Abbott Panbio COVID-19 Ag Rapid Test Device
 Introduction
Introduction
The Coronavirus disease (COVID-19) is an infectious disease caused by a newly discovered coronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)1. The SARS-CoV-2 is a β-coronavirus, which is an enveloped non-segmented positive-sense RNA virus2. It is spread by human-to-human transmission via droplets or direct contact, and infection has been estimated to have a mean incubation period of 6.4 days and a basic reproduction number of 2.24-3.58. Among patients with pneumonia caused by SARS-CoV-2, fever was the most common symptom, followed by cough3. The main IVD assays used for COVID-19 employ real-time reverse transcriptase-polymerase chain reaction (RT-PCR) that takes a few hours4. The availability of a cost-effective, rapid pointofcare diagnostic test is critical to enable healthcare professionals to aid in the diagnosis of patients and prevent further spread of the virus5. Antigen tests will play a critical role in the fight against COVID-196.
Test Principle
Panbio™ COVID-19 Ag Rapid Test Device contains a membrane strip, which is pre-coated with immobilized anti-SARS-CoV-2 antibody on the test line and mouse monoclonal anti-chicken IgY on the control line. Two types of conjugates (human IgG specific to SARS-CoV-2 Ag gold conjugate (binds to the nucleocapsid protein) and chicken IgY gold conjugate) move upward on the membrane chromatographically and react with anti-SARS-CoV-2 antibody and pre-coated mouse monoclonal anti-chicken IgY respectively. For a positive result, human IgG specific to SARS-CoV-2 Ag gold conjugate and anti-SARS-CoV-2 antibody will form a test line in the result window. Neither the test line nor the control line are visible in the result window prior to applying the patient specimen. A visible control line is required to indicate a test result is valid.
Intended Use
Panbio™ COVID-19 Ag Rapid Test Device is an in vitro diagnostic rapid test for the qualitative detection of SARS-CoV-2 antigen (Ag) in human nasal swab specimens from individuals who meet COVID-19 clinical and / or epidemiological criteria. Panbio™ COVID-19 Ag Rapid Test Device is for professional use only and is intended to be used as an aid in the diagnosis of SARS-CoV-2 infection.
The product may be used in any laboratory and non-laboratory environment that meets the requirements specified in the Instructions for Use and local regulation.
The test provides preliminary test results. Negative results don’t preclude SARSCoV-2 infection and they cannot be used as the sole basis for treatment or other management decisions. Negative results must be combined with clinical observations, patient history, and epidemiological information. The test is not intended to be used as a donor screening test for SARS-CoV-2.
Kit Variants
• 41FK11 No 2D barcode printed on the contained test devices
• 41FK21 Contains test devices with a 2D barcode printed on the test device,
which encodes traceability information for the product
Materials Provided
• 25 Test devices with desiccant in individual foil pouch
• Buffer (1 x 9 ml/bottle)
• 25 Extraction tubes
• 25 Extraction tube caps
• 1 Positive control swab
• 1 Negative control swab
• 25 Sterilized nasal swabs for sample collection
• 1 Tube rack
• 1 Quick Reference Guide
• 1 Instructions for use
Materials Required but not Provided
- Personal Protective Equipment per local recommendations (i.e. gown/ lab coat, face mask, face shield/eye goggles and gloves), Timer, Biohazard container
Active Ingredients of Main Components - 1 Test device Gold conjugate: Human IgG specific to SARS-CoV-2 Ag gold colloid and Chicken IgY – gold colloid, Test line: Mouse monoclonal anti-SARS-CoV-2, Control line: Mouse monoclonal anti-Chicken IgY
- Buffer Tricine, Sodium Chloride, Tween 20, Sodium Azide (<0.1%), Proclin 300
Storage and Stability
- The test kit should be stored at a temperature between 2-30 °C. Do not freeze the kit or its components.
Note: When stored in a refrigerator, all kit components must be brought to room temperature (15-30 °C) for a minimum of 30 minutes prior to performing the test. Do not open the pouch while components come to room temperature. - The Buffer bottle may be opened and resealed for each assay. The Buffer cap should be firmly sealed between each use. The Buffer is stable until expiration date if kept at 2-30 °C.
