SofTIP™ Guide Cannula
SofTIP™ Guide Cannula
Instructions for Use
SofTIP Guide Cannula
FIGURE 1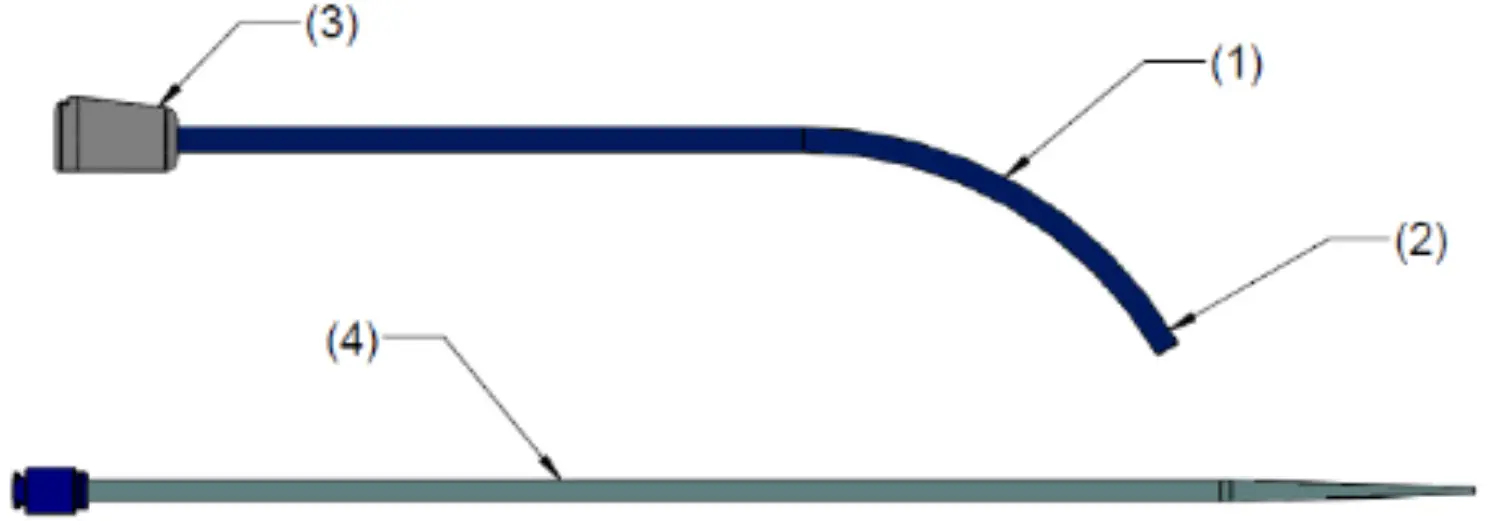 Carefully Read All Instructions Prior to Use.
Carefully Read All Instructions Prior to Use.
Caution: Federal (US) law restricts this device to sale by or on the order of a physician.
PACKAGING AND STORAGE
The SofTIP is sterile (using ethylene oxide gas) and non-pyro-genic in unopened packaging that is designed to maintain sterility unless the primary product pouch has been opened or damaged.
Handle with care. Do not store in excessive heat. After use, this product may be a potential biohazard. Handle and dispose of all such devices in accordance with accepted medical practice and applicable local, state, and federal laws and regulations.
INDICATIONS
The SofTIP Guide Cannula is indicated for use in introduction, guiding, and/or placement of the Lariat RS Suture Delivery Device.
