![]()
![]()
ElisaRSRTM 21-OH Ab
21-Hydroxylase (21-OH) Autoantibody ELISA Kit – Instructions for use
![]()
RSR Limited
Parc Ty Glas, Llanishen, Cardiff
CF14 5DU United Kingdom
Tel.: +44 29 2068 9299
Fax: +44 29 2075 7770
Email: [email protected]
Website: www.rsrltd.com
![]() Advena Ltd. Tower Business Centre, 2nd Flr., Tower Street, Swatar, BKR 4013 Malta.
Advena Ltd. Tower Business Centre, 2nd Flr., Tower Street, Swatar, BKR 4013 Malta.
INTENDED USE
The RSR 21-hydroxylase autoantibody (21-OH Ab) ELISA kit is intended for use by professional persons only, for the quantitative determination of 21-OH Ab in human serum. Autoimmune destruction of the adrenal cortex is the most common cause of Addison’s disease and autoantibodies to the adrenal specific enzyme steroid 21 hydroxylase are important markers of adrenal autoimmunity. This can be the case if the disease presents as Addison’s disease or as part of the autoimmune polyglandular syndromes (APS) type I or type II.
REFERENCES
J. Furmaniak and B. Rees Smith
Editorial: Adrenal and Gonadal Autoimmune Diseases.
J. Clin. Endocrinol. Metab. 1995 80: 1502 1505
S. Chen et al
Autoantibodies to Steroidogenic Enzymes in Autoimmune Polyglandular Syndrome, Addison’s Disease, and Premature Ovarian Failure.
J. Clin. Endocrinol. Metab. 1996 81: 1871-1876
H. Tanaka et al
Steroid 21-Hydroxylase
Autoantibodies: Measurements with a New Immunoprecipitation Assay.
J. Clin. Endocrinol. Metab. 1997 82: 1440-1446
G. Coco et al
Estimated Risk for Developing Autoimmune Addison’s Disease in Patients with Adrenal Cortex Autoantibodies.
J. Clin. Endocrinol. Metab. 2006 91: 1637-1645
E. S. Husebye et al
Consensus Statement on the Diagnosis, Treatment, and Follow-up of Patients with Primary Adrenal Insufficiency.
J. Intern. Med. 2014 275: 104-115
ASSAY PRINCIPLE
In RSR’s 21-OH Ab ELISA kit, 21-OH Ab in patients’ sera, reference preparation or calibrators (optional), and controls are allowed to interact with 21-OH coated onto ELISA plate wells. After a 16 – 20 hour incubation, the samples are discarded leaving 21-OH Ab bound to the 21-OH coated on the wells. 21OH-Biotin is added in a 2nd incubation step where, through the ability of 21-OH Ab to act divalently, a bridge is formed between the 21-OH immobilized on the plate and 21-OH-Biotin. The amount of 21-OHBiotin bound is then determined in a 3rd incubation step involving the addition of streptavidin peroxidase (SA-POD), which binds specifically to biotin. Excess, unbound SA-POD has then washed away and the addition of the peroxidase substrate 3,3′,5,5’tetramethlybenzidine (TMB) results in the formation of a blue color. This reaction is stopped by the addition of a stop solution, causing the good contents to turn yellow. The absorbance of the yellow reaction mixture at 450nm and 405nm is then read using an ELISA plate reader. A higher absorbance indicates the presence of 21-OH Ab in the test sample. Reading at 405nm allows quantitation of high absorbances. It is recommended that low absorbance values be measured at 450nm. If it is possible to read at only one wavelength 405nm may be used.
STORAGE AND PREPARATION OF TEST SERUM SAMPLES
Sera to be analyzed should be assayed soon after separation or stored, preferably in aliquots, at or below 20oC. 100 L is sufficient for one assay (duplicate 50 L determinations). Repeated freeze-thawing or increases in storage temperature should be avoided. Do not use lipemic or haemolysed serum samples. Do not use plasma in the assay. When required, bring test sera to room temperature and mix gently to ensure homogeneity. Centrifuge serum prior to assay (preferably for 5 min at about 10,000 rpm i.e. about 10,000 g in a microfuge) to remove particulate matter. Please do not omit this centrifugation step if sera are cloudy or contain particulates.