- Perform the test immediately after removing the test device from the foil pouch.
- Do not use the test kit beyond its expiration date.
- The shelf life of the kit is as indicated on the outer package.
- Do not use the test kit if the pouch is damaged or the seal is broken.
- Direct swab specimens should be tested immediately after collection. If immediate testing is not possible, the swab specimen can be kept in an extraction tube filled with extraction buffer (300 μl) at room temperature (15-30 °C) for up to two hours prior to testing.
Warnings
- For in vitro diagnostic use only. Do not reuse the test device and kit components.
- These instructions must be strictly followed by a trained healthcare professional to achieve accurate results. All users have to read the instruction prior to performing a test.
- Do not eat or smoke while handling specimens.
- Wear protective gloves while handling specimens and wash hands thoroughly afterwards.
- Avoid splashing or aerosol formation of specimen and buffer.
- Clean up spills thoroughly using an appropriate disinfectant.
- Decontaminate and dispose of all specimens, reaction kits and potentially contaminated materials (i.e. swab, extraction tube, test device) in a biohazard container as if they were infectious waste and dispose according to applicable local regulations.
- Do not mix or interchange different specimens.
- Do not mix reagent of different lots or those for other products.
- Do not store the test kit in direct sunlight.
- To avoid contamination, do not touch the head of provided swab when opening the swab pouch.
- The sterilized swabs should be used only for nasal specimen collection.
- To avoid cross-contamination, do not reuse the sterilized swabs for specimen collection.
- Do not dilute the collected swab with any solution except for the provided extraction buffer.
- The buffer contains <0.1% sodium azide as a preservative which may be toxic if ingested. When disposed of through a sink, flush with a large volume of water.7
- Do not use the positive or negative control swab for specimen collection.
Test Procedure (Refer to Figure)
Nasal swab Specimens
Note: Healthcare professionals should comply with personal safety guidelines including the use of personal protective equipment.
Test Preparation
- Allow all kit components to reach a temperature between 15-30 °C prior to testing for 30 minutes.
- Remove the test device from the foil pouch prior to use. Place on a flat, horizontal and clean surface.
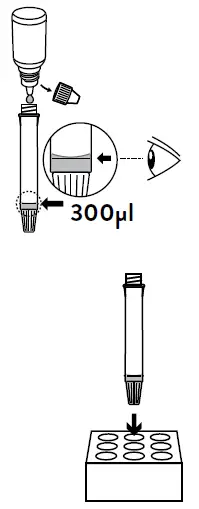
- Hold the buffer bottle vertically and fill the extraction tube with buffer fluid until it flows up to the Fill-line of the extraction tube (300 μl).
Caution: If the amount of buffer is excessive or insufficient, an improper test result may occur. - Place the extraction tube in the tube rack.
Nasal Mid-Turbinate (NMT) Specimen Collection & Extraction
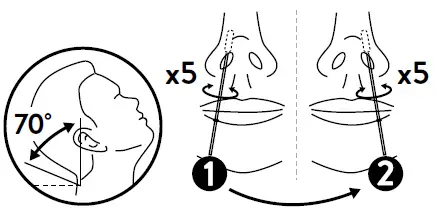
- Tilt the patient’s head back 70 degrees. While gently rotating the swab, insert swab less than one inch (about 2 cm) into nostril (until resistance is met atthe turbinates).
- Rotate the swab five times against the nasal wall then slowly remove from the nostril.
- Using the same swab repeat the collection procedure with the second nostril.
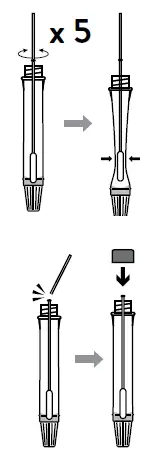
Caution: If the swab stick breaks during specimen collection, repeat specimen collection with a new swab. - Swirl the swab tip in the buffer fluid inside the extraction tube, pushing into the wall of the extraction tube at least five times and then squeeze out the swab by squeezing the extraction tube with your fingers.