DESCRIPTION
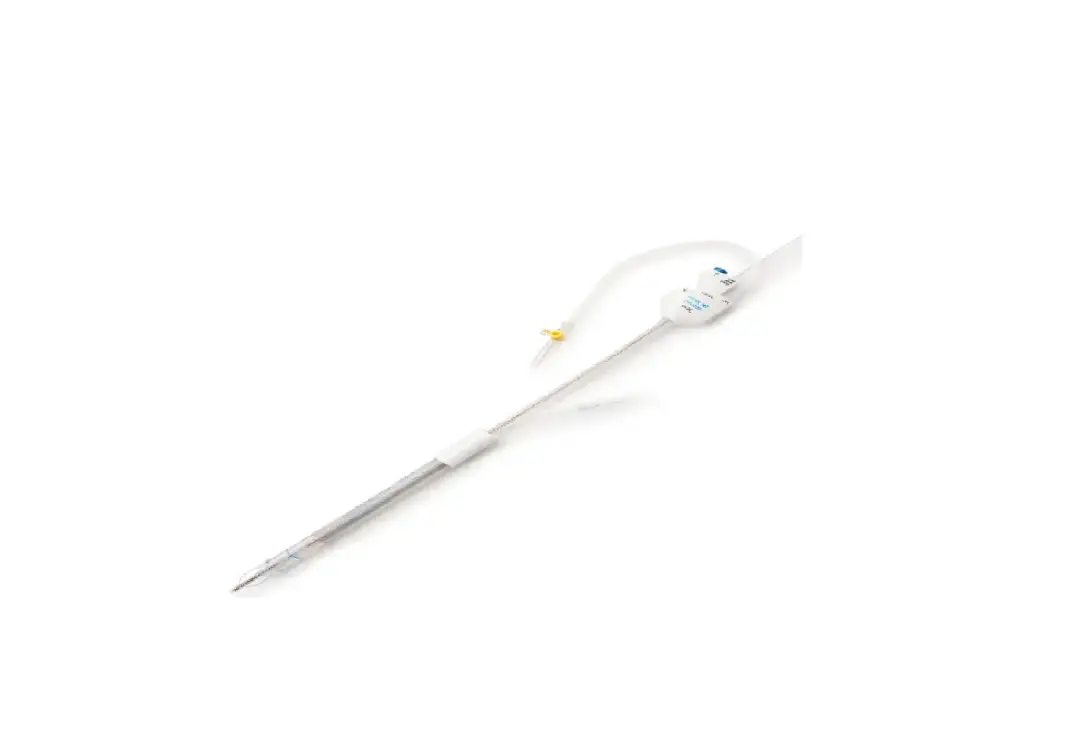
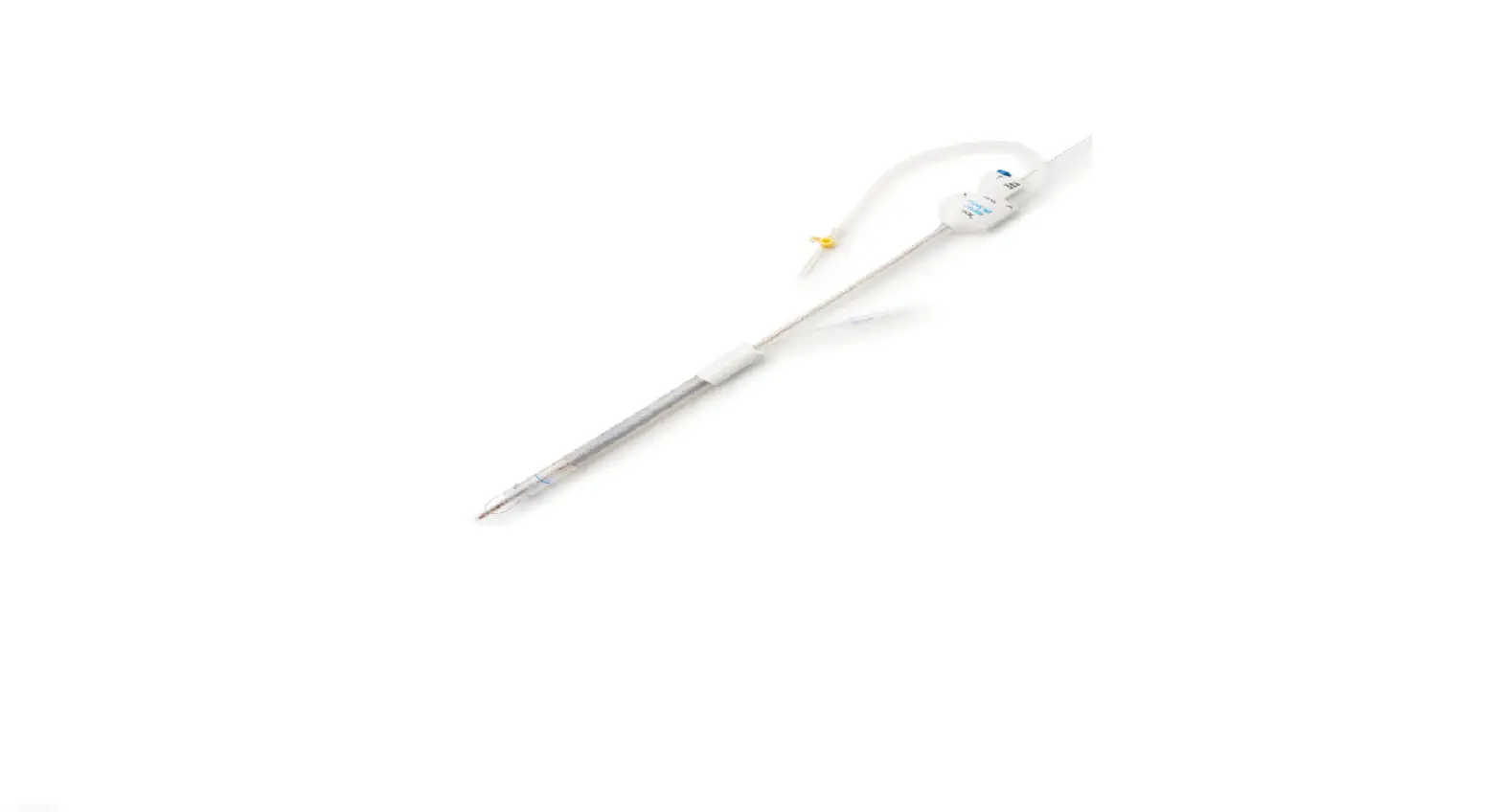
The SofTIP Guide Cannula is a single-use, sterile device. The device is comprised of a pre-formed polymer shaft (1) with a radiopaque marker (2) at the distal end, and a hub (3) at the proximal end that includes an orientation indicator. The SofTIP is packaged with a dilator (4) to aid introduction of the device over a guide wire. See Figure 1.