SYMBOLS
| Symbol | Meaning |
| EC Declaration of Conformity |
| In Vitro Diagnostic Device | |
| Catalog Number | |
| Lot Number | |
| Consult Instructions |
| Manufactured by | |
| Sufficient for |
| Expiry Date |
| Store |
| Negative Control | |
| Positive Control |
MATERIALS REQUIRED AND NOT SUPPLIED
Pipettes capable of dispensing 50 L and 100 L. Means of measuring various volumes to reconstitute or dilute reagents supplied.
Pure water.
ELISA Plate reader is suitable for 96 well formats and capable of measuring at 450nm and 405nm.
ELISA Plate shaker, capable of 500 shakes/min (not an orbital shaker).
ELISA Plate cover.
ELISA Plate washing machine.
PREPARATION OF REAGENTS SUPPLIED
Store unopened kit and all kit components (A-N) at 2-8°C.
| A | 21-OH Coated Wells 12 break-apart strips of 8 wells (96 in total) in a frame and sealed in a foil bag. Allow foil bag to stand at room temperature (20- 25oC) for 30 minutes before opening. |
| Ensure wells are firmly fitted in the frame provided. After opening return any unused wells to the original foil bag with desiccant provided and seal with adhesive tape. Place foil bag in the self-seal plastic bag provided and store at 2-8oC for up to 6 months. | |
| B | Negative Control 0.7 mL Ready for use |
| C1-2 | Positive Controls I & II 2 x 0.7 mL Ready for use |
| D | Reference Preparation 0.7 mL Ready for use |
| E1-4 | Calibrators (optional) 0.3, 1.0, 10, 100 u/mL (arbitrary RSR units) 4 x 0.7 mL Ready for use |
| F | Reaction Enhancer 6 mL, colored red Ready for use |
| G | 21-OH–Biotin 3 vials Lyophilised |
| Reconstitute with room temperature reconstitution buffer (H) immediately before use (within 30 minutes), 5.5 mL per vial. When more than one vial is to be used, pool the vials and mix gently. | |
| H | Reconstitution Buffer for 21-OH-Biotin 2 x 10 mL Ready for use |
| J | Streptavidin Peroxidase (SA-POD) 0.7 mL Concentrated |
| Dilute 1 in 20 with diluent for SA-POD (K). For example, 0.5 mL (J) + 9.5 mL (K). Store for up to 16 weeks at 2-8oC after dilution. | |
| K | Diluent for SA-POD 15 mL Ready for use |
| L | Peroxidase Substrate (TMB) 15 mL Ready for use |
| M | Stop Solution 12 mL Ready for use |
| N | Concentrated Wash Solution 125 mL Concentrated |
| Dilute 1 in 10 with pure water before use. Store at 2-8oC up to kit expiry date. |
ASSAY PROCEDURE
On day 1 allow all the reagents required for steps 1-3 to stand at room temperature (20-25 o C) for at least 30 minutes prior to use. On day 2 allow all the reagents required for steps 4-13 (except for the coated wells) to stand at room temperature (20-25 o C) for at least 30 minutes prior to use.
The coated wells from day 1 must remain at 2–8 o C until ready to proceed with step 4 below. Do not reconstitute 21-OH-Biotin until step 5 below. A repeating Eppendorf-type pipette is recommended for steps 2, 5, 8, 11, and 12.
| Day 1 | 1. | Pipette 50 µL (in duplicate) of patient sera, negative control (B), positive controls (C1–2), reference preparation (D), and (if used) calibrators (E1-4) into respective wells (A). Leave one well empty for blank (see step 13). |
| 2. | Pipette 50 µL reaction enhancer (F) into each well (except blank). | |
| 3. | Cover the frame and shake the wells on an ELISA plate shaker (500 shakes per min) for 1 minute. Incubate overnight (16-20 hours) at 2-8oC without shaking. | |
| Day 2 | 4. | Aspirate and wash/aspirate the wells three times with diluted wash solution (N) by use of an ELISA plate washing machine. |
| 5. | Reconstitute 21-OH-Biotin (G) using reconstitution buffer (H) which has reached room temperature. Pipette 100 µL into each well (except blank). | |
| 6. | Cover the frame and shake the wells for 1 hour at room temperature on an ELISA plate shaker (500 shakes per min). | |
| 7. | Repeat wash step 4. | |
| 8. | Pipette 100 µL of diluted SA-POD (J) into each well (except blank). | |
| 9. | Cover the frame and shake the wells for 20 minutes at room temperature on an ELISA plate shaker (500 shakes per min). | |
| 10. | Repeat wash step 4. | |
| 11. | Pipette 100 µL of TMB (L) into each well (including blank) and incubate for 20 minutes in the dark at room temperature without shaking. | |
| 12. | Pipette 50 µL of stop solution (M) into each well (including blank), cover the frame, and shake for approximately 5 seconds on an ELISA plate shaker. Ensure substrate incubations are the same for each well. | |
| 13. | Within 20 minutes, read the absorbance of each well at 450nm and 405nm using an ELISA plate reader, blanked against the well containing 100 µL of TMB (L) and 50 µL stop solution (M) only. |
RESULT ANALYSIS
Calculation of results without calibrators
Index Calculation
The index values are calculated as follows:
| Index= | test sample absorbance at 450nm | x 100 |
| reference preparation absorbance at 450nm |
The index value can also be calculated using absorbance data at 405nm.