- Break the swab at the breakpoint and close the cap of extraction tube.
Reaction with Test Device
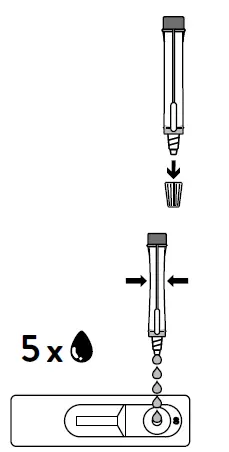
- Open the dropping nozzle cap at the bottom of the extraction tube.
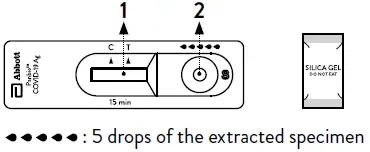
- Dispense 5 drops of extracted specimens vertically into the specimen well (S) on the device. Do not handle or move the test device until the test is complete and ready for reading.
Caution: Bubbles that occur in the extraction tube can lead to inaccurate results. If you are unable to create sufficient drops, this may be caused by clogging in the dispensing nozzle. Shake the tube gently to release the blockage until you observe free drop formation. - Close the nozzle and dispose of the extraction tube containing the used swab according to your local regulations and biohazard waste disposal protocol.
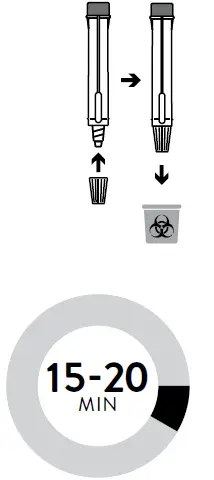
- Start timer. Read result at 15 minutes. Do not read results after 20 minutes.
- Dispose of the used device according to your local regulations and biohazard waste disposal protocol.
Positive / Negative Control Swab
Caution: Control use only. Do not use the positive or negative control swab for specimen collection.
Note: Please refer to the External Quality Control section of this Instructions for use for the frequency of testing external quality control swabs.
- Hold the buffer bottle vertically and fill the extraction tube with buffer fluid until it flows up to the Fill-line of the extraction tube (300 μl).
Caution: If the amount of buffer is excessive or insufficient, an improper test result may occur. - Place the extraction tube in the tube rack.
- Insert the positive or negative control swab in the buffer fluid inside of the extraction tube and soak the swab for 1 minute. Swirl the control swab tip in the buffer fluid inside of the extraction tube, pushing into the wall of the extraction tube at least five times and then squeeze out the swab by squeezing the extraction tube with your fingers.
- Dispose of the used control swab in accordance with your biohazard waste disposal protocol.
- Close the cap of the extraction tube.
- Follow the above test procedure [Reaction with Test Device].
Test Interpretation (Refer to Figure)
- Negative result: The presence of only the control line (C) and no test line (T) within the result window indicates a negative result.
- Positive result: The presence of the test line (T) and the control line (C) within the result window, regardless of which line appears first, indicates a positive result.
Caution: The presence of any test line (T), no matter how faint, indicates a positive result. - Invalid result: If the control line (C) is not visible within the result window after performing the test, the result is considered invalid.
Test Limitations
- The contents of this kit are to be used for the professional and qualitative detection of SARS-CoV-2 antigen from nasal swab. Other specimen types may lead to incorrect results and must not be used.
- Failure to follow the instructions for test procedure and interpretation of test results may adversely affect test performance and/or produce invalid results.
- A negative test result may occur if the specimen was collected, extracted or transported improperly. A negative test result does not eliminate the possibility of SARS-CoV-2 infection and should be confirmed by viral culture or a molecular assay.
- Positive test results do not rule out co-infections with other pathogens.
- Test results must be evaluated in conjunction with other clinical data available to the physician.
- Reading the test results earlier than 15 minutes or later than 20 minutes may give incorrect results.
- Panbio™ COVID-19 Ag Rapid Test Device is not intended to detect from defective (non-infectious) virus during the later stages of viral shedding that might be detected by PCR molecular tests.8
- Positive results may occur in cases of infection with SARS-CoV.
