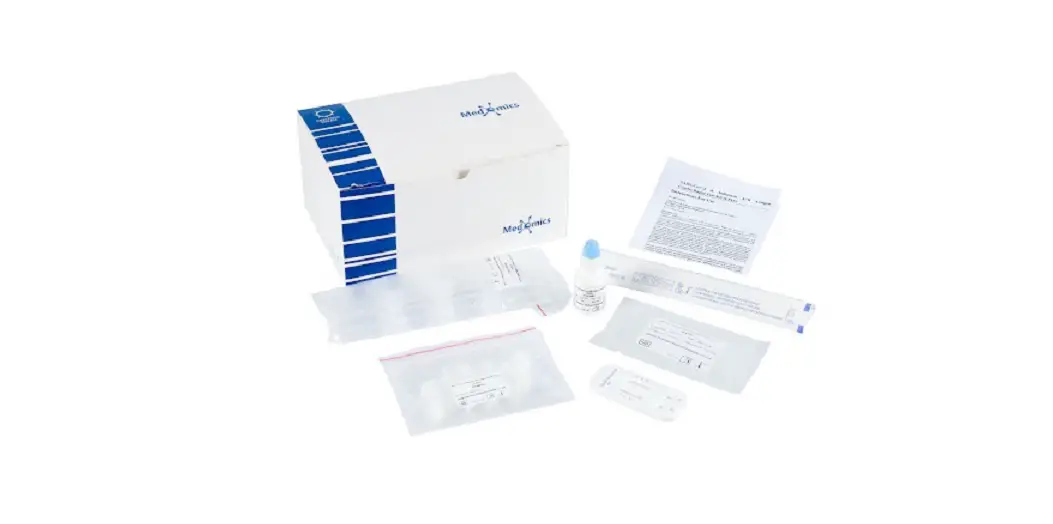
VSCD10-SC RapidFor SARS-CoV-2 and FLU A/B Combo Antigen Test Kit
Product Information: SARS-CoV-2 & FLU A/B Combo Antigen
Test Kit
The SARS-CoV-2 & FLU A/B Combo Antigen Test Kit (CAT NO:
VSCD10-SC) is an in vitro differentiative detection kit for
SARS-CoV-2 & FLU A/B antigens in human nasopharyngeal and nasal
swab samples. It helps in the diagnosis of COVID-19 and influenza
A/B viruses.
Advantages:
- Can detect both SARS-CoV-2 & FLU A/B antigens in a single
test. - Provides accurate and reliable results within 15-20
minutes. - Easy to use and interpret.
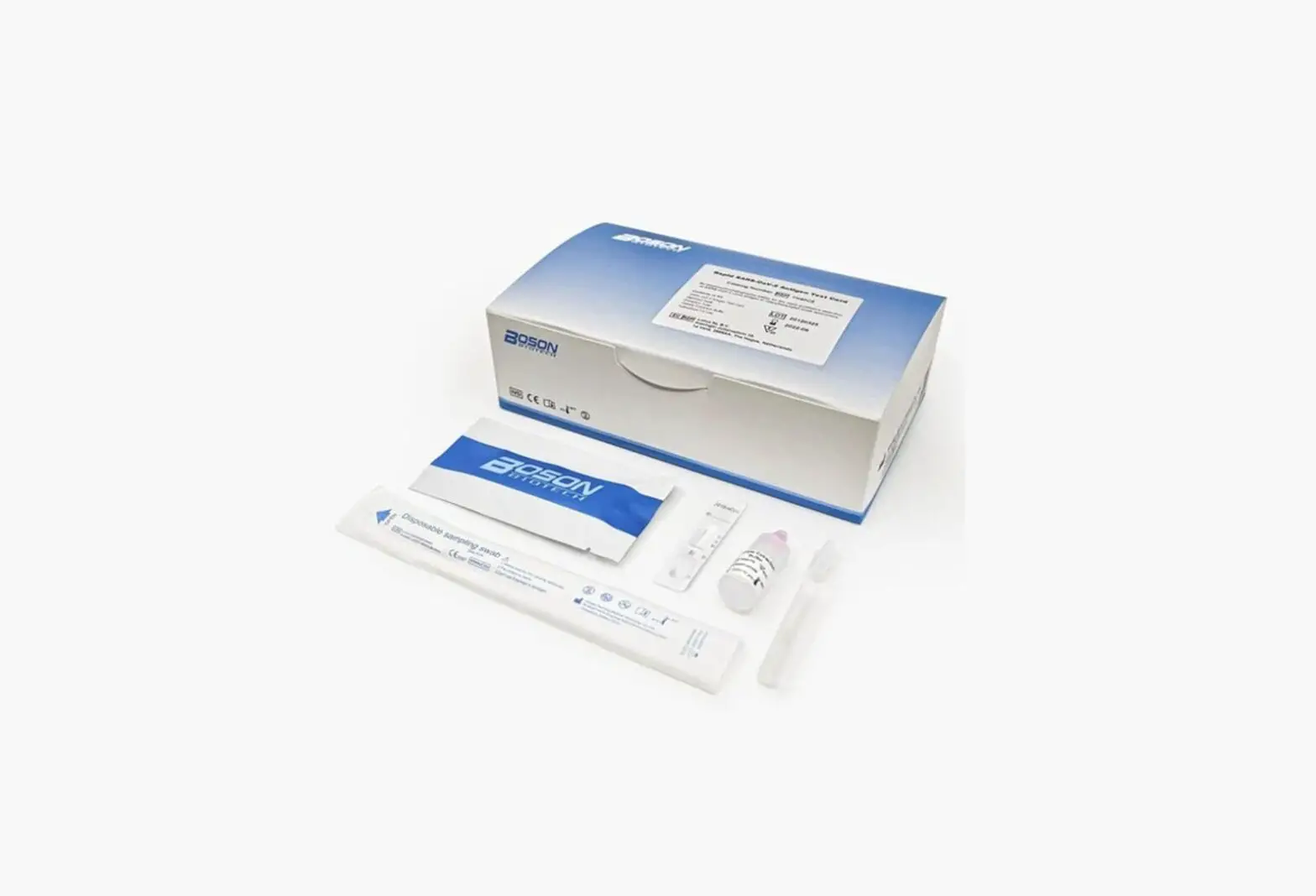
Box Content:
The kit contains the following items:
- 25 test devices
- 25 sterile swabs (nasopharyngeal and nasal)
- 25 extraction vials and caps
- 1 workstation
- 1 instruction manual
Test Procedure:
- Collect a nasopharyngeal or nasal swab specimen from the
patient. - Insert the swab into the extraction vial containing the buffer
solution provided in the kit. - Rotate the swab several times to ensure maximum release of the
specimen. - Remove the swab while squeezing the sides of the vial to
extract the liquid from the swab. - Place the dropper tip onto the extraction vial and dispense 3
drops of the extracted specimen into the sample well of the test