TYPICAL RESULTS (Example only; not to be used for calculation of actual results)
| A450nm | Index value | A405nm | Index value | |
| Reference Preparation (D) | 0.728 | 100 | 0.232 | 100 |
| Negative Control (B) | 0.090 | 12 | 0.028 | 12 |
| Positive Control (C1) | 0.464 | 64 | 0.151 | 65 |
| Positive Control (C2) | 1.684 | 231 | 0.541 | 233 |
ASSAY CUT OFF
| Negative | < 45 |
| Positive | ≥ 45 |
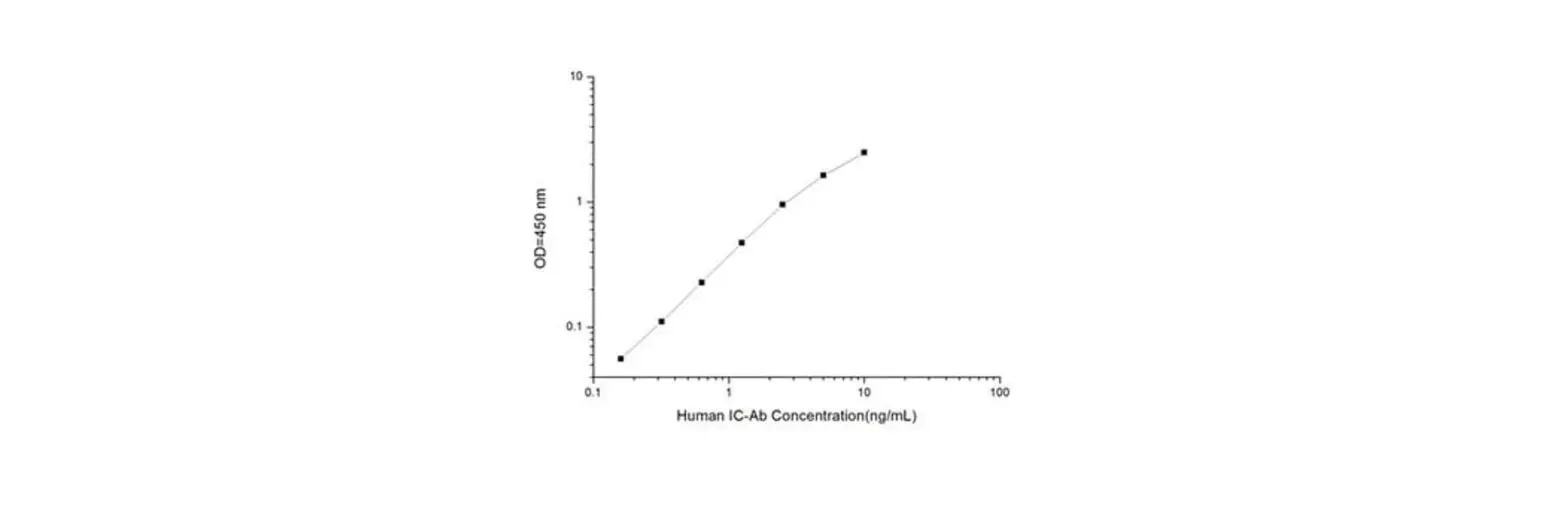
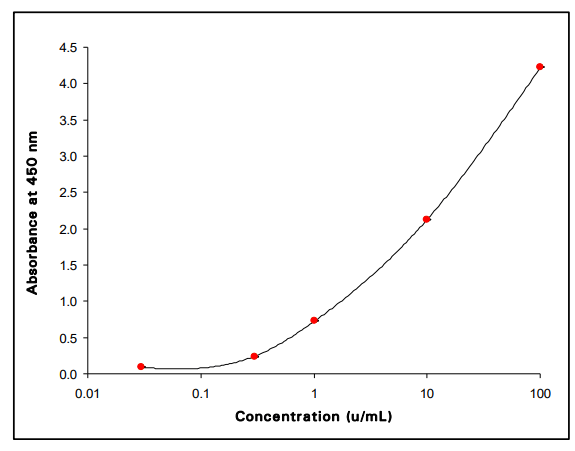
Calculation of results with calibrators (optional)
A calibration curve can be established by plotting calibrator concentration on the x-axis (log scale) against the absorbance of the calibrators on the y-axis (linear scale). The 21-OH Ab concentrations in patients’ sera can then be read off the calibration curve [plotted at RSR as a spline log/lin curve (smoothing factor = 0)]. Other data reduction systems can be used. The negative control (B) can be assigned a value of 0.03 u/mL to assist in the computer processing of assay results.