COMPLICATIONS
When used as directed, no adverse effects are expected with use of this device.![]() Warnings
Warnings ![]()
Never advance the SofTIP without the dilator in place as this may result in potential perforation of cardiac structures causing bleeding or embolism.
Always use standard visualization techniques (e.g., fluoroscopy and echocardiography) for guidance when moving the device (e.g., advancing, withdrawing, and re-locating) to minimize the risk of injury.
Do not advance, withdraw, torque, or move the device when resistance is met without first determining and resolving the cause. Manipulation when resistance is met may lead to perforation of cardiac tissues causing bleeding or embolism.
For single use only. Do not reuse, reprocess or resterilize. Reuse, reprocessing or resterilization may compromise the structural integrity of the device and/or lead to device failure which in turn may result in patient injury, illness or death. Re-use, reprocessing or resterilization may also create a risk of contamination of the device and/or cause patient infection or crossinfection, including, but not limited to, the transmission of infectious disease(s) from one patient to another. Contamination of the device may lead to injury, illness, or death of the patient.
Do not advance the SofTIP and dilator without the guide wire beyond the tip of the dilator.![]() PRECAUTIONS
PRECAUTIONS
• Care should be taken to avoid damage when handling the device. Do not use if the device is visibly kinked, damaged or deformed.
• Review the Instructions for Use before using.
INSTRUCTIONS FOR USE
Preparation
- Carefully inspect the SofTIP Guide Cannula and the packaging prior to use. Do not use a device that is damaged in any way or if its packaging is damaged.
- Remove the SofTIP and dilator from the sterile packaging and remove from the packaging card.
- Insert the dilator into the SofTIP and rotate the dilator hub to lock in place.
Guide Wire Placement - Using standard percutaneous techniques, deliver an .035″ guide wire to the desired anatomical location.
Device Delivery - Serial dilate over the guide wire as required to facilite introduction of the SofTIP.
- Backload the guidwire into the SofTIP dilator, and advance the SofTIP/dilator together to the desired anatomical location.
Note: Use standard visualization techniques (e.g. fluoroscopy) for guidance when advancing the device to the desired anatomical location. - When the desired location is reached, rotate the dilator to unlock and withdraw from the SofTIP.
- During use, the flat side of the hub can be used to indicate the orientation of the curve in the shaft.
Device Disposal - Dispose of the SofTIP device as appropriate in accordance with accepted medical practice and applicable local, state and federal laws and regulations.
All rights reserved Patent(s) pending
SYMBOLS GLOSSARY
| Manufacturer | Catalog Number | Use-By Date | |||
| Lot Number | Contents of the package | Do Not Reuse | |||
| Do Not Use if Package is Damaged | Keep Away From Sunlight | Do Not Resterilize | |||
| Keep Dry | Caution | Caution: Federal law (US) restricts this device to sale by or on the order of a physician. | |||
| Sterilized using ethylene oxide | Consult Instructions For Use | Authorized European Representative | |||
| Product complies with the requirements of directive 93/42/EEC | Non pyrogenic |

 Manufacturer
Manufacturer
AtriCure Incorporated
7555 Innovation Way
Mason, Ohio 45040 USA
Customer Service:
1-866-349-2342 (toll free)
+1 513-755-4100 (phone)![]() European Representative:
European Representative:
AtriCure Europe B.V.
De entree 260
1101 EE Amsterdam
NL
+31 20 7005560
[email protected]
IFU-0120.B
2021/12![]()