Quality Control
- Internal Quality Control:
The test device has a test line (T) and a control line (C) on the surface of the test device. Neither the test line nor the control line are visible in the result window before applying a specimen. The control line is used for procedural control and should always appear if the test procedure is performed properly and the test reagents of the control line are working. - External Quality Control:
The controls are specifically formulated and manufactured to ensure performance of the Panbio™ COVID-19 Ag Rapid Test Device and are used to verify the user’s ability to properly perform the test and interpret the results.
The Positive Control contains recombinant SARS-CoV-2 nucleocapsid protein, which is not contagious. The Positive Control will produce a positive test result and has been manufactured to produce a visible test line (T). The Negative Control will produce a negative test result. Control swabs are not specific for a particular Panbio™ COVID-19 Ag Rapid Test Device lot and may be used between test device lots until the swabs’ expiry dates.
Good laboratory practice suggests the use of positive and negative controls to ensure that:- Test reagents are working, and
- The test is correctly performed.
The external controls can be run under any of the following circumstances: - By a new operator prior to performing testing on patient specimens,
- When receiving a new test shipment,
- At periodic intervals as dictated by local requirements, and/or by the user’s Quality Control procedures.
Performance Characteristics
- External evaluation of Panbio™ COVID-19 Ag Rapid Test Device (Symptomatic) Clinical performance of Panbio™ COVID-19 Ag Rapid Test Device was determined by testing 104 positive nasal swab specimens and 404 negative specimens for SARS-CoV-2 antigen (Ag) to have a sensitivity of 98.1% (95%
CI: 93.2-99.8%) and a specificity of 99.8% (95% CI: 98.6-100.0%). Clinical specimens were determined to be positive or negative using an FDA EUA RT-PCR reference method. The individuals on which the reported sensitivity and specificity are based also had a nasopharyngeal swab taken, which was tested in the FDA EUA-approved RT-PCR.
Panbio™ COVID-19 Ag Rapid Test Device ResultsNasal PCR Test Result Positive Negative Total Panbio™ COVID-19 Positive 102 1 103 Ag Rapid Test Device Negative 2 403 405 Result (nasal swab Total 104 404 508 specimens) Sensitivity Specificity Overall Percent Agreement 98.1% 99.8% 99.4% [93.2%; 99.8%] [98.6%; 100.0%] [98.3%; 99.9%] - Performance data was calculated from a study of individuals suspected of exposure to COVID-19 or who have presented with symptoms in the last 7 days.
- Stratification of the positive specimens post onset of symptoms or suspected exposure between 0-3 days has a sensitivity of 100.0% (95% CI: 92.3- 100.0%; n=46) and 4-7 days has a sensitivity of 96.6% (95% CI: 88.1- 99.6%; n=58).
- Positive agreement of the Panbio™ COVID-19 Ag Rapid Test Device is higher with samples of Ct values ≤30 with a sensitivity of 100.0% (95% CI: 96.0-100.0%) and Ct values ≤33 with a sensitivity of 99.0% (95% CI: 94.5- 100.0%). As indicated in References 8-10, patients with Ct value >30 are no longer contagious. 8, 9, 10
- The clinical performance data was also calculated vs nasopharyngeal swab specimens using an FDA EUA RT-PCR reference and has a sensitivity of 91.1% (95% CI: 84.2-95.6%) and specificity of 99.7% (95% CI: 98.6- 100.0%).
- External evaluation of Panbio™ COVID-19 Ag Rapid Test Device (Asymptomatic) Clinical performance of Panbio™ COVID-19 Ag Rapid Test Device was determined by testing 483 asymptomatic subjects for SARS-CoV-2 antigen (Ag). Clinical specimens were determined to be positive or negative using an FDA EUA RT-PCR reference method.
The positive results (n=50) were stratified by the comparator method cycle threshold (Ct) counts and assessed to better understand the correlation of product performance, as a surrogate for the amount of virus present in the clinical sample. A lower Ct value corresponds to a higher virus concentration.
As presented in the table below, the positive agreement increases with lower Ct values.
The specificity (n=433) was 100% with 95% CI [99.2%; 100.0%].