TYPICAL RESULTS (Example only; not for calculation of actual results)
| A450 nm | Conc. u/mL | A405 nm | Conc. u/mL | |
| Negative Control (B) | 0.090 | 0.028 | ||
| E1 | 0.231 | 0.3 | 0.073 | 0.3 |
| E2 | 0.728 | 1 | 0.232 | 1 |
| E3 | 2.121 | 10 | 0.679 | 10 |
| E4 | 4.223 | 100 | 1.242 | 100 |
| Positive Control (C1) | 0.464 | 0.57 | 0.151 | 0.59 |
| Positive Control (C2) | 1.684 | 5.37 | 0.541 | 5.32 |
For absorbance readings at 450nm above 3.0, the absorbance reading at 405nm can be converted to 450nm absorbance values by multiplying by the appropriate factor (3.4 in the case of equipment used at RSR).
Samples with 21-OH Ab concentrations above 100 u/mL can be diluted (e.g. 10 x and/or 100 x) in 21OH Ab negative serum. Some sera will not dilute in a linear way.
ASSAY CUT OFF
| Negative | < 0.4 u/mL |
| Positive | ≥ 0.4 u/mL |
This cut-off and the cut-off based on index value (see above) have been validated at RSR. However, each laboratory should establish its own normal and pathological reference ranges for 21-OH Ab levels. Also, it is recommended that each laboratory include its own panel of control samples in the assay.
CLINICAL EVALUATION
Clinical Specificity
Sera from 928 healthy blood donors were tested in the 21-OH Ab ELISA kit. 922 (99.4%) sera were identified as being negative for 21-OH Ab. The remaining 6 (0.6%) of healthy blood donor sera (0.59, 0.93, 1.2, 2.4, >100 and >100 u/mL) were all found to contain IgM antibodies to 21-OH.
Clinical Sensitivity
Sera from 100 patients diagnosed with autoimmune Addison’s disease were tested in the 21-OH Ab ELISA kit. 86 (86%) were identified as being positive for 21-OH Ab.
Lower Detection
Limit The negative control was assayed 20 times and the mean and standard deviation were calculated. The lower detection limit at +2 standard deviations was 0.13 u/mL, the index value was 12.
Intra Assay Precision
| Sample | Mean u/mL (n=25) | CV (%) | Mean index (n=25) | CV (%) |
| 1 | 0.30 | 2.7 | 39 | 2.2 |
| 2 | 0.89 | 6.1 | 92 | 4.8 |
| 3 | 2.0 | 6.3 | 154 | 3.5 |
| 4 | 5.4 | 18.1 | 249 | 7.3 |
| 5 | 55 | 9.9 | 512 | 2.3 |
Sample | Mean u/mL (n=20) | CV (%) | Mean index (n=20) | CV (%) |
| A | 0.39 | 4.1 | 43 | 4.9 |
| B | 1.0 | 7.4 | 102 | 4.6 |
| C | 2.7 | 17.9 | 164 | 8.9 |
| D | 10.7 | 11.5 | 284 | 6.2 |
| E | 58.7 | 14.0 | 500 | 8.4 |
Clinical Accuracy
Analysis of 185 sera from patients with autoimmune diseases other than Addison’s disease indicated no interference from autoantibodies to thyroglobulin, thyroid peroxidase, TSH receptor, glutamic acid decarboxylase, zinc transporter 8, aquaporin-4, voltage-gated potassium channel, double-stranded DNA, acetylcholine receptor or from rheumatoid factor. A serum sample from a further patient with Type 1 DM (GADAb positive) gave an index value of 409 and a concentration of 44 u/mL. This sample was assayed in RSR’s 21-OH Ab RIA kit and was positive with a 21-OH Ab concentration of 100 u/mL. A serum sample from a further patient with Type 1 DM (ZnT8 Ab positive) gave an index value of 60 and a concentration of 0.53 u/mL. This sample was assayed in the 21-OH Ab RIA and was negative. A further sample that was AChRAb positive gave an index value of 68 and a concentration of 0.61 u/mL.