device. - Wait for 15-20 minutes and read the results.
Interpretation of Results:
The test results can be interpreted as follows:
| SARS | FLU | |||||
|---|---|---|---|---|---|---|
| + | + | C | C | T | A/B | Positive |
| – | – | C | C | T | A/B | Negative |
| C | C | T | A/B | Invalid | ||
*Note: SARS + indicates the presence of SARS-CoV-2 antigen, SARS
– indicates the absence of SARS-CoV-2 antigen, FLU + indicates the
presence of influenza A/B antigen, FLU – indicates the absence of
influenza A/B antigen, C indicates the control line, T indicates
the test line, and A/B indicates the type of influenza A/B
virus.
SARS-CoV-2 & FLU A/B COMBO ANTIGEN TEST KIT
CAT NO: VSCD10-SC
It is created as in vitro differentiative detection for SARS-CoV-2 & FLU A/B antigens in human nasopharyngeal and nasal swab samples.
Interpretation
SARS +
FLU +
C
C
T
A
B
Positive
SARS – FLU –
C
C
T
A
B
Negative
C
C
T
A
B
Invalid
Advantages
· For professional use only · Already filled buffer tubes · 2 sampling options · Easy-to-follow procedure · Variant detection · Accurate and rapid results
Box Content
· 25x Test Cassette · 25x Tube · 25x Swab · 25x Dropper · 1x Instructions For Use
Test Procedure
Open
x10 OR
Cat. No
VSCD10-SC
Target
SARS-CoV-2 & FLU A/B Antigens
*NP: Nasopharyngeal Swab, N: Nasal Swab
Specimen*
NP,N
Kit Size
25T
CE Status
CE
vitrosens.com