The results for sensitivity are summarized in the following table:All Nasal PCR Positive Samples (n=50) Ct values ≤ 33 (n=40) Ct values ≤ 30 Ct (n=32) Sensitivity 66.0% 80.0% 93.8% [CI 95%] [51.2%; 78.8%] [64.4%; 90.9%] [79.2%; 99.2%] As indicated in References 8-10, patients with Ct value >30 are no longer contagious. 8, 9, 10
- External evaluation of Panbio™ COVID-19 Ag Rapid Test Device (Self-Collected Swab)
The clinical performance of Panbio™ COVID-19 Ag Rapid Test Device was assessed in 287 symptomatic subjects (≥16 years of age) who collected their swab specimen (self swabbing) under the direction and supervision of a trained professional. The swab was then handed to the trained professional who executed the remaining steps of the procedure. The trained professional also collected a nasopharyngeal swab from each subject to be used as a reference specimen. The reference specimen was tested on the Panbio™ COVID-19
Ag Rapid Test Device.
The results are summarized in the following table:Panbio™ COVID-19 Ag Rapid Test Device (Nasopharyngeal) Positive Negative Total Panbio™ COVID-19 Positive 110 0 110 Ag Rapid Test Device Negative 2 175 177 (Nasal) – Self-collected Total 112 175 287 Swab Positive Negative Overall Percent Agreement Agreement Agreement 98.2% 100.0% 99.3% [93.7%; 99.8%] [97.9%; 100.0%] [97.5%; 99.9%] - Detection Limit
Panbio™ COVID-19 Ag Rapid Test Device was confirmed to detect 2.5X101.8 TCID50/ml of SARS-CoV-2 which was isolated from a COVID-19-confirmed patient in Korea. - Hook Effect
There is no hook effect at 1.0×105.8 TCID50/ml of SARS-CoV-2 which was isolated from a COVID-19-confirmed patient in Korea. - Cross Reactivity
Cross-reactivity of Panbio™ COVID-19 Ag Rapid Test Device was evaluated by testing 28 viruses and 13 other microorganisms. The final test concentrations of viruses and other microorganisms are documented in the Table below. The following viruses and other microorganisms except the Human SARS-coronavirus Nucleoprotein have no effect on the test results of Panbio™ COVID-19 Ag Rapid Test Device.
Panbio™ COVID-19 Ag Rapid Test Device has cross-reactivity with Human- SARS-coronavirus Nucleoprotein at a concentration of 25 ng/ml or more because SARS-CoV has high homology (79.6%) to SARS-CoV-2.No. Types of Specimen Cross Reaction Substance Final Test Concentration Test Result 1 Virus
Adenovirus Type 1 2.2 X 107 TCID50/ml
No cross reaction 2 Adenovirus Type 5 5.71 X 108 TCID50/ml
No cross reaction 3 Adenovirus Type 7 2.86 X 109 TCID50/ml
No cross reaction 4 Enterovirus (EV68) 2.81 X 107 TCID50/ml
No cross reaction 5 Echovirus2 1.0 X 106.5 TCID50/ml
No cross reaction 6 Echovirus11 5.0 X106.25 TCID50/ml
No cross reaction 7 Enterovirus D68 2.81 X 107 TCID50/ml
No cross reaction 8 Human herpesvirus (HSV) 1 5.0 X 107.5 TCID50/ml
No cross reaction 9 Human herpesvirus (HSV) 2 5.0 X 105.75 TCID50/ml
No cross reaction 10 Mumps Virus Ag 1.58 X 105 TCID50/ml
No cross reaction 11 Influenza virus A (H1N1) Strain (A/Virginia/ATCC1/2009) 3.71 X 105 PFU/ml No cross reaction 12 Influenza virus A (H1N1) Strain (A/WS/33) 5.0 X 107.25 TCID50/ml
No cross reaction 13 Influenza virus A(H1N1) Strain (A/California/08/2009/pdm09) 1.6 X 108 TCID50/ml
No cross reaction 14 Influenza virus B Strain (B/ Lee/40) 5.0 X 106.25 TCID50/ml
No cross reaction 15 Parainfluenza Type 1 3.06 X 108 TCID50/ml
No cross reaction 16 Parainfluenza Type 2 5.0 X 105 TCID50/ml
No cross reaction No. Types of Specimen Cross Reaction Substance Final Test Concentration Test Result 17 Virus