Interference
No interference was observed when samples were spiked with the following materials; hemoglobin at 500 mg/dL, bilirubin at 20 mg/dL, or Intralipid up to 3000 mg/dL.
SAFETY CONSIDERATIONS
Streptavidin Peroxidase (SA-POD)
Signal word: Warning ![]()
Hazard statement(s)
H317: May cause an allergic skin reaction
Precautionary statement(s)
P280: Wear protective gloves/protective clothing/eye protection/face protection
P302 + P352: IF ON SKIN: Wash with plenty of soap and water
P333 + P313: If skin irritation or rash occurs: Get medical advice/attention
P362 + P364: Take off contaminated clothing and wash it before reuse
Peroxidase Substrate (TMB)
Signal word: Danger ![]()
Hazard statement(s)
H360: May damage fertility or the unborn child
Precautionary statement(s)
P280: Wear protective gloves/protective clothing/eye protection/face protection
P308 + P313: IF exposed or concerned: Get medical advice/attention
This kit is intended for use by professional persons only. Follow the instructions carefully. Observe expiry dates stated on the labels and the specified shelf life for coated wells, diluted and reconstituted reagents. Refer to the Safety Data Sheet for more detailed safety information. Avoid all actions likely to lead to ingestion. Avoid contact with skin and clothing. Wear protective clothing. Material of human origin used in the preparation of the kit has been tested and found non-reactive for HIV1 and 2 and HCV antibodies and HBsAg but should, nonetheless, be handled as potentially infectious. Wash hands thoroughly if contamination has occurred before leaving the laboratory. Sterilize all potentially contaminated waste, including test specimens before disposal. Material of animal origin used in the preparation of the kit has been obtained from animals certified as healthy but these materials should be handled as potentially infectious. Some components contain small quantities of sodium azide as the preservative. As with all kit components, avoid ingestion, inhalation, injection, and contact with skin, eyes, and clothing. Avoid the formation of heavy metal azides in the drainage system by flushing any kit component away with copious amounts of water.
ASSAY PLAN
| Allow all reagents and samples to reach room temperature (20 – 25°C) before use | ||
| Day 1 | Pipette: | 50 µL negative and positive controls (B and C1-2), reference preparation (D) or calibrators (if used E1-4), and patient sera (except blank) |
| Pipette: | 50 µL reaction enhancer (F) (except blank) | |
| Mix: | Shake on an ELISA plate shaker at 500 shakes/min for 1 minute | |
| Incubate: | Overnight (16 – 20 hours) at 2 – 8°C without shaking | |
| Day 2 | Aspirate/Wash: | ELISA plate (A) three times |
| Pipette: | 100 µL 21-OH-Biotin (G) reconstituted with room temperature reconstitution buffer (H) into each well (except blank) | |
| Incubate: | 1 hour at room temperature on an ELISA plate shaker at 500 shakes/min | |
| Aspirate/Wash: | ELISA plate (A) three times | |
| Pipette: | 100 µL SA-POD (J) (diluted 1:20) into each well (except blank) | |
| Incubate: | 20 minutes at room temperature on an ELISA plate shaker at 500 shakes/min | |
| Aspirate/Wash: | ELISA plate (A) three times | |
| Pipette: | 100 µL TMB (L) into each well (including blank) | |
| Incubate: | 20 minutes at room temperature in the dark (without shaking) | |
| Pipette: | 50 µL stop solution (M) into each well (including blank) and shake for 5 seconds | |
| Read absorbance at 450nm and 405nm within 20 minutes of adding stop solution | ||