Parainfluenza Type 3 6.6 X 107 TCID50/ml
No cross reaction 18 Parainfluenza Type 4A 2.81 X 107 TCID50/ml
No cross reaction 19 Respiratory syncytial virus (RSV) type A 4.22 X 105 TCID50/ml
No cross reaction 20 Respiratory syncytial virus (RSV) type B 5.62 X 105 TCID50/ml
No cross reaction 21 Rhinovirus A16 1.26 X 106 TCID50/ml
No cross reaction 22 HCoV-HKU1 1.5mg/ml No cross reaction 23 HCoV-NL63 1.7 X 105 TCID50/ml
No cross reaction 24 HCoV-OC43 8.9 X 105 TCID50/ml
No cross reaction 25 HCoV-229E 1.51 X 106 TCID50/ml
No cross reaction 26 Human SARS-coronavirus Nucleoprotein 25ng/ml Cross reaction 27 MERS-CoV Nucleoprotein 0.25mg/ml No cross reaction 28 Human Metapneumovirus (hMPV) 16 Type A1 1.51 X 106 TCID50/ml
No cross reaction No. Types of Specimen Cross Reaction Substance Final Test Concentration Test Result 1 Other Microorganism
Staphylococcus saprophyticus 1.9 X 107 CFU/ ml No cross reaction 2 Neisseria sp.(Neisseria lactamica) 1.7 X 108 CFU/ ml No cross reaction 3 Staphylococcus haemolyticus 3.5 X 109 CFU/ ml No cross reaction 4 Streptococcus salivarius 1.96 X 107 CFU/ ml No cross reaction 5 Hemophilus parahaemolyticus 2.2 X 108 CFU/ ml No cross reaction 6 Proteus vulgaris 7.2 X 106 CFU/ ml No cross reaction 7 Moraxella catarrhalis 4.7 X 107 CFU/ ml No cross reaction 8 Klebsiella pneumoniae 5.0 X 106 CFU/ ml No cross reaction 9 Fusobacterium necrophorum 1.75 X 108 CFU/ ml No cross reaction 10 Mycobacterum tuberculosis 10mg/ml No cross reaction 11 Pooled human nasal wash N/A* No cross reaction 12 Streptococcus pyogenes 3.6 X 107 CFU/ ml No cross reaction 13 Mycoplasma pneumoniae 4 X 108 CFU/ml No cross reaction No concentration provided by supplier. Undiluted stock solution was tested.
- Interfering Substances
The following 43 potentially interfering substances have no impact on Panbio™ COVID-19 Ag Rapid Test Device. The final test concentrations of the interfering substances are documented in the Table below.No. Types of Specimen Interfering Substances Final Test Concentration Test Result 1 Endogenous Substance
Mucin 0.5% No Interference 2 Hemoglobin 100 mg/L No Interference 3 Triglycerides 1.5 mg/L No Interference 4 Icteric (Bilirubin) 40 mg/dL No Interference 5 Rheumatoid factor 200 IU/ml No Interference 6 Anti-nuclear antibody >1:40 No Interference 7 Pregnant 10-fold dilution No Interference 8 Exogenous Substance
Guaiacol glyceryl ether 1 μg/ml No Interference 9 Albuterol 0.005 mg/dL No Interference 10 Ephedrine 0.1 mg/ml No Interference 11 Chlorpheniramine 0.08 mg/dL No Interference 12 Diphenhydramine 0.08 mg/dL No Interference 13 Ribavirin 26.7 μg /ml No Interference 14 Oseltamivir 0.04 mg/dL No Interference 15 Zanamivir 17.3 μg /ml No Interference 16 Phenylephrine hydrochloride 15% v/v No Interference 17 Oxymetazolin hydrochloride 15% v/v No Interference 18 Amoxicillin 5.4 mg/dL No Interference 19 Acetylsalicylic acid 3 mg/dL No Interference 20 Ibuprofen 21.9 mg/dL No Interference 21 Chlorothiazide 2.7 mg/dL No Interference 22 Indapamide 140 ng/ml No Interference 23 Glimepiride (Sulfonylureas) 0.164 mg/dL No Interference 24 Acarbose 0.03 mg/dL No Interference 25 Ivermectin 4.4 mg/L No Interference 26 Lopinavir 16.4 μg/L No Interference 27 Ritonavir 16.4 μg/L No Interference 28 Chloroquine phosphate 0.99 mg/L No Interference No. Types of Specimen Interfering Substances Final Test Concentration Test Result 29 Exogenous Substance
Sodium chloride with preservatives 4.44 mg/ml No Interference 30 Beclomethasone 4.79 ng/ml No Interference 31 Dexamethasone 0.6 µg/ml No Interference 32 Flunisolide 0.61 µg/ml No Interference 33 Triamcinolone 1.18 ng/ml No Interference 34 Budesonide 2.76 ng/ml No Interference 35 Mometasone 1.28 ng/ml No Interference 36 Fluticasone 2.31 ng/ml No Interference 37 Sulfur 9.23 µg/ml No Interference 38 Benzocaine 0.13 mg/ml No Interference 39 Menthol 0.15 mg/ml No Interference 40 Mupirocin 10 µg/ml No Interference 41 Tobramycin 24.03 µg/ml No Interference 42 Biotin 1.2 µg/ml No Interference 43 HAMA 63.0 ng/ml No Interference - Repeatability & Reproducibility
Repeatability & Reproducibility of Panbio™ COVID-19 Ag Rapid Test Device was established using in-house reference panels containing negative specimens and a range of positive specimens. There were no differences observed within-run, between-run, between-lots, between-sites, and between-days.
PREPARATION
- Allow all kit components to reach a temperature between 15- 30°C prior to testing for 30 minutes.
Note: Healthcare professionals should comply with personal safety guidelines including the use of personal protective equipment. - Open the package and look for the following:
- Test device with desiccant in individual foil pouch
- Buffer
- Extraction tube
- Extraction tube cap
- Positive control swab
- Negative control swab
- Sterilized nasal swabs for sample collection
- Tube rack
- Quick reference guide
- Instructions for use
- Carefully read these instructions prior to using Panbio™
COVID-19 Ag Rapid Test Device kit. - Look at the expiration date of the kit box. If the expiration date has passed, use another kit.
- Open the foil pouch and look for the following:
- Result window
- Specimen well
Then, label the device with the patient identifier.
TEST PROCEDURE
- Hold the buffer bottle vertically and fill the extraction tube with buffer fluid until it flows up to the Fill-line of the extraction tube (300μl).
Caution: If the amount of buffer is excessive or insufficient, an improper test result may occur.
- Place the extraction tube in the tube rack.
- Tilt the patient’s head back 70 degrees. While gently rotating the swab, insert swab less than one inch (about 2 cm) into nostril (until resistance is met at the turbinates). Rotate the swab five times against the nasal wall. Using the same swab repeat the collection procedure with the second nostril. Slowly remove swab from the nostril.
Caution: If the swab stick breaks during specimen collection, repeat specimen collection with a new swab.
- Insert the swab specimen in the extraction tube. Swirl the swab tip in the buffer fluid inside the extraction tube, pushing into the wall of the extraction tube at least five times and then squeeze out the swab by squeezing the extraction tube with your fingers.

- Break the swab at the breakpoint and close the cap of extraction tube.
- Open the dropping nozzle cap at the bottom of the extraction tube.

- Dispense 5 drops of extracted specimens vertically into the specimen well (S) on the device. Do not handle or move the test device until the test is complete and ready for reading.
Caution: Bubbles that occur in the extraction tube can lead to inaccurate results. If you are unable to create sufficient drops, this may be caused by clogging in the dispensing nozzle. Shake the tube gently to release the blockage until you observe free drop formation. - Close the nozzle and dispose of the extraction tube containing the used swab according to your local regulations and biohazard waste disposal protocol.

- Start timer. Read result at 15 minutes. Do not read results after 20 minutes.
- Dispose of the used device according to your local regulations and biohazard waste disposal protocol.
TEST INTERPRETATION
NEGATIVE
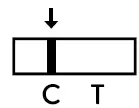
The presence of only the control line (C) and no test line (T) within the result window indicates a negative result.
POSITIVE
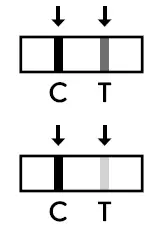
The presence of the test line (T) and the control line (C) within the result window, regardless of which line appears first, indicates a positive result.
Caution: The presence of any test line (T), no matter how faint, indicates a positive result.
INVALID
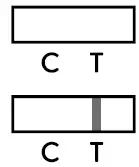
If the control line (C) is not visible within the result window after performing the test, the result is considered invalid. Instructions may not have been followed correctly. It is recommended to read the IFU again before re-testing the specimen with a new test device.
REFERENCES
- Rothan HA, Byrareddy SN. The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J Autoimmun. 2020; Feb 26:102433. doi: 10.1016/j.jaut.2020.102433.
- Guo YR, Cao QD, Hong ZS, et al. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak-an update on the status. Mil Med Res. 2020; Mar 13; 7(1):11.doi:10.1186/s40779-020-00240-0.
- Lai CC, Shih TP, Ko WC, et al. Severe acute respiratory syndrome coronavirus 2 (SARSCov- 2) and coronavirus disease-2019 (COVID-19): The epidemic and the challenges. Int J Antimicrob Agents. 2020; Mar 55(3): 105924.doi: 10.1016/j.ijantimicag.2020.105924.
- In Vitro Diagnostic Assays for COVID-19: Recent Advances and Emerging Trends (Sandeep Kumar Vashist, 2020 April 05: diagnostics)
- Nano Research for COVID-19 (http://dx.doi.org/10.1021/acsnano.0c02540)
- Coronavirus (COVID-19) Update: FDA Authorizes First Antigen Test to Help in the Rapid
Detection of the Virus that Causes COVID-19 in Patients (Stephen M, Hahn M.D. 2020 May 09: Commisioner of Food and Drugs - Current Intelligence Bulletin 13: Explosive Azide Hazard DHHS (NIOSH) Publication Number 78-127 August 16, 1976
- CDC. Discontinuation of Transmission-Based Precautions and Disposition of Patients with COVID-19 in Healthcare Settings (Interim Guidance). (2020).
- CDC. Duration of Isolation and Precautions for Adults with COVID-19. (2020).
- Bullard, et al. Predicting Infectious Severe Acute Respiratory Syndrome Coronavirus 2 From
Diagnostic Samples. CID. 2020; Nov 15;71 (10); DOI:10.1093/cid/ciaa638
GLOSSARY OF SYMBOLS
| Temperature limitation | |
| For in vitro diagnostic use only | |
 | Do not reuse |
 | Do not use if package is damaged |
| Lot Number | |
| Catalog Number | |
 | Consult instructions for use |
 | Keep dry |
 | Biological Risks |
 | Use By |
| Manufacturer | |
| Date of manufacture | |
 | Keep away from sunlight |
| CE mark | |
 | Contains sufficient for X tests |
 | Caution |
| Sterilized using ethylene oxide | |||
| Sterilized using irradiation | |||
 | Do not re-sterilize | ||
| Negative control | |||
| Positive control | |||
TECHNICAL SUPPORT
| TECHNICAL SUPPORT | |
| Europe & Middle East | +44 161 483 9032 |
| Africa, Russia & CIS | +27 10 500 9700 |
| Asia Pacific | +61 7 3363 7711 |
| Latin America | +57 2 661 8797 |
Abbott Rapid Diagnostics Jena GmbH Orlaweg 1, D-07743 Jena, Germany
abbott.com/poct
Date Issued : 2021.01
41FK11/41FK21-01-EN-A1
2021 Abbott. All rights reserved.
All trademarks referenced are trademarks of either the Abbott group of companies or their respective owners.


 Introduction
Introduction